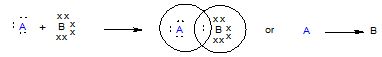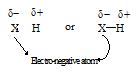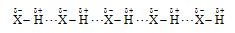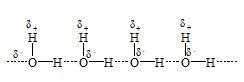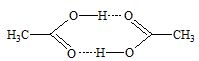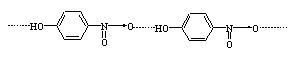-
Chemical bonding and Molecular structure
Coordinate bond, Hydrogen Bonding and Metallic Bonding
Coordinate bond
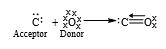
It is a special type of covalent bond in which both the shared electrons are contributed by one atom only. The atom which contributes electron pair is called the donor while the atom which accepts it is called acceptor. It may be defined as “a covalent bond in which both electrons of the shared pair are contributed by one of the two atoms”. Such a bond is also called as dative bond. A coordinate or a dative bond is established between two such atoms, one of which has a complete octet and possesses a pair of valence electrons while the other is short of a pair of electrons.
This bond is represented by an arrow (\(\rightarrow\)) pointing towards acceptor atom.
The compound consisting of the coordinate bond is termed coordinate compound. Some examples of coordinate bond formation are given below:
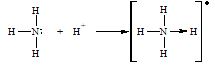
i) Formation of ammonium ion : Hydrogen ion (H+) has no electrons and thus accepts a lone pair donated by nitrogen.
ii) Formation of CO : Carbon has four valency electrons and oxygen has six. They combine to form two double bond and a coordinate bond as to achieve their octet completed.
Characteristics of Coordinate Compounds: The properties of coordinate compounds are intermediate between the properties of electrovalent compounds and covalent compounds. The main properties are described below:
i) Melting and Boiling Points: Their melting and boiling points are higher than purely covalent compounds and lower than ionic compounds.
ii) Solubility: These are sparingly soluble in polar solvents like water but readily soluble in non-polar (organic) solvents.
iii) Conductivity: Like covalent compounds, these are also bad conductors of electricity. The solutions or fused mass do not allow the passage of electricity.
Illustration:
Which of the following compound contains both ionic as well as covalent bonds only .
(a) NaCl (b) NaOH (c) CH4 (d) NH4Cl
Sol.(b) NaOH conations an ionic bond between Na+ and OH¯ and a covalent bond between Oand H.
Hydrogen Bonding
In 1920, Latimer and Rodebush introduced the idea of “hydrogen bond” to explain the nature of association in liquid state of substance like water, hydrogen fluoride, ammonia, formic acid etc. In a hydrogen compound, when hydrogen is bonded to highly electronegative atom (such as F, O, N) by a covalent bond, the electron pair is attracted towards electronegative atom so strongly that a dipole results i.e., one end carries a positive charge (H-end) and other end carries a negative charge (high electronegative atom i.e. X-end)
If a number of such molecules are brought nearer to each other, the positive end of one molecule and negative end of the other molecule will attract each other and weak electrostatic force will develop. Thus, these molecules will associate together to form a cluster of molecules.
The attractive force that binds hydrogen atom of one molecule with electronegative atom of the other molecule of the same or different substance is known as hydrogen bond.
Hydrogen bonding is of two types:
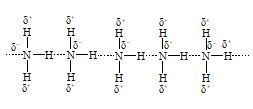
a) Intermolecular hydrogen bonding: This type of bonding results between the positive and negative ends of different molecules of the same or different substances.
Example :
i) Ammonia
ii) Water
iii) Acetic acid
This type of hydrogen bonding increases the boiling point of the compound and also its solubility in water. The increase in boiling point is due to association of several molecules of the compound.
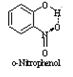
b) Intermolecular hydrogen bonding: This type of bonding results between hydrogen and an electronegative element both present in the same molecule. This type of bonding is generally present in organic compounds. Examples are o-nitro-phenol, o-hydroxyl benzoic acid, etc.
This type of bonding decreases the boiling point of the compound. The solubility of the compound also decreases. Hence compound becomes more volatile.
Properties Explained by Hydrogen Bonding
a) Strength of certain acids and bases can be explained on the basis of hydrogen bonding.
b) Solubility: An organic substance is said to be insoluble in water if it does not form hydrogen bonding with water. The organic compounds like alkanes, alkenes, ethers, etc., are insoluble in water as they do not form hydrogen bonding with water, while alcohols and acids are soluble because they readily form hydrogen bonds with water.
i) Melting and boiling points of hydrides of N, O and F. If the melting points and boiling points of the hydrides of the elements of IVA, VA, VIA and VIIA groups are plotted against the molecular weights of these hydride.
From these plots it may be seen that although in case of \(\text{Sb}{{\text{H}}_{\text{3}}},\text{ As}{{\text{H}}_{\text{3}}},\text{ P}{{\text{H}}_{\text{3}}}\) (VA group elements hydrides),\({{\text{H}}_{\text{2}}}\text{Te},\text{ }{{\text{H}}_{\text{2}}}\text{Se},\text{ }{{\text{H}}_{\text{2}}}\text{S}\) (VI A group elements hydrides) and HI, HBr, HCl (VII group elements hydrides) there is a progressive decrease in their melting points and boiling with the decrease in their molecular weights, the mp’s and b.p’s of \(\text{N}{{\text{H}}_{\text{3}}},\text{ }{{\text{H}}_{\text{2}}}\text{O}\) and HF hydrides suddenly increase with a further decrease of their molecular weights. The sudden increase in mp’s and bp’s in these hydrides is due to the inter-molecular H-bonding in between H and F in case of HF, in between H and O in case of \({{H}_{2}}O\) and in between H and N in case of \(N{{H}_{3}}\) respectively. The existence of H-bonding in these molecules gives polymerized molecules \({{(N{{H}_{3}})}_{n}}.\)Thus melting points and boiling of these molecules are suddenly raised.
Having no power to form H-bonds, the simple carbon family hydrides \((\text{Sn}{{\text{H}}_{\text{4}}},\text{ Ge}{{\text{H}}_{\text{4}}},\text{ Si}{{\text{H}}_{\text{4}}}\,\,\text{and C}{{\text{H}}_{\text{4}}})\) show a decrease in their bp’s and mp’s with the decrease in their molecular weights.
ii) Ice has less density than water. The explanation of this fact is as follows: In the crystal structure of ice, the O-atom is surrounded by four H-atoms. Two H-atoms are linked to oxygen-atom by covalent bonds as shown (by normal covalent bond) and the remaining two H-atoms are linked to oxygen-atom by two H-bonds shown by dotted lines. Thus in ice every water molecule is associated with four other water molecules by H-bonding in a tetrahedral fashion. Ice has an open cage like structure with a large empty space due to the existence of H-bonds. As ice melts at 00C, a number of H-bonds are broken down and the space between water molecules decreases so that water molecules move closer together. The density of water increases, from 00 to 40C, and at 40C it is maximum. Above 40C the increase in kinetic energy of the molecules is sufficient to cause the molecules to begin to disperse and the result is that the density decrease with increasing temperature.
Illustration :
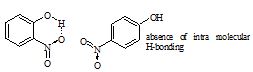
Both o-nitrophenol and p-nitrophenol has hydrogen bonding in their molecules? Explain which of the two has higher boiling point?
Sol. Both have hydrogen bonding. o-Nitrophenol has intramolecular hydrogen bonds. But due to larger distance between —NO2 and —OH group in p-nitrophenol, there is no such bonding.
However,there is intermolecular hydrogen bonding in p-nitrophenol and therefore, it exists as associated molecule. In o-nitrophenol, intramolecular hydrogen bonding is possible.
Due to associated nature of p-nitrophenol, it is less volatile and has high boiling point.
Metallic Bond:
The force that binds a metal ion to a number of electrons with in its sphere of influence is known as metallic bond.
Electron - Sea Theory (Drude - Lorentz theory)
Valency electrons are weakly bound to the kernel in metals. The valency electrons are completely delocalized and frequently exchanged between the atoms. The valence electrons belong to all the atoms, they move freely from one kernel to the other. Valence electrons form a poor or sea of electrons.
The consequences of metallic bond are
1. Metallic lusture : When light falls on metal surface, electrons get excited. When deexcited emission of visible light takes place. This is called metallic lusture.
2. Electrical cuductivity: In the presence of external field the free electrons in metal move towards a particular direction
3. Thermal conductivity:On heating the free electrons acquire energy and start moving in zigzag way transferring heat energy in all the directions.
4.Due to strong bond energy one layer of atoms slide over the other with out serious disturbance at any stage on applying force.
5. Elasticity: The property of a substance which can resist a deformation and recover to its original form after the removal of force. This is the result of metallic bond.