-
Thermal Properties of Matter
Thermal Expansion
Most substance expand when they are heated. Thermal expansion is a consequence of the change in average separation between the constituent atoms of an object. Atoms of an object can be imagined to be connected to one another by stiff springs as shown in figure. At ordinary temperatures, the atoms in a solid oscillate about their equilibrium positions with an amplitude of approximately 10–11 m.The average spacing between the atoms is about 10–10 m. As the temperature of solid increases, the atoms oscillate with greater amplitudes, as a result the average separation between them increases, consequently the object expands.
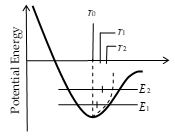
More precisely, thermal expansion arises from the asymmetrical nature of the potential energy curve.
At the atomic level, thermal expansion may be understood by considering how the potential energy of the atoms varies with distance. The equilibrium position of an atom will be at the minimum of the potential energy well if the well is symmetric. At a given temperature each atom vibrates about its equilibrium position and its average position remains at the minimum point. If the shape of the well is not symmetrical, as shown in figure, the average position of an atom will not be at the minimum point. When the temperature is raised the amplitude of the vibrations increases and the average position is located at a greater interatomic separation. This increased separation is manifested as expansion of the material.
The potential energy of an atom. Thermal arises because the ‘‘wall’’ is not symmetrical about the equilibrium position r0. As the temperature rises, the energy of the atom changes. The average position r when the energy is E2 is not the same as that when the E1.
Linear Expansion
Suppose that the temperature of a thin rod of length l is charged from T to T + \(\Delta \)T. It is found experimentally that, if \(\Delta \)T is not too large, the corresponding change in length \(\Delta\)l of the rod is directly proportional to \(\Delta\)T and l. Thus,
\(\Delta l \propto \Delta T \,\,and \,\,\Delta l \propto l\)
Introducing a proportionality constant \(\alpha\) (which is different for different materials) we may write \(\Delta\)l as \(\Delta\)l = l \(\alpha\Delta\)T …(i)
Here, the constant \(\alpha\) is called the coefficient of linear expansion of the material of the rod and its units are K–1 or [(°C)–1]. Remember that \(\Delta T = \Delta \)TC.
Actually, \(\alpha\) does depend slightly on the temperature, but its variation is usually small enough to be negligible, even over a temperature range of 100°C. We will always assume that \(\alpha\) is a constant.
Consider a solid in the form of a rod. Let its length be L0 at 0ºC. Let the rod be heated through tºC. Let its length increase to Lt. Then, the linear expansion is (Lt – L0). It is observed that the linear expansion is proportional to original length L0 and the rise in temperature t.
\(\therefore L_t – L_0 = L_0 \alpha t\)Lt = L0 (1 + \(\alpha \)t)
Also,\(\therefore\,\alpha=\frac{{{L}_{t}}-{{L}_{0}}}{{{L}_{0}}t}\)
The coefficient of linear expansion of the material of a rod is defined as the change in length per unit length per unit change in temperature.
Superficial Expansion
Let S0 be the surface area of a body at 0ºC. Let St be its surface area at tºC.
Then St – S0 = S0 \(\beta\)t
Where \(\beta \) is called coefficient of superficial expansion of the body.
Now St = S0 (1 + \(\beta\)t)
The value of \(\beta \) depends upon the nature of material and temperature.
Also,\(\beta =\frac{{{S}_{t}}-{{S}_{0}}}{{{S}_{0}}t}\)
Cubical Expansion
Because the linear dimensions of an object change with temperature, it follows that surface area and volume change as well. Just as with linear expansion, experiments show that if the temperature change\(\Delta\)T is not too great (less than 100°C or so), the increase in volume $\Delta $V is proportional to both the temperature change \(\Delta\)T and the initial volume V. Thus,
\(\Delta V\propto \Delta T\ \ \text{and}\ \ \Delta V\propto V\)
Introducing a proportionality constant \(\gamma\), we may write \(\Delta\)V as,
\(\Delta V = V \times \gamma \times\Delta \)T …(ii)
Here, \(\gamma\)is called the coefficient of volume expansion. The units of \(\gamma\) are K–1 or (°C)–1.
Let V0 be the value of a body at 0ºC. Let Vt be its volume at tºC.
Then Vt – V0 = V0 \(\gamma\)t
Now, Vt = V0 (1+ \(\gamma\)t)
Also, \(\gamma =\frac{{{V}_{t}}-{{V}_{0}}}{{{V}_{0}}t}\)
Relation between \(\gamma\) and \(\alpha\)
For an isotropic solid (which has the same value of a in all directions) \(\gamma\)= 3\(\alpha\). To see that \(\gamma \) = 3\(\alpha\) for a solid, consider a cube of length l and volume V = l3.
When the temperature of the cube is increased by dT, the side length increases by dl and the volume increases by an amount dV given by
\(dV=\left( \frac{dV}{dl} \right).dl=3{{l}^{2}}.dl\)
Now, dl = l\(\alpha\)dT
\(\therefore \) dV = 3l3\(\alpha\).dT = (3\(\alpha\))VdT
This is consistent with Eq. (ii), dV = \(\gamma \)VdT, only if
\( \gamma = 3\alpha \) …(iii)
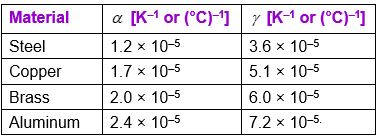
Average values of \(\alpha\) and \(\gamma\) for some materials are listed in Table. You can check the relation \(\gamma = 3\alpha\), for the materials given in the table.
Illustrations:
Illustration 1
A metallic bob weighs 50 g in air. If it is immersed in a liquid at a temperature of 25ºC, it weights 45 g. When the temperature of the liquid is raised to 100ºC. It weighs 45.1 g. Calculate the coefficient of cubical expansion of the liquid, given the coefficient of linear expansion of the metal to be 12 × 10–6 (ºC)–1.
Solution
Let \(\gamma \)m be the coefficient of cubical expansion of the metal.
Then,\({{\gamma }_{m}}=3\alpha =3\times 12\times {{10}^{-6}}{{{{(}^{0}}C)}^{-1}}=36\times {{10}^{-6}}{{{{(}^{0}}C)}^{-1}}\)
If V1 and V2 are the volumes of the metal bob at t1 = 25ºC and t2 = 100ºC respectively, then,\(\frac{{{V}_{2}}}{{{V}_{1}}}=1+{{\gamma }_{m}}({{t}_{2}}-{{t}_{1}})=1+36\times {{10}^{-6}}\times 75=1.0027\)
Now, upthrust = loss of weight of the bob in the liquid
\(\therefore {{V}_{1}}{{\rho }_{1}}=50-45=5g\,\,\,\,and\,\,{{V}_{2}}{{\rho }_{2}}=50-45.1=4.9g\,\)
Here \({{\rho }_{1}}\) and \({{\rho }_{2}}\) are the densities of the liquid at 25ºC and 100ºC respectively.
Therefore,\(\frac{{{\rho }_{1}}}{{{\rho }_{2}}}=\frac{5}{4.9}\times \frac{{{V}_{2}}}{{{V}_{1}}}=\frac{5}{4.9}\times 1.002\)
But \({{\rho }_{1}}={{\rho }_{2}}(1+75\gamma )\)
\(\gamma =\frac{\left( \frac{{{\rho }_{1}}}{{{\rho }_{2}}}-1 \right)}{75}\)
\(\gamma =2.993{{\mathbf{K}}^{\mathbf{1}}}\mathbf{or}\text{ }{{\left( {}^\circ \mathbf{C} \right)}^{\mathbf{1}}}\)
Illustration 2
The volume of a metal ball increases by 0.18% when its temperature is increased by 30ºC. What is the coefficient of linear expansion of the metal.
Solution
The increase in volume of ball is given by
\(\Delta V=V\gamma \Delta T\)
where \(\gamma\) is the coefficient of cubical expansion of the metal. Thus,
\(\frac{\Delta V}{V}=\gamma \Delta t\)
Given,
\(\frac{\Delta V}{V}=\frac{0.18}{100}\)
and \(\Delta t={{30}^{0}}C\)
Therefore,\(\gamma \,\,\,=\,\,\,\frac{\Delta V}{V\Delta t}\,\,\,\,=\,\,\,\frac{0.18}{100\,\,\times \,\,30}\,\,=\,\,6\,\,\times \,\,{{10}^{-5}}\,\,\,per{}^\text{o}C\)
Now,\(\alpha \,\,\,=\,\,\frac{\gamma }{3}\,\,\,=\,\,\,\frac{6\,\,\times \,\,{{10}^{-5}}}{3}\,\,\,\,=\,\,2\,\,\times \,\,{{10}^{-5}}\,\,\,per\,\,{}^\text{o}C\)
Stress in Objects Due to Thermal Expansion
\(F\,\,=\,\,YS\alpha \Delta T\)
The Anomalous Expansion of Water
Most liquids also expand when their temperatures increase. Their expansion can also be described by Eq. (ii). The volume expansion coefficients for liquids are about 100 times larger than those for solids.
Some substances contract when heated over a certain temperature range. The most common example is water.
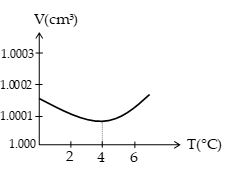
Figure shows how the volume of 1 gm of waste varies with temperature at atmospheric pressure. The volume decreases as the temperature is raised from 0°C to about 4°C, at which point the volume is a minimum and the density is a maximum (1000 kg/m3). Above 4°C, water expands with increasing temperature like most substances.
This anomalous behaviour of water causes ice to form first at the surface of a lake in a cold weather. As winter approaches, the water temperature increases initially at the surface. The water there sinks because of its increased density. Consequently, the surface reaches 0°C first and the lake becomes covered with ice. Aquatic life is able to survive the cold winter as the lake bottom remains unfrozen at a temperature of about 4°C.
Isotropic and Anisotropic Expansion
Thus, when a solid expands uniformly in all three dimensions, this is called isotropic expansion of solid. In isotropic expansion the coefficient of linear expansion \(\alpha\) remains same in all three dimensions and thus the volume coefficient of thermal expansion can be written as
\(\gamma \,\,=\,3\alpha\)
When expansion of solid is different in the three dimensions due to atomic arrangement is solid lattice structure, this is termed as anisotropic expansion of solids. In anisotropic expansion the coefficient of linear expansion of solid is different in different dimensions of solid.
If \(\alpha\)1, \(\alpha\) 2 and \(\alpha\)3 are the coefficient linear expansion along three axes then the value of \(\gamma\) is given as
\(\gamma \,\,=\,\,{{\alpha }_{1}}+{{\alpha }_{2}}+{{\alpha }_{3}}\)
The anisotropic expansion is due to the properties of a lattice structure in atomic arrangement, it can be seen only in solids not in liquids or gases.
Application of Thermal Expansion in Solids
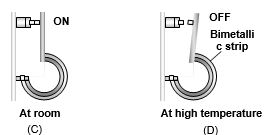
(1) Bi-metallic strip : Two strips of equal lengths but of different materials (different coefficient of linear expansion) when join together, it is called “bi-metallic strip”, and can be used in thermostat to break or make electrical contact. This strip has the characteristic property of bending on heating due to unequal linear expansion of the two metal. The strip will bend with metal of greater \(\alpha\) on outer side i.e. convex side.
(2) Effect of temperature on the time period of a simple pendulum : A pendulum clock keeps proper time at temperature \(\theta\). If temperature is increased to \(\theta'\)>\(\theta\) then due to linear expansion, length of pendulum and hence its time period will increase.
Fractional change in time period \(\frac{\Delta T}{T}=\frac{1}{2}\alpha \,\Delta \theta \)
(i) Due to increment in its time period, a pendulum clock becomes slow in summer and will lose time.
Loss of time in a time period \(\Delta T=\frac{1}{2}\alpha \,\,\Delta \theta \,\,T\)
(ii) Time lost by the clock in a day (t = 86400 sec)
\(\Delta t=\frac{1}{2}\alpha \,\,\Delta \theta \,\,t=\frac{1}{2}\alpha \,\,\Delta \theta \,\,(86400)=43200\,\alpha \,\,\Delta \theta \,\,sec\)
(iii) The clock will lose time i.e. will become slow if \({\theta }'\)>\(\theta\)(in summer)
and will gain time i.e. will become fast if \({\theta }'\)<\(\theta \) (in winter).
(iv) The gain or loss in time is independent of time period T and depends on the time interval t.
(v) Since coefficient of linear expansion (\(\alpha\)) is very small for invar, hence pendulums are made of invar to show the correct time in all seasons.
(3) Thermal stress in a rigidly fixed rod : When a rod whose ends are rigidly fixed such as to prevent expansion or contraction, undergoes a change in temperature, due to thermal expansion or contraction, a compressive or tensile stress is developed in it. Due to this thermal stress the rod will exert a large force on the supports. If the change in temperature of a rod of length L is \(\Delta\theta\) then
Thermal strain \(=\frac{\Delta L}{L}=\alpha \Delta \theta\) \(\left[ \text{As }\alpha =\frac{\Delta L}{L}\times \frac{1}{\Delta \theta } \right]\)
So Thermal stress \(=Y\alpha \Delta \theta\) \(\left[ \text{As }Y=\frac{\text{stress}}{\text{strain}} \right]\)
or Force on the supports \(F=YA\alpha \,\Delta \theta \)
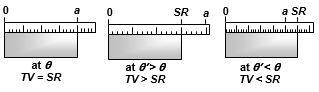
(4) Error in scale reading due to expansion or contraction : If a scale gives correct reading at temperature \(\theta\), at temperature \({\theta }'\)>\(\theta )\) due to linear expansion of scale, the scale will expand and scale reading will be lesser than true value so that,
True value = Scale reading \([1+\alpha \,({\theta }'-\theta )]\)
i.e.\(\text{TV}=SR\,[1+\alpha \,\Delta \theta ]\) with \(\Delta \theta =({\theta }'-\theta )\)
However, if \({\theta }'\)<\(\theta \), due to contraction of scale, scale reading will be more than true value, so true value will be lesser than scale reading and will still be given by above equation with \(\Delta \theta =({\theta }'-\theta )\) negative.
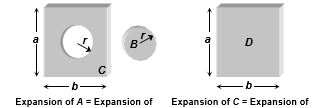
(5) Expansion of cavity : Thermal expansion of an isotropic object may be imagined as a photographic enlargement. So if there is a hole A in a plate C (or cavity A inside a body C), the area of hole (or volume of cavity) will increase when body expands on heating, just as if the hole (or cavity) were solid B of the same material. Also the expansion of area (or volume) of the body C will be independent of shape and size of hole (or cavity), i.e., will be equal to that of D.
(6) Some other application
(i)When rails are laid down on the ground, space is left between the ends of two rails.
(ii)The transmission cable are not tightly fixed to the poles.
(iii)Test tubes, beakers and crucibles are made of Pyrex-glass or silica because they have very low value of coefficient of linear expansion.
(iv)The iron rim to be put on a cart wheel is always of slightly smaller diameter than that of wheel.
(v)A glass stopper jammed in the neck of a glass bottle can be taken out by warming the neck of the bottle