-
Thermal Properties of Matter
Ideal Gas Equation and Absolute Temperature
Concept of an Ideal gas
A gas has no shape and size and can be contained in a vessel of any size or shape. It expands indefinitely and uniformly to fill the available space. It exerts pressure on its surroundings.
The gas whose molecules are point masses (mass without volume) and do not attract each other are called ideal or perfect gases. It is a hypothetical concept which can’t exist in reality. The gases such as hydrogen, oxygen or helium which cannot be liquefied easily are called permanent gases. An actual gas behaves as ideal gas most closely at low pressure and high temperature.
Gas Laws
Assuming permanent gases to be ideal, through experiments, it was established that gases irrespective of their nature obey the following laws:
Boyle’s Law
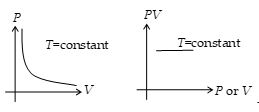
According to this law, for a given mass of a gas the volume of a gas at constant temperature (called isothermal process) is inversely proportional to its pressure, i.e.,
\(V\propto \frac{1}{P}\) (T = constant)
or PV = constant
or PiVi = PfVf
Thus, P-V graph in an isothermal process is a rectangular hyperbola. Or PV versus P or V graph is a straight line parallel to P or V axis.
Charle’s Law
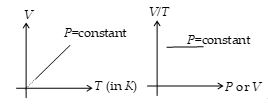
According to this law, for a given mass of a gas the volume of a gas at constant pressure (called isobaric process) is directly proportional to its absolute temperature, i.e.,
\(V\propto T\) (P = constant)
or \(\frac{V}{T}=\text{constant}\)
or \(\frac{{{V}_{i}}}{{{T}_{i}}}=\frac{{{V}_{f}}}{{{T}_{f}}}\)
Thus, V–T graph in an isobaric process is a straight line passing through origin. Or V/T versus V or T graph is straight line parallel to V or T axis.
Gay Lussac’s Law or Pressure Law
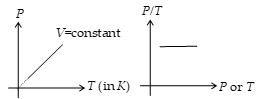
According to this law, for a given mass of a gas the pressure of a gas at constant volume (called isochoric process) is directly proportional to its absolute temperature, i.e.,
\(P\propto T\) (V = constant)
or \(\frac{P}{T}=\text{constant}\)
or \(\frac{{{P}_{i}}}{{{T}_{i}}}=\frac{{{P}_{f}}}{{{V}_{f}}}\)
Thus, P–T graph in an isochoric process is a straight line passing through origin or P/T versus P or T graph is straight line parallel to P or T axis.
Avogadro’s Law
According to this law, at same temperature and pressure equal volumes of all gases contain equal number of molecules.
Ideal Gas Equation
All the above four laws can be written in one single equation known as ideal gas equation. According to this equation.
\(PV=nRT=\frac{m}{M}RT\)
In this equation n = number of moles of the gas
\(=\frac{m}{M}\)
m = total mass of the gas,
M = molecular mass of the gas
And R = Universal gas constant
= 8.31 J/mol-K = 2.0 cal/mol-K
The above four laws can be derived from this single equation. For example, for a given mass of a gas (m = constant)
PV = constant at constant temperature (Boyle’s law)
\(\frac{P}{T}=\) constant at constant volume (Pressure law)
\(\frac{V}{T}=\) constant at constant pressure (Charle’s law)
and if P, V and T are constants then n = constant for all gases.
And since, equal number of moles contain equal number of molecules. So, at constant pressure, volume and temperature all gases will contain equal number of molecules. Which is nothing but Avogadro’s law.
Degrees of Freedom (f)
The term degree of freedom refers to the number of possible independent ways in which a system can have energy.
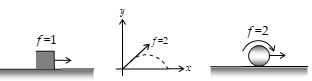
For example : In figure (a) block has one degree of freedom, because it is confined to move in a straight line and has only one translational degree of freedom.
In figure (b), the projectile has two degrees of freedom because it is confined to move in a plane and so it has two translational degrees of freedom.
In figure (c), the sphere has two degrees of freedom one rotational and another translational. Similarly, a particle free to move in space will have three translational degrees of freedom.
Degree of freedom of gas molecules
A gas molecule can have following types of energies:
(i) translational kinetic energy
(ii) rotational kinetic energy
(iii)vibrational energy (potential + kinetic)
Vibrational Energy
The forces between different atoms of a gas molecule may be visualized by imagining every atom as being connected to its neighbors by springs. Each atom can vibrate along the line joining the atoms. Energy associated with this is called vibrational energy.
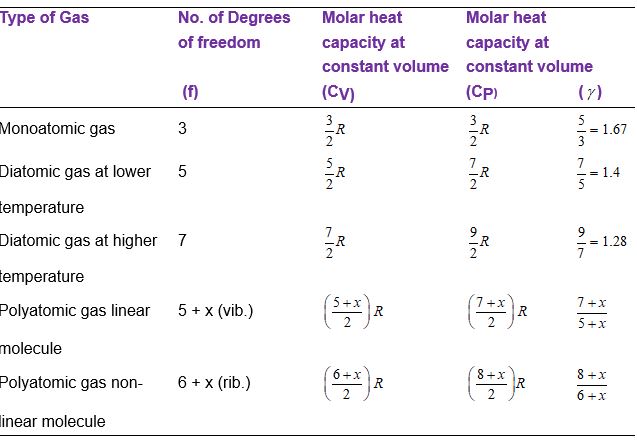
Degree of freedom of monoatomic gas
A monoatomic gas molecule (like He) consists of a single atom. It can have translational motion in any direction in space. Thus, it has 3 translational degrees of freedom.
f = 3 (all translational)
A can also rotate but due to its small moment of inertia, rotational kinetic energy is neglected.
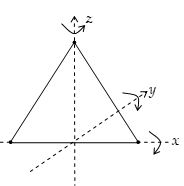
Degree of freedom of a diatomic and linear polyatomic gas
The molecules of a diatomic and linear polyatomic gas (like O2, CO2 and H2) cannot only move bodily but also rotate about any one of the three co-ordinate axes as shown in figure. However, its moment of inertia about the axis joining the two atoms (x-axis) is negligible. Hence, it can have only two rotational degrees of freedom. Thus, a diatomic molecule has 5 degrees of freedom : 3 translational and 2 rotational. At sufficiently
High temperatures it has vibrational energy as well providing it two more degrees of freedom (one vibrational kinetic energy and another vibrational potential energy). Thus, at high temperatures a diatomic molecule has 7 degrees of freedom, 3 translational, 2 rotational and 2 vibrational. Thus,
f = 5
(3 translational + 2 rotational) at room temperatures
and f = 7
(3 translational + 2 rotational + 2 vibrational) at high temperatures
Degree of freedom of nonlinear polyatomic gas
A nonlinear polyatomic molecule (such as NH3) can rotate about any of three co-ordinate axes. Hence, it has 6 degrees of freedom 3 translational and 3 rotational. At room temperatures a polyatomic gas molecule has vibrational energy greater than that of a diatomic gas. But at high enough temperatures it is also significant. So it has 8 degrees of freedom 3 rotational, 3 translational and 2 vibrational. Thus, f = 6
(3 translational + 3 rotational) at room temperatures
and f = 8
(3 translational + 3 rotational + 2 vibrational) at high temperatures
Degree of freedom of a solid
An atom in a solid has no degrees of freedom for translational and rotational motion. At high temperatures due to vibration along 3 axes it has 3 × 2 = 6 degrees of freedom.
f = 6 (all vibrational) at high temperatures
Internal energy of an Ideal Gas
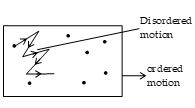
Suppose a gas is contained in a closed vessel as shown in figure. If the container as a whole is moving with some speed, then this motion is called the ordered motion of the gas. Source of this motion is some external force. The zig zag motion of gas molecules within the vessel is known as the disordered motion. This motion is directly related to the temperature of the gas. As the temperature is increased, the disordered motion of the gas molecules gets fast. The internal
energy (U) of the gas is concerned only with its disordered motion. It is in no way concerned with its ordered motion. When the temperature of the gas is increased, its disordered motion and hence its internal energy is increased.
Intermolecular forces in an ideal gas is zero. Thus, P.E. due to intermolecular forces of an ideal gas is zero. A monoatomic gas is having a single atom. Hence its vibrational energy is zero. For dia and polyatomic gases vibrational energy is significant only at high temperatures. So, they also have only translational and rotational K.E. We may thus conclude that at room temperature the internal energy of an ideal gas (whether it is mono, dia or poly) consists of only translational and rotational K.E. Thus,
U (of an ideal gas) = KT + KR at room temperatures.
Later in the next article we will see that KT (translational K.E.) and KR (rotational K.E.) depends on T only. They are directly proportional to the absolute temperature of the gas. Thus, internal energy of an ideal gas depends only on its absolute temperature (T) and is directly proportional to T. Or, U\(\propto\)T
Law of Equipartition of Energy
An ideal gas is just like an ideal father. As an ideal father distributes whole of its assets equally among his children. Same is the case with an ideal gas. It distributes its internal energy equally in all degrees of freedom. In each degree of freedom energy of one mole of an ideal gas is \(\frac{1}{2}RT\) where T is the absolute temperature of the gas. Thus, if f be the number of degrees of freedom, the internal energy of 1 mole of the gas will be \(\frac{f}{2}\)RT or internal energy of n moles of the gas will be \(\frac{n}{2}fRT\). Thus,
\(U=\frac{n}{2}fRT\) …(i)
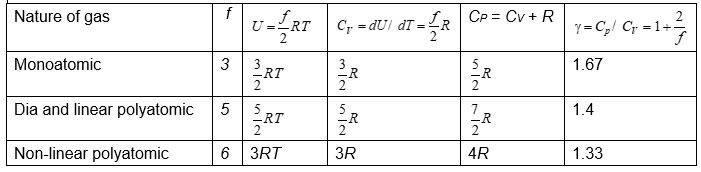
For a monoatomic gas, f = 3.
Therefore,\(U=\frac{3}{2}RT\) (for 1 mole of monoatomic gas)
For a dia and linear polyatomic gas at low temperatures, f = 5, so,
\(U=\frac{5}{2}RT\) (for 1 mole)
and for nonlinear polyatomic gas at low temperatures, f = 6, so
\(U=\frac{6}{2}RT=3RT\) (for 1 mole)
Molar Heat Capacity
‘‘Molar heat capacity C is the heat required to raise the temperature of 1 mole of a gas by 1°C (or 1 K).’’ Thus,\(C=\frac{\Delta Q}{n\Delta T}\ \ \text{or}\ \ \ \Delta Q=nC\Delta T\)
For a gas the value of C depends on the process through which its temperature is raised.
For example, in an isothermal process \(\Delta\)T’ = 0 or Ciso = \(\infty\). In an adiabatic process (we will discuss it later) \(\Delta\)Q = 0. Hence, Cadi = 0. Thus, molar heat capacity of a gas varies from 0 to \(\infty\) depending on the process. In general experiments are made either at constant volume or at constant pressure. In case of solids and liquids, due to small thermal expansion, the difference in measured values of molar heat capacities is very small and is usually neglected. However, in case of gases molar heat capacity at constant volume CV is quite different from that at constant pressure Cp. Later in the next chapter we will derive the following relations, for an ideal gas
\({{C}_{V}}=\frac{dU}{dT}=\frac{f}{2}R=\frac{R}{\gamma -1}\)
\({{C}_{P}}={{C}_{V}}+R\)
\(\gamma =\frac{{{C}_{P}}}{{{C}_{V}}}=1+\frac{2}{f}\)
Here U is the internal energy of one mole of the gas. He most general expression for C in the process PVx = constant is,
\(C=\frac{R}{\gamma -1}+\frac{R}{1-x}\) (we will derive it later)
For example : For isobaric process P = constant or x = 0 and
\(C={{C}_{p}}=\frac{R}{\gamma -1}+R={{C}_{V}}+R\)
For isothermal process, PV = constant or x = 1
\(\therefore\) C =\(\infty\) and for adiabatic process PV’ = constant or x = \(\gamma\)
\(\therefore\) C = 0
Values of f, U, CV, CP and \(\gamma\) for different gases are shown in table.