-
Thermal Properties of Matter
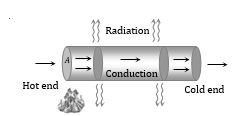
Conduction
The process of transmission of heat energy in which the heat is transferred from one particle to other particle without dislocation of the particle from their equilibrium position is called conduction.
(1) Heat flows from hot end to cold end. Particles of the medium
simply oscillate but do not leave their place.
(2) Medium is necessary for conduction
(3) It is a slow process
(4) The temperature of the medium increases through which heat flows
(5) Conduction is a process which is possible in all states of matter.
(6) When liquid and gases are heated from the top, they conduct heat from top to bottom.
(7) In solids, only conduction takes place
(8) In non-metallic solids and fluids, the conduction takes place only due to vibrations of molecules, therefore they are poor conductors.
(9) In metallic solids free electrons carry the heat energy, therefore they are good conductor of heat.
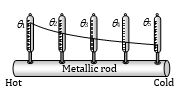
Conduction in Metallic Rod
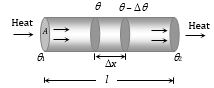
When one end of a metallic rod is heated, heat flows by conduction from the hot end to the cold end.
(1) Variable state : In this state Temperature of every part of the rod increases
Heat received by each cross-section of the rod from hotter end used in three ways.
(i) A part increases temperature of itself.
(ii) Another part transferred to neighboring cross-section.
(iii) Remaining part radiates.
· \(\theta\)1 > \(\theta\)2 > \(\theta\)3 > \(\theta\)4 > \(\theta\)5
· \(\theta\) \(\rightarrow\) Changing
(2) Steady state: After some time, a state is reached when the temperature of every cross-section of the rod becomes constant. In this state, no heat is absorbed by the rod. The heat that reaches any cross-section is transmitted to the next except that a small part of heat is lost to surrounding from the sides by convection & radiation. This state of the rod is called steady state.
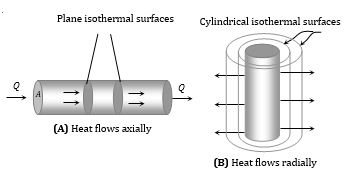
(3) Isothermal surface: Any surface (within a conductor) having its all points at the same temperature, is called isothermal surface. The direction of flow of heat through a conductor at any point is perpendicular to the isothermal surface passing through that point.
(4) Temperature gradient (T.G.): The rate of change of temperature with distance between two isothermal surfaces is called temperature gradient. Hence
(i) Temperature gradient = \(\frac{-\Delta \theta }{\Delta x}\)
(ii) The negative sign show that temperature \(\theta\) decreases as the distance x increases in the direction of heat flow.
(iii) For uniform temperature fall \(\frac{{{\theta }_{1}}-{{\theta }_{2}}}{l}=\frac{\Delta \theta }{\Delta x}\)
(iv) Unit: K/m or °C/m (S.I.) and Dimensions \([{{L}^{-1}}\theta ]\)
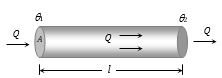
(5) Law of thermal conductivity: Consider a rod of length l and area of cross-section A whose faces are maintained at temperature \(\theta\)1 and \(\theta\)2 respectively. The curved surface of rod is kept insulated from surrounding to avoid leakage of heat
(i) In steady state the amount of heat flowing from one face to the other face in time t is given by \(Q=\frac{KA({{\theta }_{1}}-{{\theta }_{2}})\,t}{l}\) where K is coefficient of thermal conductivity of material of rod.
(ii) Rate of flow of heat i.e. heat current \(\frac{Q}{t}=H=\frac{KA({{\theta }_{1}}-{{\theta }_{2}})\,}{l}\)
(iii) In case of non-steady state or variable cross-section, a more general equation can be used to solve problems.
\(\frac{dQ}{dt}=-\,KA\frac{d\theta }{dx}\)
(6) More about K : It is the measure of the ability of a substance to conduct heat through it.
(i) Units : Cal/cm-sec oC (in C.G.S.), kcal/m-sec-K (in M.K.S.) and W/m- K (in S.I.). Dimension : \([ML{{T}^{-3}}{{\theta }^{-1}}]\)
(ii) The magnitude of K depends only on nature of the material.
(iii) Substances in which heat flows quickly and easily are known as good conductor of heat. They possess large thermal conductivity due to large number of free electrons e.g. Silver, brass etc. For perfect conductors,\(K=\infty \).
(iv) Substances which do not permit easy flow of heat are called bad conductors. They possess low thermal conductivity due to very few free electrons e.g. Glass, wood etc. and for perfect insulators, \(K=0\).
(v) The thermal conductivity of pure metals decreases with rise in temperature but for alloys thermal conductivity increases with increase of temperature.
(vi) Human body is a bad conductor of heat (but it is a good conductor of electricity).
(7) Decreasing order of conductivity: For some special cases it is as follows
(a) \({{K}_{Ag}}\)>\({{K}_{Cu}}\)>\({{K}_{Al}}\)
(b) \({{K}_{Solid}}\)>\({{K}_{Liquid}}\)>\({{K}_{Gas}}\)
(c) \({{K}_{Metals}}\)>\({{K}_{Non-metals}}\)
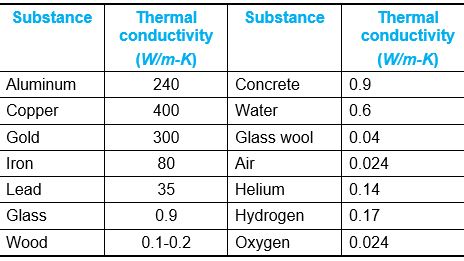
Table: Thermal conductivity of some material
(7) Relation between temperature gradient and thermal conductivity : In steady state, rate of flow of heat \(\frac{dQ}{dt}=-KA\frac{d\theta }{dx}\)= – KA \(\times\) (T.G.)
\(\Rightarrow\) (T.G.)\(\propto \frac{1}{\mathbf{K}} (\frac{dQ}{dt}\)= constant)
Temperature difference between the hot end and the cold end in steady state is inversely proportional to K, i.e. in case of good conductors’ temperature of the cold end will be very near to hot end. In ideal conductor where K =\(\infty\), temperature difference in steady state will be zero.
(8) Thermal resistance (R) : The thermal resistance of a body is a measure of its opposition to the flow of heat through it.
It is defined as the ratio of temperature difference to the heat current (= Rate of flow of heat)
(i) Hence \(R=\frac{{{\theta }_{1}}-{{\theta }_{2}}}{H}=\frac{{{\theta }_{1}}-{{\theta }_{2}}}{KA({{\theta }_{1}}-{{\theta }_{2}})/l}=\frac{l}{KA}\)
(ii) Unit : \(^{o}C\times sec/cal\) or \(K\times sec/kcal\) and Dimension : \([{{M}^{-1}}{{L}^{-2}}{{T}^{3}}\theta ]\)
(9) Wiedmann-Franz law: At a given temperature T, the ratio of thermal conductivity to electrical conductivity is constant i.e., \((K/\sigma T)\)= constant, i.e., a substance which is a good conductor of heat (e.g., silver) is also a good conductor of electricity. Mica is an exception to above law.
(10) Thermometric conductivity or diffusivity: It is a measure of rate of change of temperature (with time) when the body is not in steady state (i.e., in variable state)
It is defined as the ratio of the coefficient of thermal conductivity to the thermal capacity per unit volume of the material. Thermal capacity per unit volume =\(\frac{mc}{V}= \rho \,c\)
(\(\rho\) = density of substance) \(\Rightarrow\) Diffusivity (D) = \(\frac{K}{\rho \,c}\)
Unit : m2/sec and Dimension : \([{{L}^{2}}{{T}^{-1}}]\)
Table: Electrical Analogy for Thermal Conduction
Applications of Conductivity in Daily Life
(1) Cooking utensils are provided with wooden handles, because wood is a poor conductor of heat. The hot utensils can be easily handled from the wooden handles and our hands are saved from burning.
(2) We feel warmer in a fur coat. The air enclosed in the fur coat being bad conductor heat does not allow the body heat to flow outside. Hence, we feel warmer in a fur coat.
(3) Eskimos make double walled houses of the blocks of ice. Air enclosed in between the double walls prevents transmission of heat from the house to the cold surroundings.
For exactly the same reason, two thin blankets are warmer than one blanket of their combined thickness. The layer of air enclosed in between the two blankets makes the difference
(4) Wire gauze is placed over the flame of Bunsen burner while heating the flask or a beaker so that the flame does not go beyond the gauze and hence there is no direct contact between the flame and the flask. The wire gauze being a good conductor of heat, absorb the heat of the flame and transmit it to the flask.
Davy's safety lamp has been designed on this principle. The gases in the mines burn inside the gauze placed around the flame of the lamp. The temperature outside the gauze is not high, so the gases outside the gauze do not catch fire.
(5) Birds often swell their feathers in winter. By doing so, they enclose more air between their bodies and the feathers. The air, being bad conductor of heat prevents the out flow of their body heat. Thus, birds feel warmer in winter by swelling their feathers.
Combination of Metallic Rods
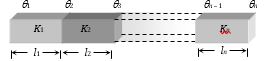
(1) Series combination: Let n slabs each of cross-sectional area A, lengths \({{l}_{1}},\,{{l}_{2}},\,{{l}_{3}},......{{l}_{n}}\) and conductivities \({{K}_{1}},\,{{K}_{2}},\,{{K}_{3}}......{{K}_{n}}\) respectively be connected in the series.
(i) Heat current: Heat current is the same in all the conductors. i.e.,\(\frac{Q}{t}={{H}_{1}}={{H}_{2}}={{H}_{3}}.........={{H}_{n}}\)
\(\frac{{{K}_{1}}A({{\theta }_{1}}-{{\theta }_{2}})}{{{l}_{1}}}=\frac{{{K}_{2}}A({{\theta }_{2}}-{{\theta }_{3}})}{{{l}_{2}}}=\frac{{{K}_{n}}A({{\theta }_{n-1}}-{{\theta }_{n}})}{{{l}_{n}}}\)
(ii) Equivalent thermal resistance:\(R={{R}_{1}}+{{R}_{2}}+.....{{R}_{n}}\)
(iii) Equivalent thermal conductivity: It can be calculated as follows
From \({{R}_{S}}={{R}_{1}}+{{R}_{2}}+{{R}_{3}}+...\)
\(\frac{{{l}_{1}}+{{l}_{2}}+...{{l}_{n}}}{{{K}_{s}}}=\frac{{{l}_{1}}}{{{K}_{1}}A}+\frac{{{l}_{2}}}{{{K}_{2}}A}+....+\frac{{{l}_{n}}}{{{K}_{n}}A}\)
\(\Rightarrow{{K}_{s}}=\frac{{{l}_{1}}+{{l}_{2}}+......\,{{l}_{n}}}{\frac{{{l}_{1}}}{{{K}_{1}}}+\frac{{{l}_{2}}}{{{K}_{2}}}+........\frac{{{l}_{n}}}{{{K}_{n}}}}\)
(a) For n slabs of equal length \({{K}_{s}}=\frac{n}{\frac{1}{{{K}_{1}}}+\frac{1}{{{K}_{2}}}+\frac{1}{{{K}_{3}}}+.....\frac{1}{{{K}_{n}}}}\)
(b) For two slabs of equal length, \({{K}_{s}}=\frac{2{{K}_{1}}{{K}_{2}}}{{{K}_{1}}+{{K}_{2}}}\)
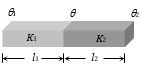
(iv) Temperature of interface of composite bar : Let the two bars are arranged in series as shown in the figure.
Then heat current is same in the two conductors.
i.e.,\(\frac{Q}{t}=\frac{{{K}_{1}}A({{\theta }_{1}}-\theta )}{{{l}_{1}}}=\frac{{{K}_{2}}A(\theta -{{\theta }_{2}})}{{{l}_{2}}}\)
By solving we get \(\theta =\frac{\frac{{{K}_{1}}}{{{l}_{1}}}{{\theta }_{1}}+\frac{{{K}_{2}}}{{{l}_{2}}}{{\theta }_{2}}}{\frac{{{K}_{1}}}{{{l}_{1}}}+\frac{{{K}_{2}}}{{{l}_{2}}}}\)
(a) If l1 = l2 then \(\theta =\frac{{{K}_{1}}{{\theta }_{1}}+{{K}_{2}}{{\theta }_{2}}}{{{K}_{1}}+{{K}_{2}}}\)
(b) If K1 = K2 and l1 = l2 then \(\theta =\frac{{{\theta }_{1}}+{{\theta }_{2}}}{2}\)
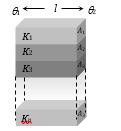
(2) Parallel Combination: Let n slabs each of length l, areas \({{A}_{1}},{{A}_{2}},{{A}_{3}},.....{{A}_{n}}\) and thermal conductivities \({{K}_{1}},{{K}_{2}},{{K}_{3}},.....{{K}_{n}}\) are connected in parallel then
(i) Equivalent resistance :\(\frac{1}{{{R}_{s}}}=\frac{1}{{{R}_{1}}}+\frac{1}{{{R}_{2}}}+\frac{1}{{{R}_{3}}}+.....\frac{1}{{{R}_{n}}}\)
For two slabs \({{R}_{s}}=\frac{{{R}_{1}}{{R}_{2}}}{{{R}_{1}}+{{R}_{2}}}\)
(ii) Temperature gradient : Same across each slab.
(iii) Heat current : in each slab will be different. Net heat current will be the sum of heat currents through individual slabs. i.e.,\(H={{H}_{1}}+{{H}_{2}}+{{H}_{3}}+....{{H}_{n}}\)
\(\frac{K({{A}_{1}}+{{A}_{2}}+.....+{{A}_{n}})\,({{\theta }_{1}}-{{\theta }_{2}})}{l}\) = \(\frac{{{K}_{1}}{{A}_{1}}({{\theta }_{1}}-{{\theta }_{2}})}{l}+\frac{{{K}_{2}}{{A}_{2}}({{\theta }_{1}}-{{\theta }_{2}})}{l}+...+\frac{{{K}_{n}}{{A}_{n}}\,({{\theta }_{1}}-{{\theta }_{2}})}{l}\)
\(\Rightarrow K=\frac{{{K}_{1}}{{A}_{1}}+{{K}_{2}}{{A}_{2}}+{{K}_{3}}{{A}_{3}}+.....{{K}_{n}}{{A}_{n}}}{{{A}_{1}}+{{A}_{2}}+{{A}_{3}}+.....{{A}_{n}}}\)
(a) For n slabs of equal area \(K=\frac{{{K}_{1}}+{{K}_{2}}+{{K}_{3}}+.....{{K}_{n}}}{n}\)
(b) For two slabs of equal area \(K=\frac{{{K}_{1}}+{{K}_{2}}}{2}\).
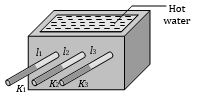
Ingen-Hauz Experiment
It is used to compare thermal conductivities of different materials. If \({{l}_{1}}, {{l}_{2}}\) and l3 are the lengths of wax melted on rods as shown in the figure, then the ratio of thermal conductivities is \({{K}_{1}}:{{K}_{2}}:{{K}_{3}}=l_{1}^{2}:l_{2}^{2}:l_{3}^{2}\)
\(\Rightarrow\) Thermal conductivity (K) \(\propto \)(Melted length l )2
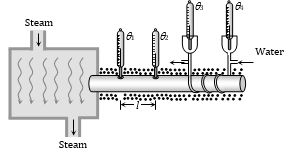
Searle's Experiment
It is a method of determination of K of a metallic rod.
(1) In this experiment a temperature difference \(({{\theta }_{1}}-{{\theta }_{2}})\) is maintained across a rod of length l and area of cross section A. If the thermal conductivity of the material of the rod is K, then the amount of heat transmitted by the rod from the hot end to the cold end in time t is given by,\(Q=\frac{KA({{\theta }_{1}}-{{\theta }_{2}})\,t}{l}\) ......(i)
(2) In Searle's experiment, this heat reaching the other end is utilized to raise the temperature of certain amount of water flowing through pipes circulating around the other end of the rod. If temperature of the water at the inlet is \({{\theta }_{3}}\) and at the outlet is \({{\theta }_{4}}\), then the amount of heat absorbed by water is given by, \(Q=mc({{\theta }_{4}}-{{\theta }_{3}})\) ......(ii)
(3) Where, m is the mass of the water which has absorbed this heat and temperature is raised and c is the specific heat of the water
Equating (i) and (ii), K can be determined i.e., \(K=\frac{mc({{\theta }_{4}}-{{\theta }_{3}})\,l}{A\,({{\theta }_{1}}-{{\theta }_{2}})\,t}\)
(4) In numericals we may have the situation where the amount of heat travelling to the other end may be required to do some other work e.g.,it may be required to melt the given amount of ice. In that case equation (i) will have to be equated to mL.
i.e.\(mL=\frac{KA({{\theta }_{1}}-{{\theta }_{2}})\,t}{l}\)
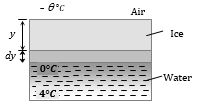
Growth of Ice on Lake
(1) Water in a lake starts freezing if the atmospheric temperature drops below \({{0}^{o}}C\). Let y be the thickness of ice layer in the lake at any instant t and atmospheric temperature is \(-{{\theta }^{o}}C\).
(2) The temperature of water in contact with lower surface of ice will be zero.
(3) If A is the area of lake, heat escaping through ice in time dt is \(d{{Q}_{1}}=\frac{KA[0-(-\theta )]\,dt}{y}\)
(4) Suppose the thickness of ice layer increases by dy in time dt, due to escaping of above heat. Then \(d{{Q}_{2}}=mL=\rho (dy\,A)\,L\)
(5) As \(d{{Q}_{1}}=d{{Q}_{2}}\), hence, rate of growth of ice will be \((dy/dt)=(K\theta /\rho Ly)\)
So, the time taken by ice to grow to a thickness y is
\(t=\frac{\rho L}{K\theta }\int_{\,0}^{\,y}{\,y\,dy}=\frac{\rho L}{2K\theta }{{y}^{2}}\)
(6)If the thickness is increased from \({{y}_{1}}\) to \({{y}_{2}}\) then time taken \(t=\frac{\rho L}{K\theta }\int_{\,{{y}_{1}}}^{\,{{y}_{2}}}{\,ydy}=\frac{\rho L}{2K\theta }(y_{2}^{2}-y_{1}^{2})\)
(7) Take care and do not apply a negative sign for putting values of temperature in formula and also do not convert it to absolute scale.
(8) Ice is a poor conductor of heat, therefore the rate of increase of thickness of ice on ponds decreases with time.
(9) It follows from the above equation that time taken to double and triple the thickness, will be in the ratio of
\({{t}_{1}}:{{t}_{2}}:{{t}_{3}}::{{1}^{2}}:{{2}^{2}}:{{3}^{2}},\) i.e.,\({{t}_{1}}:{{t}_{2}}:{{t}_{3}}::1:4:9\)
(10) The time intervals to change the thickness from 0 to y, from y to 2y and so on will be in the ratio
\(\Delta {{t}_{1}}:\Delta {{t}_{2}}:\Delta {{t}_{3}}::({{1}^{2}}-{{0}^{2}}):({{2}^{2}}-{{1}^{2}}):({{3}^{2}}:{{2}^{2}})\)
\(\Rightarrow\Delta {{t}_{1}}:\Delta {{t}_{2}}:\Delta {{t}_{3}}::1:3:5\)