-
Explanation of Valence Bond Theory
Expalnation of Valence Bond Theory
As already explained, Lewis concept is unable to explain the shapes of molecules. This theory provides a simple procedure to predict the shapes of covalent molecules. Sidgwick
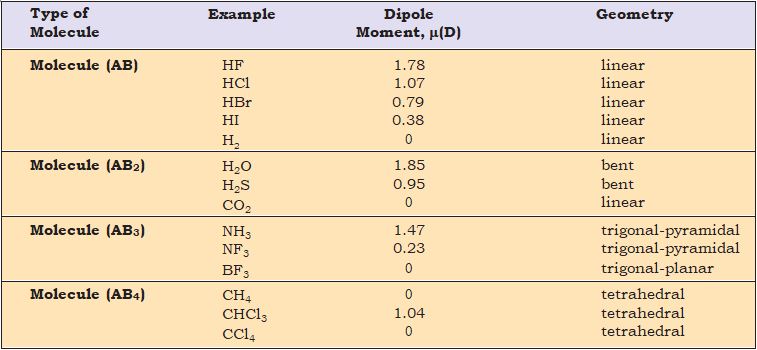
Table 4.5 Dipole Moments of Selected Molecules
and Powell in 1940, proposed a simple theory based on the repulsive interactions of the electron pairs in the valence shell of the atoms. It was further developed and redefined by Nyholm and Gillespie (1957).
The main postulates of VSEPR theory are as follows:
1. The shape of a molecule depends upon the number of valence shell electron pairs (bonded or nonbonded) around the central atom.
2. Pairs of electrons in the valence shell repel one another since their electron clouds are negatively charged.
3. These pairs of electrons tend to occupy such positions in space that minimise repulsion and thus maximise distance between them.
4. The valence shell is taken as a sphere with the electron pairs localising on the spherical surface at maximum distance from one another.
5. A multiple bond is treated as if it is a single electron pair and the two or three electron pairs of a multiple bond are treated as a single super pair.
6. Where two or more resonance structures can represent a molecule, the VSEPR model is applicable to any such structure.
The repulsive interaction of electron pairs decrease in the order:
Lone pair (lp) – Lone pair (lp) > Lone pair (lp)
– Bond pair (bp) > Bond pair (bp) – Bond pair (bp)
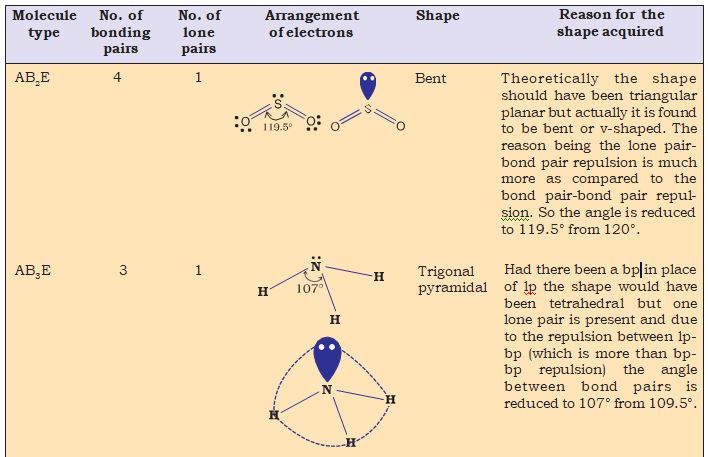
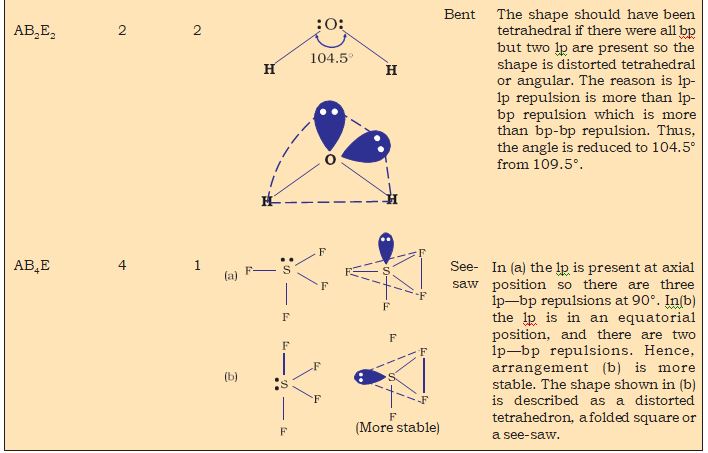
Nyholm and Gillespie (1957) refined the VSEPR model by explaining the important difference between the lone pairs and bonding pairs of electrons. While the lone pairs are localised on the central atom, each bonded pair is shared between two atoms. As a result, the lone pair electrons in a molecule occupy more space as compared to the bonding pairs of electrons.This results in greater repulsion between lone pairs of electrons as compared to the lone pair - bond pair and bond pair - bond pair repulsions.These repulsion effects result in deviations from idealised shapes and alterations in bond angles in molecules.
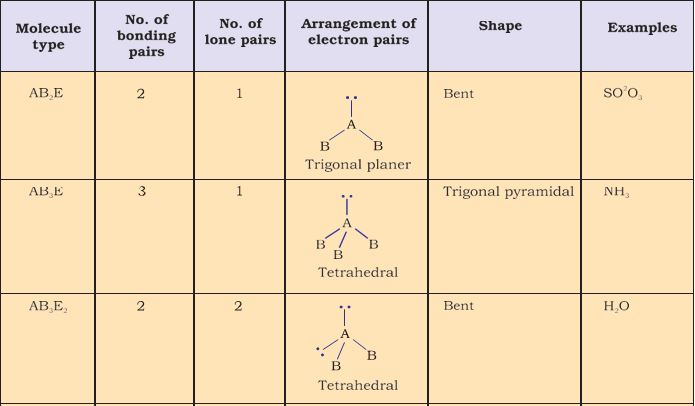
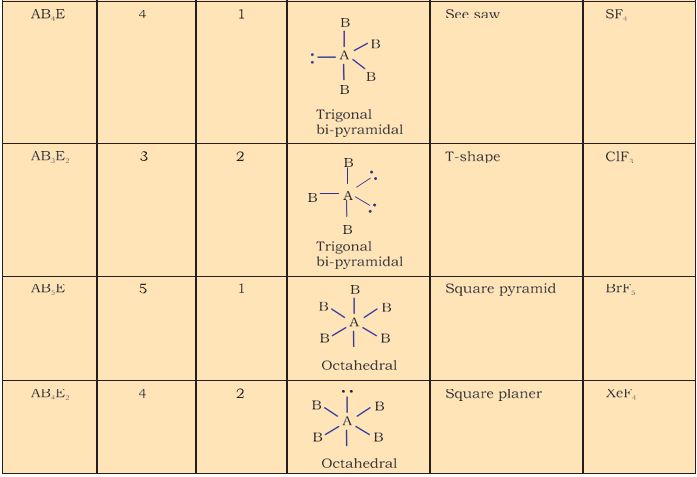
For the prediction of geometrical shapes of molecules with the help of VSEPR theory, it is convenient to divide molecules into two categories as (i) molecules in which the central atom has no lone pair and (ii) molecules in which the central atom has one or more lone pairs.
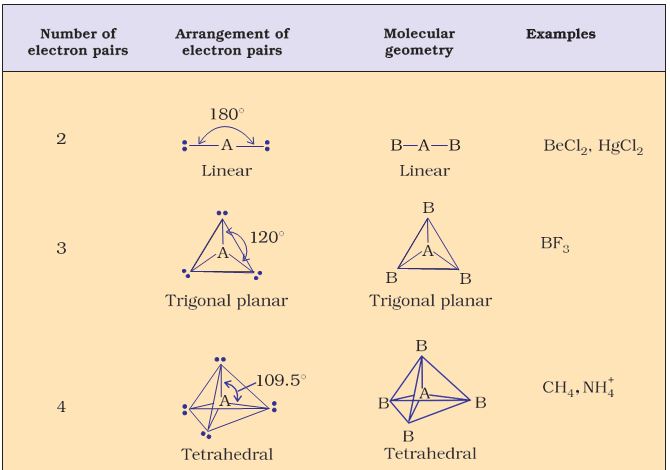
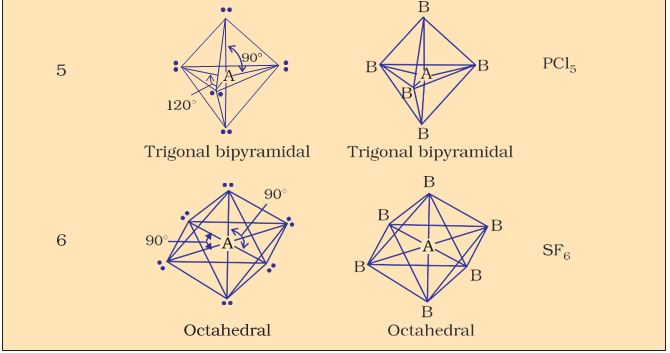
Table 4.6 (page114) shows the arrangement of electron pairs about a central atom A (without any lone pairs) and geometries of some molecules/ions of the type AB. Table 4.7 (page 115) shows shapes of some simple molecules and ions in which the central atom has one or more lone pairs. Table 4.8 (page 116) explains the reasons for the distortions in the geometry of the molecule.
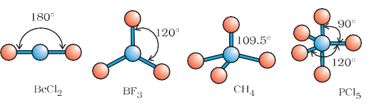
As depicted in Table 4.6, in the compounds of AB2, AB3, AB4, AB5 and AB6, the arrangement of electron pairs and the B atoms around the central atom A are : linear, trigonal planar, tetrahedral, trigonal- bipyramidal and octahedral, respectively. Such arrangement can be seen in the molecules like BF3 (AB3), CH4 (AB4) and PCl5 (AB5) as depicted below by their ball and stick models.
Fig. 4.6 The shapes of molecules in which central atom has no lone pair
The VSEPR Theory is able to predict geometry of a large number of molecules, especially the compounds of p-block elements accurately. It is also quite successful in determining the geometry quite-accurately even when the energy difference between possible structures is very small. The theoretical basis of the VSEPR theory regarding the effects of electron pair repulsions on molecular shapes is not clear and continues to be a subject of doubt and discussion.
Table 4.6 Geometry of Molecules in which the Central Atom has No Lone Pair of Electrons
TableTable 4.7 Shape (geometry) of Some Simple Molecules/Ions with Central Ions having One or More Lone Pairs of Electrons(E).
Table 4.8 Shapes of Molecules containing Bond Pair and Lone Pair
VALENCE BOND THEORY
As we know that Lewis approach helps in writing the structure of molecules but it fails to explain the formation of chemical bond. It also does not give any reason for the difference in bond dissociation enthalpies and bond lengths in molecules like H2 (435.8 kJ mol ,74 pm) and F2 (155 kJ mol , 144 pm), although in both the cases a single covalent bond is formed by the sharing of an electron pair between the respective atoms. It also gives no idea about the shapes of polyatomic molecules.
Similarly the VSEPR theory gives the geometry of simple molecules but theoretically, it does not explain them and also it has limited applications. To overcome these limitations the two important theories based on quantum mechanical principles are introduced. These are valence bond (VB) theory and molecular orbital (MO) theory.
Valence bond theory was introduced by Heitler and London (1927) and developed further by Pauling and others. A discussion of the valence bond theory is based on the knowledge of atomic orbitals, electronic configurations of elements (Units 2), the overlap criteria of atomic orbitals, the hybridization of atomic orbitals and the principles of variation and superposition. A rigorous treatment of the VB theory in terms of these aspects is beyond the scope of this book. Therefore, for the sake of convenience, valence bond theory has been discussed in terms of qualitative and non-mathematical treatment only. To start with, let us consider the formation of hydrogen molecule which is the simplest of all molecules.
Consider two hydrogen atoms A and B approaching each other having nuclei NA and NB and electrons present in them are represented by eA and eB. When the two atoms are at large distance from each other, there is no interaction between them. As these two atoms approach each other, new attractive and repulsive forces begin to operate.
Attractive forces arise between:
1. nucleus of one atom and its own electron that is NA – eA and NB– eB.
2. nucleus of one atom and electron of other atom i.e., NA– eB, NB– eA.
Similarly repulsive forces arise between
3. electrons of two atoms like eA – eB,
4.nuclei of two atoms NA – NB.Attractive forces tend to bring the two atoms close to each other whereas repulsive forces tend to push them apart (Fig. 4.7).
Fig. 4.7 Forces of attraction and repulsion during the formation of H2 molecule.
Experimentally it has been found that the magnitude of new attractive force is more than the new repulsive forces.As a result, two atoms approach each other and potential energy decreases.Ultimately a stage is reached where the net force of attraction balances the force of repulsion and system acquires minimum energy. At this stage two hydrogen atoms are said to be bonded together to form a stable molecule having the bond length of 74 pm.
Since the energy gets released when the bond is formed between two hydrogen atoms, the hydrogen molecule is more stable than that of isolated hydrogen atoms. The energy so released is called as bond enthalpy, which is corresponding to minimum in the curve depicted in Fig. 4.8. Conversely, 435.8 kJ of energy is required to dissociate one mole of H2 molecule.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamisamaaBa % aaleaacaaIYaaabeaakmaabmaabaGaam4zaaGaayjkaiaawMcaaiab % gUcaRiaaisdacaaIZaGaaGynaiaac6cacaaI4aGaam4AaiaadQeaca % WGTbGaam4BaiaadYgadaahaaWcbeqaaiabgkHiTiaaigdaaaGccqGH % sgIRcaWGibWaaeWaaeaacaWGNbaacaGLOaGaayzkaaGaey4kaSIaam % isamaabmaabaGaam4zaaGaayjkaiaawMcaaaaa!4E82! {H_2}\left( g \right) + 435.8kJmo{l^{ - 1}} \to H\left( g \right) + H\left( g \right)\)
Fig. 4.8 The potential energy curve for the formation of H2 molecule as a function of internuclear distance of the H atoms. The minimum in the curve corresponds to the most stable state of H2.
-
Orbital Overlap Concept
Orbital Overlap Concept
In the formation of hydrogen molecule, there is a minimum energy state when two hydrogen atoms are so near that their atomic orbitals undergo partial interpenetration. This partial merging of atomic orbitals is called overlapping of atomic orbitals which results in the pairing of electrons. The extent of overlap decides the strength of a covalent bond. In general, greater the overlap the stronger is the bond formed between two atoms. Therefore, according to orbital overlap concept, the formation of a covalent bond between two atoms results by pairing of electrons present in the valence shell having opposite spins.
-
Directional Properties of Bonds
Directional Properties of Bonds
As we have already seen, the covalent bond is formed by overlapping of atomic orbitals.The molecule of hydrogen is formed due to the overlap of 1s-orbitals of two H atoms.
In case of polyatomic molecules like CH4, NH3 and H2O, the geometry of the molecules is also important in addition to the bond formation. For example why is it so that CH4 molecule has tetrahedral shape and HCH bond angles are 109.5°? Why is the shape of NH3 molecule pyramidal ?
The valence bond theory explains the shape, the formation and directional properties of bonds in polyatomic molecules like CH4, NH3 and H2O, etc. in terms of overlap and hybridisation of atomic orbitals.
-
Overlapping of Atomic Orbitals
Overlapping of Atomic Orbitals
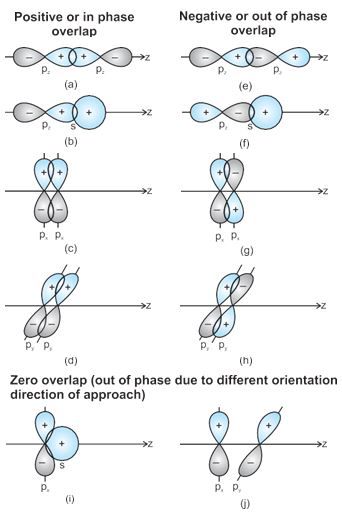
When orbitals of two atoms come close to form bond, their overlap may be positive, negative or zero depending upon the sign (phase) and direction of orientation of amplitude of orbital wave function in space (Fig. 4.9). Positive and negative sign on boundary surface diagrams in the Fig. 4.9 show the sign (phase) of orbital wave function and are not related to charge. Orbitals forming bond should have same sign (phase) and orientation in space. This is called positive overlap. Various overlaps of s and p orbitals are depicted in Fig. 4.9.
The criterion of overlap, as the main factor for the formation of covalent bonds applies uniformly to the homonuclear/heteronuclear diatomic molecules and polyatomic molecules.We know that the shapes of CH4, NH3, and H2O molecules are tetrahedral, pyramidal and bent respectively. It would be therefore interesting to use VB theory to find out if these geometrical shapes can be explained in terms of the orbital overlaps.
Let us first consider the CH4 (methane) molecule. The electronic configuration of carbon in its ground state is [He]2s2 2p2 which in the excited state becomes [He] 2s1 2p 1 2p 1 \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaaGOmaiaadc % hadaqhaaWcbaGaamOEaaqaaiaaigdaaaaaaa!398E! 2p_z^1\).The energy required for this excitation is compensated by the release of energy due to overlap between the orbitals of carbon and the
Fig.4.9 Positive, negative and zero overlaps of s and p atomic orbitals
hydrogen.The four atomic orbitals of carbon, each with an unpaired electron can overlap with the 1s orbitals of the four H atoms which are also singly occupied. This will result in the formation of four C-H bonds. It will, however, be observed that while the three p orbitals of carbon are at 90° to one another, the HCH angle for these will also be 90°. That is three C-H bonds will be oriented at 90° to one another.The 2s orbital of carbon and the 1s orbital of H are spherically symmetrical and they can overlap in any direction. Therefore the direction of the fourth C-H bond cannot be ascertained. This description does not fit in with the tetrahedral HCH angles of 109.5°. Clearly, it follows that simple atomic orbital overlap does not account for the direcional characteristics of bonds in CH4. Using similar procedure and arguments, it can be seen that in the case of NH3 and H2O molecules, the HNH and HOH angles should be 90°.This is in disagreement with the actual bond angles of
107° and 104.5° in the NH3 and H2O molecules respectively.
-
Types of Overlapping and Nature of Covalent Bonds and Strength of Bonds
Types of Overlapping and Nature of Covalent Bonds and Strength of Bonds
The covalent bond may be classified into two types depending upon the types of overlapping:
(i) Sigma(\(\sigma\)) bond, and (ii) pi(\(\pi\)) bond
1. Sigma(\(\sigma\)) bond : This type of covalent bond is formed by the end to end (head-on) overlap of bonding orbitals along the internuclear axis. This is called as head on overlap or axial overlap. This can be formed by any one of the following types of combinations of atomic orbitals.
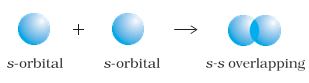
2. s-s overlapping : In this case, there is overlap of two half filled s-orbitals along the internuclear axis as shown below :
3. s-p overlapping: This type of overlap occurs between half filled s-orbitals of one atom and half filled p-orbitals of another atom.
4. p–p overlapping : This type of overlap takes place between half filled p-orbitals of the two approaching atoms.
3. pi(\(\pi\)) bond : In the formation of \(\pi\) bond the atomic orbitals overlap in such a way that their axes remain parallel to each other and perpendicular to the internuclear axis.The orbitals formed due to sidewise overlapping consists of two saucer type charged clouds above and below the plane of the participating atoms.
Strength of Sigma and pi Bonds
Basically the strength of a bond depends upon the extent of overlapping. In case of sigma bond, the overlapping of orbitals takes place to a larger extent.Hence, it is stronger as compared to the pi bond where the extent of overlapping occurs to a smaller extent.Further, it is important to note that in the formation of multiple bonds between two atoms of a molecule, pi bond(s) is formed in addition to a sigma bond.