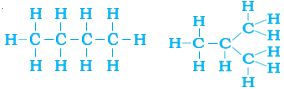-
Allotropes of Carbon
ALLOTROPY:
Allotropes of Carbon
The element carbon occurs in different forms in nature with widely varying physical properties. Both diamond and graphite are formed by carbon atoms, the difference lies in the manner in which the carbon atoms are bonded to one another.
Diamond

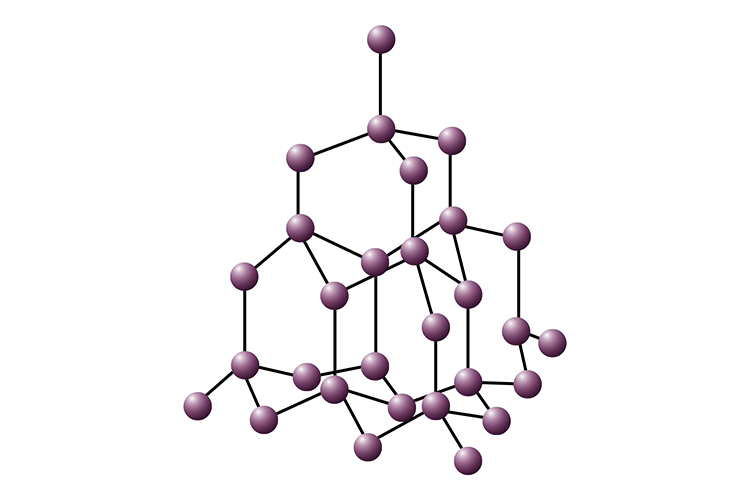
In diamond, each carbon atom is bonded to four other carbon atoms forming a rigid three-dimensional structure.
Structure of Diamond
Graphite
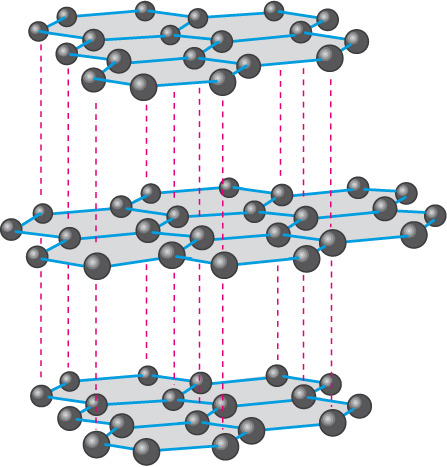
In graphite, each carbon atom is bonded to three other carbon atoms in the same plane giving a hexagonal array. One of these bonds is a double-bond, and thus the valency of carbon is satisfied. Graphite structure is formed by the hexagonal arrays being placed in layers one above the other.
Structure of Graphite
These two different structures result in diamond and graphite having very different physical properties even though their chemical properties are the same. Diamond is the hardest substance known while graphite is smooth and slippery. Graphite is also a very good conductor of electricity, unlike other non-metals that you studied in the previous Chapter.
Diamonds can be synthesised by subjecting pure carbon to very high pressure and temperature. These synthetic diamonds are small but are otherwise indistinguishable from natural diamonds
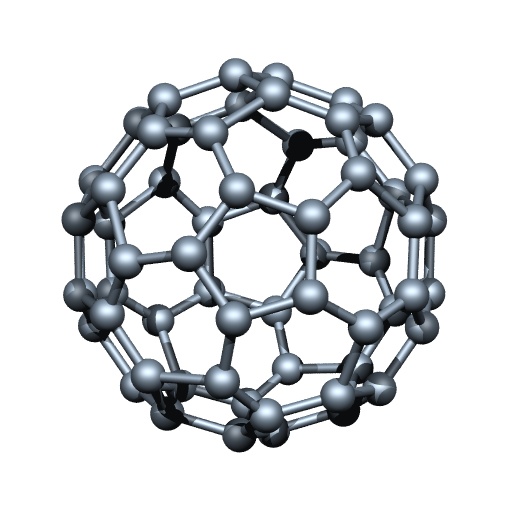
Fullerenes
Fullerenes form another class of carbon allotropes. The first one to be identified was C-60 which has carbon atoms arranged in the shape of a football. Since this looked like the geodesic dome designed by the US architect Buckminster Fuller, the molecule was named fullerene.
Structure of Buckminster Fullerence (C60)
QUESTIONS
1. What would be the electron dot structure of carbon dioxide which has the formula CO2?
2. What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur?
(Hint – The eight atoms of sulphur are joined together in the form of a ring.)
-
VERSATILE NATURE OF CARBON
VERSATILE NATURE OF CARBON
We have seen the formation of covalent bonds by the sharing of electrons in various elements and compounds. We have also seen the structure of a simple carbon compound, methane. At the beginning of the Chapter, we saw how many things we use contain carbon. In fact, we ourselves are made up of carbon compounds. The numbers of carbon compounds whose formulae are known to chemists were recently estimated to be in millions! This outnumbers by a large margin the compounds formed by all the other elements put together. Why is it that this property is seen in carbon and no other element? The nature of the covalent bond enables carbon to form a large number of compounds. Two factors noticed in the case of carbon are –
1. Carbon has the unique ability to form bonds with other atoms of carbon, giving rise to large molecules. This property is called catenation. These compounds may have long chains of carbon, branched chains of carbon, or even carbon atoms arranged in rings. In addition, carbon atoms may be linked by single, double, or triple bonds. Compounds of carbon, which are linked by only single bonds between the carbon atoms are called saturated compounds. Compounds of carbon having double or triple bonds between their carbon atoms are called unsaturated compounds.
No other element exhibits the property of catenation to the extent seen in carbon compounds. Silicon forms compounds with hydrogen which have chains of upto seven or eight atoms, but these compounds are very reactive. The carbon-carbon bond is very strong and hence stable. This gives us a large number of compounds with many carbon atoms linked to each other.
2. Since carbon has a valency of four, it is capable of bonding with four other atoms of carbon or atoms of some other mono-valent element. Compounds of carbon are formed with oxygen, hydrogen, nitrogen, sulphur, chlorine, and many other elements giving rise to compounds with specific properties that depend on the elements other than carbon present in the molecule.
Again the bonds that carbon forms with most other elements are very strong making these compounds exceptionally stable. One reason for the formation of strong bonds by carbon is its small size. This enables the nucleus to hold on to the shared pairs of electrons strongly. The bonds formed by elements having bigger atoms are much weaker.
More to know
Organic compounds
The two characteristic features are seen in carbon, that is, tetravalency and catenation, put together give rise to a large number of compounds. Many have the same non-carbon atom or group of atoms attached to different carbon chains. These compounds were initially extracted from natural substances and it was thought that these carbon compounds or organic compounds could only be formed within a living system. That is, it was postulated that a ‘ vital force ’ was necessary for their synthesis. Friedrich Wöhler disproved this in 1828 by preparing urea from ammonium cyanate. But carbon compounds, except for carbides, oxides of carbon, carbonate, and hydrogencarbonate salts continue to be studied under organic chemistry.
-
Saturated and Unsaturated Carbon Compounds
Saturated and Unsaturated Carbon Compounds
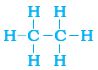
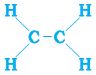
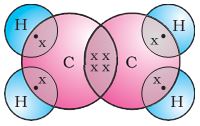
We have already seen the structure of methane. Another compound formed between carbon and hydrogen is ethane with a formula of C2H6. In order to arrive at the structure of simple carbon compounds, the first step is to link the carbon atoms together with a single bond (Fig. 4.6a) and then use the hydrogen atoms to satisfy the remaining valencies of carbon (Fig. 4.6b). For example, the structure of ethane is arrived in the following steps –
Step 1:
C — C
Figure 4.6 (a) Carbon atoms linked together with a single bond
Three valencies of each carbon atom remain unsatisfied, so each is bonded to three hydrogen atoms giving:
Step 2:
Figure 4.6 (b) Each carbon atom bonded to three hydrogen atoms
Step 3:
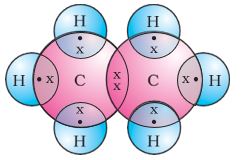
The electron dot structure of ethane is shown in Fig. 4.6(c).
Figure 4.6(c) Electron dot structure of ethane
Propane
Can you draw the structure of propane, which has the molecular formula C3H8 in a similar manner? You will see that the valencies of all the atoms are satisfied by single bonds between them. Such carbon compounds are called saturated compounds. These compounds are
normally not very reactive.
Ethene
However, another compound of carbon and hydrogen has the formula C2H4 and is called ethene. How can this molecule be depicted? We follow the same step-wise approach as above.
Step 1:
Carbon-carbon atoms linked together with a single bond (Step 1).
C — C
Step 2:
We see that one valency per carbon atom remains unsatisfied (Step 2).
Step 3:
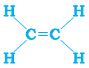
This can be satisfied only if there is a double bond between the two (Step 3)
The electron dot structure for ethene is given in Fig. 4.7.
Figure 4.7 Structure of ethene
Ethyne
Yet another compound of hydrogen and carbon has the formula C2H2 and is called ethyne. Can you draw the electron dot structure for ethyne? How many bonds are necessary between the two carbon atoms in order to satisfy their valencies? Such compounds of carbon having double or triple bonds between the carbon atoms are known as unsaturated carbon compounds and they are more reactive than the saturated carbon compounds.
-
Chains, Branches and Rings
Chains, Branches and Rings
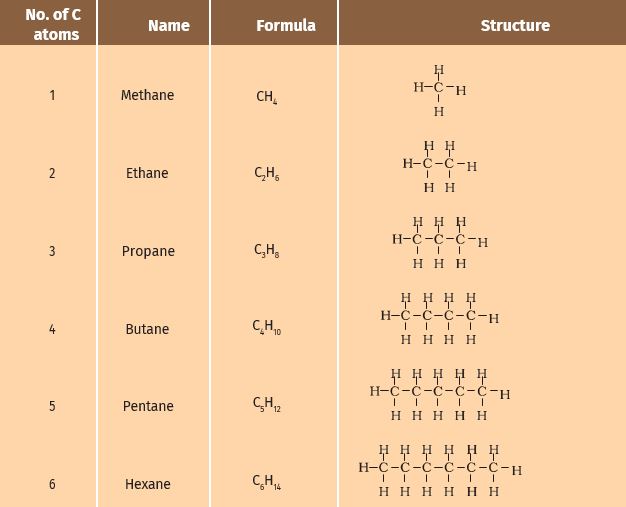
In the earlier section, we mentioned the carbon compounds methane, ethane, and propane, containing respectively 1, 2, and 3 carbon atoms. Such ‘chains’ of carbon atoms can contain many more carbon atoms. The names and structures of six of these are given in Table 4.2.
Table 4.2 Formulae and structures of saturated compounds of carbon and hydrogen
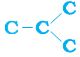
But, let us take another look at butane. If we make the carbon ‘skeleton’ with four carbon atoms, we see that two different possible ‘skeletons’ are –
C—C—C—C
Figure 4.8 (a) Two possible carbon-skeletons
Filling the remaining valencies with hydrogen gives us –
Figure 4.8 (a) Complete molecules for two structures with the formula C4H10
We see that both these structures have the same formula C4H10. Such compounds with identical molecular formula but different structures are called structural isomers.
In addition to straight and branched carbon chains, some compounds have carbon atoms arranged in the form of a ring. For example, cyclohexane has the formula C6H12 and the following structure –
(a) (b)
Figure 4.9 Structure of cyclohexane (a) carbon skeleton (b) complete molecule
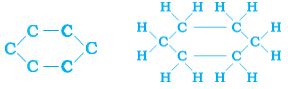
Can you draw the electron dot structure for cyclohexane? Straight chain, branched-chain, and cyclic carbon compounds, all may be saturated or unsaturated. For example, benzene, C6H6, has the following structure –
Figure 4.9 Structure of benzene
All these carbon compounds which contain only carbon and hydrogen are called hydrocarbons. Among these, the saturated hydrocarbons are called alkanes. The unsaturated hydrocarbons which contain one or more double bonds are called alkenes. Those containing one or more triple bonds are called alkynes.