-
Will you be my Friend?
Will you be my Friend?
Carbon seems to be a very friendly element. So far we have been looking at compounds containing carbon and hydrogen only. But carbon also forms bonds with other elements such as halogens, oxygen, nitrogen and sulphur. In a hydrocarbon chain, one or more hydrogens can be replaced by these elements, such that the valency of carbon remains satisfied. In such compounds, the element replacing hydrogen is referred to as a heteroatom. These heteroatoms are also present in some groups as given in Table 4.3.
Table 4.3 Some functional groups in carbon compounds
These heteroatoms and the group containing these confer specific properties to the compound, regardless of the length and nature of the carbon chain, and hence are called functional groups. Some important functional groups are given in Table 4.3. Free valency or valencies of the group are shown by the single line. The functional group is attached to the carbon chain through this valency by replacing one hydrogen atom or atoms.
-
Homologous Series
Homologous Series
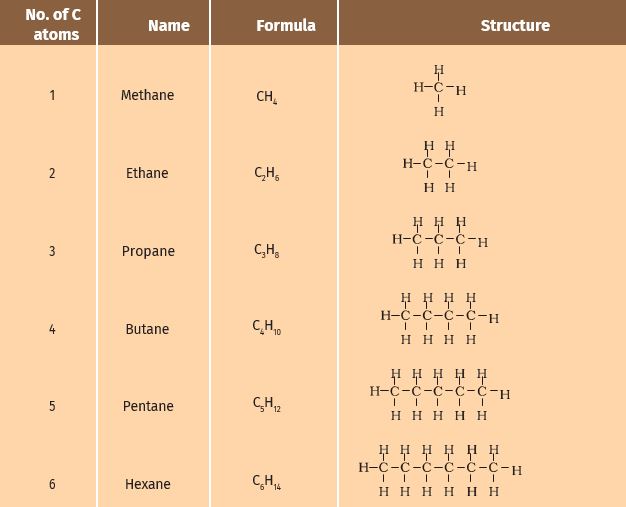
You have seen that carbon atoms can be linked together to form chains of varying lengths. These chains can be branched also. In addition, a hydrogen atom or other atoms on these carbon chains can be replaced by any of the functional groups that we saw above. The presence of a functional group such as alcohol decides the properties of the carbon compound, regardless of the length of the carbon chain. For example, the chemical properties of CH3OH, C2H5OH, C3H7OH, and C4H9OH are all very similar. Hence, such a series of compounds in which the same
functional group substitutes for hydrogen in a carbon chain is called a homologous series.
Let us look at the homologous series that we saw earlier in Table
If we look at the formulae of successive compounds, say –
CH4 and C2H6 — these differ by a –CH2- unit
C2H6 and C3H8 — these differ by a –CH2- unit
What is the difference between the next pair – propane, and butane (C4H10)? Can you find out the difference in molecular masses between these pairs (the atomic mass of carbon is 12 u and the atomic mass of hydrogen is 1 u)?
Similarly, take the homologous series for alkenes. The first member of the series is ethene which we have already come across in Section 4.2.1. What is the formula for ethene? The succeeding members have the formula C3H6, C4H8, and C5H10. Do these also differ by a –CH2– unit? Do you see any relation between the number of carbon and hydrogen atoms in these compounds?
The general formula for alkenes can be written as CnH2n, where n = 2, 3, 4. Can you similarly generate the general formula for alkanes and alkynes?
As the molecular mass increases in any homologous series, a gradation in physical properties is seen. This is because the melting and boiling points increase with increasing molecular mass. Other physical properties such as solubility in a particular solvent also show a similar gradation. But the chemical properties, which are determined solely by the functional group, remain similar in a homologous series.
Activity 4.2
Calculate the difference in the formulae and molecular masses for (a) CH3OH and C2H5OH (b) C2H5OH and C3H7OH, and (c) C3H7OH and C4H9OH.
Is there any similarity between these three?
Arrange these alcohols in the order of increasing carbon atoms to get a family. Can we call this family a homologous series?
Generate the homologous series for compounds containing up to four carbons for the other functional groups given in Table 4.3.