-
SPACE BETWEEN PARTICLES OF MATTER
SPACE BETWEEN PARTICLES OF MATTER
Activity 1.1: ( Space between particles of Matter )
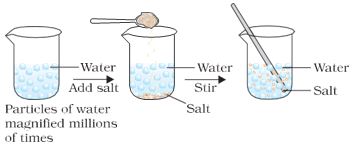
* Take a 100 mL beaker.
* Fill half the beaker with water and mark the level of water.
* Dissolve some salt/ sugar with the help of a glass rod.
* Observe any change in the water level. What do you think has happened to the salt?
* Where does it disappear?
* Does the level of water change?
Figure 1.1: When we dissolve salt in water, the particles of salt get into the spaces between particles of water.
Activity 1.2: ( Particles of matter are small )
* Take 2-3 crystals of potassium permanganate and dissolve them in 100 mL of water.
* Take out approximately 10 mL of this solution and put it into 90 mL of clear water
* Take out 10 mL of this solution and put it into another 90 mL of clear water.
* Keep diluting the solution like this 5 to 8 times.
* Is the water still coloured?
Figure 1.2: Estimating how small are the particles of matter. With every dilution, though the colour becomes light, it is still visible.
In activities 1.1 and 1.2 we saw that particles of sugar, salt, Dettol, or potassium permanganate got evenly distributed in the water. Similarly, when we make tea, coffee or lemonade (nimbu paani ), particles of one type of matter get into the spaces between particles of the other. This shows that there is enough space between particles of matter.
Source: This topic is taken from NCERT TEXTBOOK
-
MOTION OF PARTICLES OF MATTER
MOTION OF PARTICLES OF MATTER
Activity 1.3: ( Experiment with incense stick )
* Put an unlit incense stick in a corner of your class. How close do you have to go near it so as to get its smell?
* Now light the incense stick. What happens? Do you get the smell sitting at a distance?
* Record your observations
Activity 1.4: ( Diffusion of Particles )
* Take two glasses/beakers filled with water.
* Put a drop of blue or red ink slowly and carefully along the sides of the first beaker and honey in the same way in the second beaker.
* Leave them undisturbed in your house or in a corner of the class.
* Record your observations.
* What do you observe immediately after adding the ink drop?
* What do you observe immediately after adding a drop of honey?
* How many hours or days does it take for the colour of ink to spread evenly throughout the water?
Activity 1.5: ( temperature effect on diffusion )
* Drop a crystal of copper sulphate or potassium permanganate into a glass of hot water and another containing cold water. Do not stir the solution. Allow the crystals to settle at the bottom.
* What do you observe just above the solid crystal in the glass?
* What happens as time passes?
* What does this suggest about the particles of solid and liquid?
* Does the rate of mixing change with temperature? Why and how?
From the above three activities (1.3, 1.4, and 1.5), we can conclude the following:
Particles of matter are continuously moving, that is, they possess what we call kinetic energy. As the temperature rises, particles move faster. So, we can say that with an increase in temperature the kinetic energy of the particles also increases.
In the above three activities, we observe that particles of matter intermix on their own with each other. They do so by getting into the spaces between the particles. This intermixing of particles of two different types of matter on their own is called diffusion. We also observe that on heating, diffusion becomes faster. Why does this happen?
Source: This topic is taken from NCERT TEXTBOOK
-
ATTRACTION BETWEEN PARTICLES OF MATTER
ATTRACTION BETWEEN PARTICLES OF MATTER
Activity 1.6:
* Play this game in the field— make four groups and form human chains as suggested:
* The first group should hold each other from the back and lock arms like Idu-Mishmi dancers (Fig. 1.3).
Figure 1.3:
* The second group should hold hands to form a human chain.
* The third group should form a chain by touching each other with only their finger tips.
* Now, the fourth group of students should run around and try to break the three human chains one by one into as many small groups as possible.
* Which group was the easiest to break? Why?
* If we consider each student as a particle of matter, then in which group the particles held each other with the maximum force?
Activity 1.7: ( Attractive forces between particles )
* Take an iron nail, a piece of chalk and a rubber band.
* Try breaking them by hammering, cutting or stretching.
* In which of the above three substances do you think the particles are held together with greater force?
Activity 1.8: ( diver cutting surface of water )
* Take some water in a container, try cutting the surface of water with your fingers.
* Were you able to cut the surface of water?
* What could be the reason behind the surface of water remaining together?
The above three activities (1.6, 1.7 and 1.8) suggest that particles of matter have force acting between them. This force keeps the particles together. The strength of this force of attraction varies from one kind of matter to another.
Questions
1. Which of the following are matter?
(i) Chair, air, love, smell, hate, almonds, thought, cold, lemon water, smell of perfume.
2. Give reasons for the following observation:
(i) The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
3. A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
4. What are the characteristics of the particles of matter?
Source: This topic is taken from NCERT TEXTBOOK