-
INTRODUCTION
INTRODUCTION TO PURE SUBSTANCE
How do we judge whether milk, ghee, butter, salt, spices, mineral water or juice that we buy from the market are pure?
Figure 2.1: Some consumable items
Have you ever noticed the word ‘pure’ written on the packs of these consumables? For a common person pure means having no adulteration. But, for a scientist all these things are actually mixtures of different substances and hence not pure. For example, milk is actually a mixture of water, fat, proteins etc. When a scientist says that something is pure, it means that all the constituent particles of that substance are the same in their chemical nature. A pure substance consists of a single type of particles. In other words, a substance is a pure single form of matter.
As we look around, we can see that most of the matter around us exist as mixtures of two or more pure components, for example, sea water, minerals, soil etc. are all mixtures.
Source: This topic is taken from NCERT TEXTBOOK
-
WHAT IS MIXTURE
WHAT IS MIXTURE
Mixtures are constituted by more than one kind of pure form of matter. We know that dissolved sodium chloride can be separated from water by the physical process of evaporation. However, sodium chloride is itself a pure substance and cannot be separated by physical process into its chemical constituents. Similarly, sugar is a substance that contains only one kind of pure matter and its composition is the same throughout.
Soft drinks and soil are not single pure substances. Whatever the source of a pure substance may be, it will always have the same characteristic properties. Therefore, we can say that a mixture contains more than one pure substance.
Source: This topic is taken from NCERT TEXTBOOK
-
TYPES OF MIXTURE
TYPES OF MIXTURE
Depending upon the nature of the components that form a mixture, we can have different types of mixtures.
Activity 2.1: ( Homogeneous mixtures and Heterogeneous Mixtures )
* Let us divide the class into groups A, B, C and D.
* Group A takes a beaker containing 50 mL of water and one spatula full of copper sulphate powder. Group B takes 50 mL of water and two spatula full of copper sulphate powder in a beaker. Groups C and D can take different amounts of copper sulphate and potassium permanganate or common salt ( Sodium Chloride ) and mix the given components to form a mixture.
* Report the observations on the uniformity in colour and texture.
* Groups A and B have obtained a mixture that has a uniform composition throughout. Such mixtures are called homogeneous mixtures or solutions. Some other examples of such mixtures are (i) salt dissolved in water and (ii) dissolved in water. Compare the colour of the solutions of the two groups. Though both the groups have obtained copper sulphate solution the intensity of the colour of the solutions is different. This shows that a homogeneous mixture can have a variable composition.
* Groups C and D have obtained mixtures, which contain physically distinct parts and have non-uniform compositions. Such mixtures are called heterogeneous mixtures. Mixtures of Sodium Chloride and iron filings, salt and sulphur, and oil and water are examples of heterogeneous mixtures.
Activity 2.2:
* Let us again divide the class into four groups – A, B, C and D.
* Distribute the following samples to each group:
- Few crystals of copper sulphate to group A.
- One spatula full of copper sulphate to group B.
- Chalk powder or wheat flour to group C.
- Few drops of milk or ink to group D.
* Each group should add the given sample in water and stir properly using a glass rod. Are the particles in the mixture visible?
* Direct a beam of light from a torch through the beaker containing the mixture and observe from the front. Was the path of the beam of light visible?
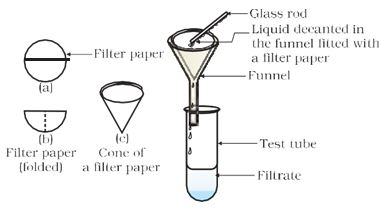
* Leave the mixtures undisturbed for a few minutes (and set up the filtration apparatus in the meantime). Is the mixture stable or do the particles begin to settle after some time?
* Filter the mixture. Is there any residue on the filter paper?
* Discuss the results and form an opinion.
* Groups A and B have got a solution.
* Group C has got a suspension.
* Group D has got a colloidal solution.
Figure 2.1: Filtration
Now, we shall learn about solutions, suspensions and colloidal solutions in the following sections.
Questions
1. What is meant by a substance?
2. List the points of difference between homogeneous and heterogeneous mixtures.
Source: This topic is taken from NCERT TEXTBOOK