-
WHAT IS SOLUTION?
WHAT IS SOLUTION?
A solution is a homogeneous mixture of two or more substances. You come across various types of solutions in your daily life. Lemonade, soda water etc. are all examples of solutions. Usually we think of a solution as a liquid that contains either a solid, liquid or a gas dissolved in it. But, we can also have solid solutions (alloys) and gaseous solutions (air). In a solution there is homogeneity at the particle level. For example, lemonade tastes the same throughout. This shows that particles of sugar or salt are evenly distributed in the solution.
A solution has a solvent and a solute as its components. The component of the solution that dissolves the other component in it (usually the component present in larger amount) is called the solvent. The component of the solution that is dissolved in the solvent (usually present in lesser quantity) is called the solute.
Examples
i) A solution of sugar in water is a solid in liquid solution. In this solution, sugar is the solute and water is the solvent.
ii) A solution of iodine in alcohol known as ‘tincture of iodine’, has iodine (solid) as the solute and alcohol (liquid) as the solvent.
iii) Aerated drinks like soda water etc., are gas in liquid solutions. These contain carbon dioxide (gas) as solute and water (liquid) as solvent.
iv) Air is a mixture of gas in gas. Air is a homogeneous mixture of a number of gases. Its two main constituents are: oxygen (21%) and nitrogen (78%). The other gases are present in very small quantities.
Alloys
Alloys are mixtures of two or more metals or a metal and a non-metal and cannot be separated into their components by physical methods. But still, an alloy is considered as a mixture because it shows the properties of its constituents and can have variable composition. For example, brass is a mixture of approximately 30% zinc and 70% copper.
Source: This topic is taken from NCERT TEXTBOOK
-
PROPERTIES OF SOLUTION
PROPERTIES OF SOLUTION
* A solution is a homogeneous mixture.
* The particles of a solution are smaller than 1 nm (10-9 metre) in diameter. So, they cannot be seen by naked eyes.
* Because of very small particle size, they do not scatter a beam of light passing through the solution. So, the path of light is not visible in a solution.
* The solute particles cannot be separated from the mixture by the process of filtration. The solute particles do not settle down when left undisturbed, that is, a solution is stable.
Source: This topic is taken from NCERT TEXTBOOK
-
WHAT IS A SUSPENSION?
WHAT IS A SUSPENSION?
Activity 2.2:
* Let us again divide the class into four groups – A, B, C and D.
* Distribute the following samples to each group:
- Few crystals of copper sulphate to group A.
- One spatula full of copper sulphate to group B.
- Chalk powder or wheat flour to group C.
- Few drops of milk or ink to group D.
* Each group should add the given sample in water and stir properly using a glass rod. Are the particles in the mixture visible?
* Direct a beam of light from a torch through the beaker containing the mixture and observe from the front. Was the path of the beam of light visible?
* Leave the mixtures undisturbed for a few minutes (and set up the filtration apparatus in the meantime). Is the mixture stable or do the particles begin to settle after some time?
* Filter the mixture. Is there any residue on the filter paper?
* Discuss the results and form an opinion.
* Groups A and B have got a solution.
* Group C has got a suspension.
* Group D has got a colloidal solution.
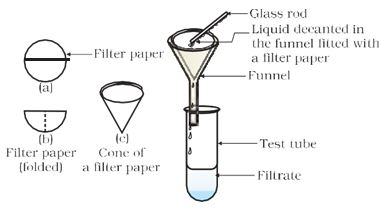
Figure 2.2: Filtration
Non-homogeneous systems, like those obtained by group C in activity 2.2, in which solids are dispersed in liquids, are called suspensions. A suspension is a heterogeneous mixture in which the solute particles do not dissolve but remain suspended throughout the bulk of the medium. Particles of a suspension are visible to the naked eye.
Properties of suspension
* Suspension is a heterogeneous mixture.
* The particles of a suspension can be seen by the naked eye
* The particles of a suspension scatter a beam of light passing through it and make its path visible.
* The solute particles settle down when a suspension is left undisturbed, that is, a suspension is unstable. They can be separated from the mixture by the process of filtration. When the particles settle down, the suspension breaks and it does not scatter light anymore.
Source: This topic is taken from NCERT TEXTBOOK
-
WHAT IS A COLLOIDAL SOLUTION?
WHAT IS A COLLOIDAL SOLUTION?
The mixture obtained by group D in the activity is called a colloid or a colloidal solution. The particles of a colloid are uniformly spread throughout the solution. Due to the relatively smaller size of particles, as compared to that of a suspension, the mixture appears to be homogeneous. But actually, a colloidal solution a heterogeneous mixture, for example, milk.
Activity 2.2:
* Let us again divide the class into four groups – A, B, C, and D.
* Distribute the following samples to each group:
- Few crystals of copper sulphate to group A.
- One spatula full of copper sulphate to group B.
- Chalk powder or wheat flour to group C.
- Few drops of milk or ink to group D.
* Each group should add the given sample in water and stir properly using a glass rod. Are the particles in the mixture visible?
* Direct a beam of light from a torch through the beaker containing the mixture and observe from the front. Was the path of the beam of light visible?
* Leave the mixtures undisturbed for a few minutes (and set up the filtration apparatus in the meantime). Is the mixture stable or do the particles begin to settle after some time?
* Filter the mixture. Is there any residue on the filter paper?
* Discuss the results and form an opinion.
* Groups A and B have got a solution.
* Group C has got a suspension.
* Group D has got a colloidal solution.
Figure 2.1: Filtration
Properties of Colloidal Solution
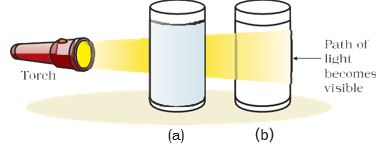
Because of the small size of colloidal particles, we cannot see them with naked eyes. But, these particles can easily scatter a beam of visible light as observed in activity 2.2. The scattering of a beam of light is called the Tyndall effect after the name of the scientist who discovered this effect.
Tyndall effect can also be observed when a fine beam of light enters a room through a small hole. This happens due to the scattering of light by the particles of dust and smoke in the air.
Figure 2.3: (a) Solution of copper sulphate does not show Tyndall effect, (b) mixture of water and milk shows Tyndall effect.
Tyndall effect can be observed when sunlight passes through the canopy of a dense forest. In the forest, mist contains tiny droplets of water, which act as particles of colloid dispersed in air.
Figure 2.4: The Tyndall effect
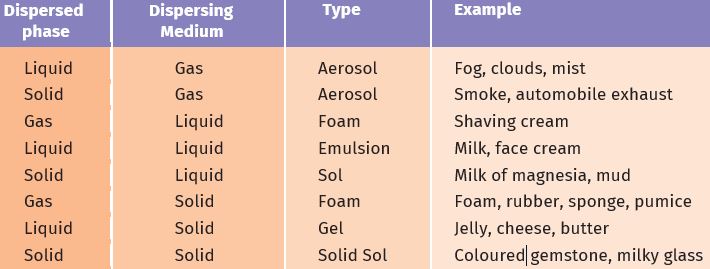
Table 2.1: Common examples of colloids
Source: This topic is taken from NCERT TEXTBOOK
-
PROPERTIES OF COLLOIDAL SOLUTION
PROPERTIES OF COLLOIDAL SOLUTION
* A colloid is a heterogeneous mixture.
* The size of particles of a colloid is too small to be individually seen by naked eyes.
* Colloids are big enough to scatter a beam of light passing through it and make its path visible
* They do not settle down when left undisturbed, that is, a colloid is quite stable.
* They cannot be separated from the mixture by the process of filtration. But, a special technique of separation known as centrifugation (perform activity 2.5), can be used to separate the colloidal particles.
The components of a colloidal solution are the dispersed phase and the dispersion medium. The solute-like component or the dispersed particles in a colloid form the dispersed phase, and the component in which the dispersed phase is suspended is known as the dispersing medium. Colloids are classified according to the state (solid, liquid or gas) of the dispersing medium and the dispersed phase. A few common examples are given in Table 2.1. From this table you can see that they are very common everyday life.
Table 2.1: Common examples of colloids
Source: This topic is taken from NCERT TEXTBOOK