-
UNSATURATED AND SATURATED SOLUTION
UNSATURATED AND SATURATED SOLUTION
Saturated solution
Activity 2.2:
* Let us again divide the class into four groups – A, B, C, and D.
* Distribute the following samples to each group:
- Few crystals of copper sulphate to group A.
- One spatula full of copper sulphate to group B.
- Chalk powder or wheat flour to group C.
- Few drops of milk or ink to group D.
* Each group should add the given sample in water and stir properly using a glass rod. Are the particles in the mixture visible?
* Direct a beam of light from a torch through the beaker containing the mixture and observe from the front. Was the path of the beam of light visible?
* Leave the mixtures undisturbed for a few minutes (and set up the filtration apparatus in the meantime). Is the mixture stable or do the particles begin to settle after some time?
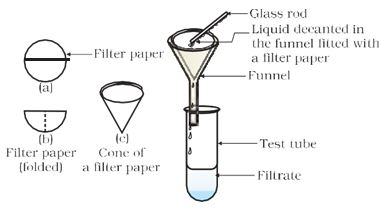
* Filter the mixture. Is there any residue on the filter paper?
* Discuss the results and form an opinion.
* Groups A and B have got a solution.
* Group C has got a suspension.
* Group D has got a colloidal solution.
Figure 2.1: Filtration
In activity 2.2, we observed that groups A and B obtained different shades of solutions. So, we understand that in a solution the relative proportion of the solute and solvent can be varied. Depending upon the amount of solute present in a solution, it can be called a dilute, concentrated or a saturated solution. Dilute and concentrated are comparative terms. In activity 2.2, the solution obtained by group A is dilute as compared to that obtained by group B.
What would happen if you were to take a saturated solution at a certain temperature and cool it slowly.
Activity 2.3:
* Take approximately 50 mL of water each in two separate beakers.
* Add salt in one beaker and sugar or barium chloride in the second beaker with continuous stirring.
* When no more solute can be dissolved, heat the contents of the beaker to raise the temperature by about 5°C.
* Start adding the solute again.
Is the amount of salt and sugar or barium chloride, that can be dissolved in water at a given temperature, the same?
At any particular temperature, a solution that has dissolved as much solute as it is capable of dissolving, is said to be a saturated solution. In other words, when no more solute can be dissolved in a solution at a given temperature, it is called a saturated solution.
Unsaturated solution
If the amount of solute contained in a solution is less than the saturation level, it is called an unsaturated solution.
Source: This topic is taken from NCERT TEXTBOOK
-
SOLUBILITY
SOLUBILITY
The amount of the solute present in the saturated solution at this temperature is called its solubility. We can infer from the above activity that different substances in a given solvent have different solubilities at the same temperature. The concentration of a solution is the amount (mass or volume) of solute present in a given amount (mass or volume) of solution.
There are various ways of expressing the concentration of a solution, but here we will learn only three methods:
i. Mass by mass percentage
ii. Volume by volume percentage
iii. Mass by volume percentage
Source: This topic is taken from NCERT TEXTBOOK