-
WHAT IS AN ATOM?
WHAT IS AN ATOM?
Have you ever observed a mason building walls, from these walls a room and then a collection of rooms to form a building? What is the building block of the huge building? What about the building block of an ant-hill? It is a small grain of sand. Similarly, the building blocks of all matter are atoms.
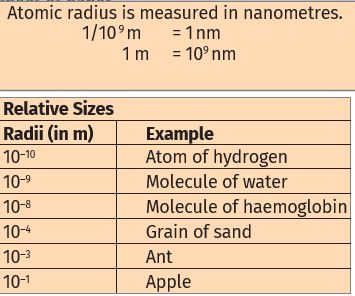
How big are atoms?
Atoms are very small, they are smaller than anything that we can imagine or compare with. More than millions of atoms when stacked would make a layer barely as thick as this sheet of paper.
We might think that if atoms are so insignificant in size, why should we care about them? This is because our entire world is made up of atoms. We may not be able to see them, but they are there, and constantly affecting whatever we do. Through modern techniques, we can now produce magnified images of surfaces of elements showing atoms.
Figure 3.2: An image of the surface of the silicon
Source: This topic is taken from NCERT TEXTBOOK
-
SYMBOLS OF ATOMS
SYMBOLS OF ATOMS
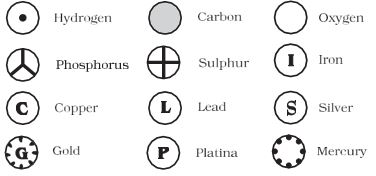
Dalton was the first scientist to use symbols for elements in a very specific sense. When he used a symbol for an element he also meant a definite quantity of that element, that is, one atom of that element. Berzilius suggested that the symbols of elements be made from one or two letters of the name of the element.
Figure 3.3: Symbols for some elements as proposed by Dalton
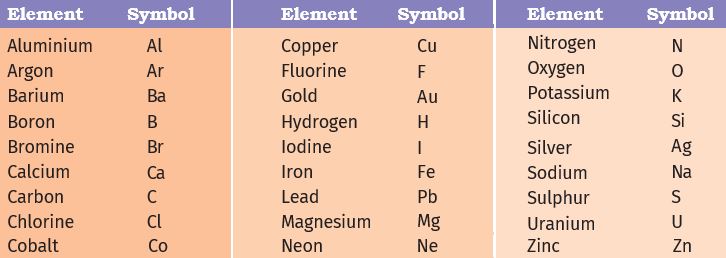
In the beginning, the names of elements were derived from the name of the place where they were found for the first time. For example, the name copper was taken from Cyprus. Some names were taken from specific colours. For example, gold was taken from the English word meaning yellow. Nowadays, IUPAC (International Union of Pure and Applied Chemistry) is an international scientific organisation that approves names of elements, symbols, and units. Many of the symbols are the first one or two letters of the element’s name in English. The first letter of a symbol is always written as a capital letter (uppercase) and the second letter as a small letter (lowercase).
Symbols of some elements are formed from the first letter of the name and a letter, appearing later in the name. Examples are: (i) chlorine, Cl, (ii) zinc, Zn etc.
Other symbols have been taken from the names of elements in Latin, German, or Greek. For example, the symbol of iron is Fe from its Latin name Ferrum, sodium is Na from natrium, potassium is K from kalium. Therefore, each element has a name and a unique chemical symbol.
(The above table is given for you to refer to whenever you study elements. Do not bother to memorize all in one go. With the passage of time and repeated usage you will automatically be able to reproduce the symbols).
Source: This topic is taken from NCERT TEXTBOOK