-
WHAT IS A MOLECULE
HOW DO ATOMS EXIST?
Atoms of most elements are not able to exist independently. Atoms form molecules and ions. These molecules or ions aggregate in large numbers to form the matter that we can see, feel or touch.
WHAT IS A MOLECULE
A molecule is in general a group of two or more atoms that are chemically bonded together, that is, tightly held together by attractive forces. A molecule can be defined as the smallest particle of an element or a compound that is capable of independent existence and shows all the properties of that substance. Atoms of the same element or of different elements can join together to form molecules.
Source: This topic is taken from NCERT TEXTBOOK
-
MOLECULES OF ELEMENTS AND ATOMICITY
MOLECULES OF ELEMENTS AND ATOMICITY
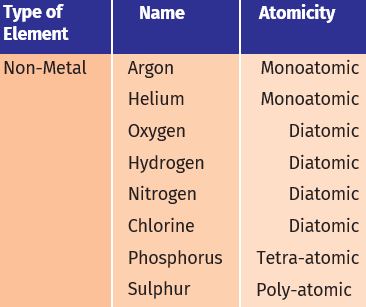
The molecules of an element are constituted by the same type of atoms. Molecules of many elements, such as argon (Ar), helium (He), etc. are made up of only one atom of that element. But this is not the case with most of the non--metals. For example, a molecule of oxygen consists of two atoms of oxygen and hence it is known as a diatomic molecule, O2. If 3 atoms of oxygen unite into a molecule, instead of the usual 2, we get ozone, O3.
The number of atoms constituting a molecule is known as its atomicity.
Metals and some other elements, such as carbon, do not have a simple structure but consist of a very large and indefinite number of atoms bonded together.
Let us look at the atomicity of some non-metals.
Source: This topic is taken from NCERT TEXTBOOK
-
MOLECULES OF COMPOUNDS
MOLECULES OF COMPOUNDS
Atoms of different elements join together in definite proportions to form molecules of compounds. Few examples are given in Table.
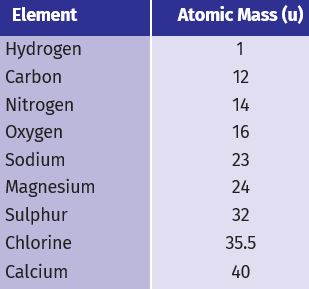
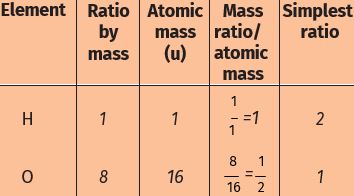
* Refer to Table for ratio by mass of atoms present in molecules and Table for atomic masses of elements. Find the ratio by the number of the atoms of elements in the molecules of compounds
i) Water ( H2O )
ii) Ammonia ( NH3 )
iii) Carbon dioxide ( CO2 )
* The ratio by the number of atoms for a water molecule can be found as follows:
* Thus, the ratio by number of atoms for water is H:O = 2:1
Source: This topic is taken from NCERT TEXTBOOK