-
DALTON’S ATOMIC THEORY
DALTON’S ATOMIC THEORY
It seemed that an atom was composed of protons and electrons, mutually balancing their charges. It also appeared that the protons were in the interior of the atom, for whereas electrons could easily be removed off but not protons. Now the big question was: what sort of structure did these particles of the atom form? We will find the answer to this question below.
We have learnt Dalton’s atomic theory in Chapter 3, which suggested that the atom was indivisible and indestructible. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of Dalton’s atomic theory. It was then considered necessary to know how electrons and protons are arranged within an atom. For explaining this, many scientists proposed various atomic models.
J.J. Thomson was the first one to propose a model for the structure of an atom.
Source: This topic is taken from NCERT TEXTBOOK
-
THOMSON’S MODEL OF AN ATOM
THOMSON ATOMIC MODEL
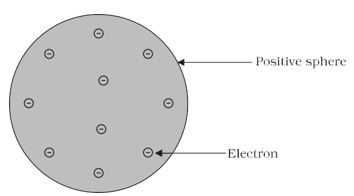
Thomson proposed the model of an atom to be similar to that of a Christmas pudding. The electrons, in a sphere of positive charge, were like currants (dry fruits) in a spherical Christmas pudding. We can also think of a watermelon, the positive charge in the atom is spread all over like the red edible part of the watermelon, while the electrons are studded in the positively charged sphere, like the seeds in the watermelon (Fig. 4.1)
Figure 4.1: Thomson’s model of an atom
Thomson proposed that:
i. An atom consists of a positively charged sphere and the electrons are embedded in it.
ii. The negative and positive charges are equal in magnitude. So, the atom as a whole is electrically neutral.
More to know
J.J. Thomson (1856- 1940), a British physicist, was born in Cheetham Hill, a suburb of Manchester, on 18 December 1856. He was awarded the Nobel prize in Physics in 1906 for his work on the discovery of electrons.
He directed the Cavendish Laboratory at Cambridge for 35 years and seven of his research assistants subsequently won Nobel prizes.
Question
On the basis of Thomson’s model of an atom, explain how the atom is neutral as a whole.
Source: This topic is taken from NCERT TEXTBOOK
-
DRAWBACKS OF THOMSON’S MODEL OF AN ATOM
DRAWBACKS OF THOMSON’S MODEL OF AN ATOM
Although Thomson's model explained that atoms are electrically neutral, the results of experiments carried out by other scientists could not be explained by this model, as we see below.
* Thomson’s atomic model failed to explain how the positive charge holds on the electrons inside the atom. It also failed to explain an atom’s stability.
* The theory did not mention anything about the nucleus of an atom.
* It was unable to explain the scattering experiment of Rutherford.
Source: This topic is taken from NCERT TEXTBOOK