-
DISTRIBUTION OF ELECTRONS IN DIFFERENT ORBITS(SHELLS)
DISTRIBUTION OF ELECTRONS IN DIFFERENT ORBITS(SHELLS)
The distribution of electrons into different orbits of an atom was suggested by Bohr and Bury.
The following rules are followed for writing the number of electrons in different energy levels or shells:
i. The maximum number of electrons present in a shell is given by the formula 2n2, where ‘n’ is the orbit number or energy level index, 1,2,3,…. Hence the maximum number of electrons in different shells are as follows:
first orbit or K-shell will be = 2 \(\times\) 12 = 2,
second orbit or L-shell will be = 2 \(\times\) 22 = 8,
third orbit or M-shell will be = 2 \(\times\) 32 = 18,
fourth orbit or N-shell will be = 2 \(\times\) 42= 32, and so on.
ii. The maximum number of electrons that can be acThe maximum number of electrons that can be accommodated in the outermost orbit is 8.
iii. Electrons are not accommodated in a given shell, unless the inner shells are filled. That is, the shells are filled in a step-wise manner.
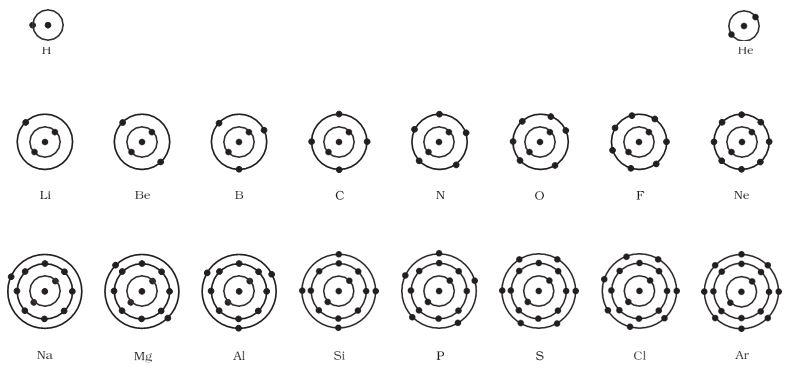
Atomic structure of the first eighteen elements is shown schematically in Fig. 4.4.
Figure 4.4: Schematic atomic structure of the first eighteen elements
Activity 4.2:
1. Make a static atomic model displaying electronic configuration of the first eighteen elements.
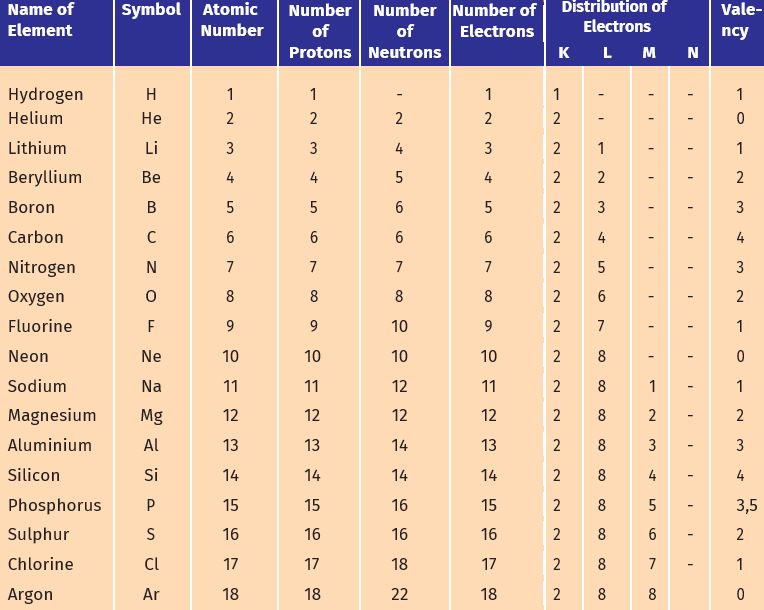
2. The composition of atoms of the first eighteen elements is given in Table 4.1
Table 4.1: Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells
Questions
1. Write the distribution of electrons in carbon and sodium atoms.
2. If K and L shells of an atom are full, then what would be the total number of electrons in the atom?
Source: This topic is taken from NCERT TEXTBOOK