-
ATOMIC NUMBER
ATOMIC NUMBER
We know that protons are present in the nucleus of an atom. It is the number of protons of an atom, which determines its atomic number. It is denoted by ‘Z’. All atoms of an element have the same atomic number, Z. In fact, elements are defined by the number of protons they possess. For hydrogen, Z = 1, because in hydrogen atom, only one proton is present in the nucleus. Similarly, for carbon, Z = 6. Therefore, the atomic number is defined as the total number of protons present in the nucleus of an atom.
Source: This topic is taken from NCERT TEXTBOOK
-
MASS NUMBER
MASS NUMBER
After studying the properties of the sub- atomic particles of an atom, we can conclude that mass of an atom is practically due to protons and neutrons alone. These are present in the nucleus of an atom. Hence protons and neutrons are also called nucleons. Therefore, the mass of an atom resides in its nucleus. For example, mass of carbon is 12 u because it has 6 protons and 6 neutrons, 6 u + 6 u = 12 u. Similarly, the mass of aluminium is 27 u (13 protons+14 neutrons). The mass number is defined as the sum of the total number of protons and neutrons present in the nucleus of an atom. It is denoted by ‘A’. In the notation for an atom, the atomic number, mass number and symbol of the element are to be written as:
For example, nitrogen is written as 14N7.
Questions
1. If number of electrons in an atom is 8 and number of protons is also 8, then (i) what is the atomic number of the atom? and (ii) what is the charge on the atom?
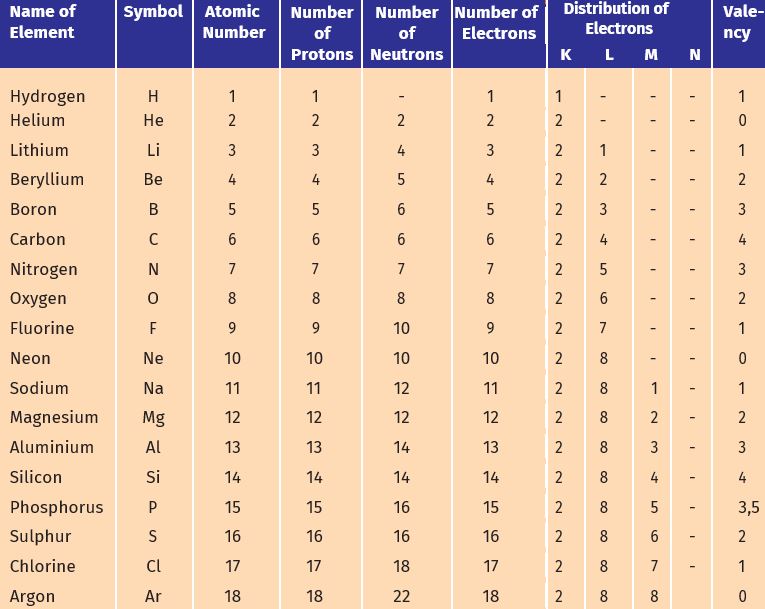
2. With the help of Table 4.1, find out the mass number of oxygen and sulphur atom.
Table 4.1: Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells
Source: This topic is taken from NCERT TEXTBOOK