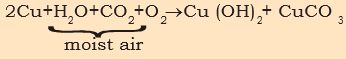-
REACTION WITH OXYGEN
REACTION WITH OXYGEN
You are familiar with the phenomenon of rusting of iron. Recall the reaction by which rust is formed. You had also performed in Class VII activity of burning a magnesium ribbon in air. You had learnt that in both the processes oxide formation takes place. Complete the following reactions of iron and magnesium with oxygen.
Iron (Fe) + Oxygen (O ) + Water (H2 O) \(\rightarrow\) ?
Magnesium (Mg) + Oxygen (O ) \(\rightarrow\) ?
Activity 4.3:
* Let us check the nature of rust formed as a result of the reaction between iron, oxygen and water.
* Collect a spoonful of rust and dissolve it in a very little amount of water.
* You will find that the rust remains suspended in water. Shake the suspension well.
* Test the solution with red and blue litmus papers (Fig. 4.3).
* What do you observe? Is the solution acidic or basic?
Figure 4.3: Testing the nature of rust
When a copper vessel is exposed to moist air for long, it acquires a dull green coating. The green material is a mixture of copper hydroxide (Cu(OH)2 ) and copper carbonate (CuCO3). The following is the reaction
Now recall the activity of burning magnesium ribbon. The ash obtained on burning magnesium ribbon is dissolved in water and tested for its acidic/basic nature. Is the solution acidic or basic? How do you ascertain this? You must have observed that the red litmus turns blue. So, oxide of magnesium is also basic in nature. In general, metallic oxides are basic in nature. Let us now observe the reaction of non-metals with oxygen.
Activity 4.4:
* (To be demonstrated by the teacher in the class)
* Take a small amount of powdered sulphur in a deflagrating spoon and heat it.
* If deflagrating spoon is not available, you may take a metallic cap of any bottle and wrap a metallic wire around it and give it the shape shown in Fig. 4.4 (a).
Figure 4.4(a): Burning of sulphur powder
Figure 4.4(b): Testing of the solution with litmus papers
The name of the product formed in the reaction of sulphur and oxygen is sulphur dioxide gas. When sulphur dioxide is dissolved in water sulphurous acid is formed. The reaction can be given as follows:
Sulphur dioxide (SO2 ) + Water (H2O) \(\rightarrow\) Sulphurous acid (H2SO3)
The sulphurous acid turns blue litmus paper red. Generally, oxides of non-metals are acidic in nature.
Source: This topic is taken from NCERT TEXTBOOK
-
REACTION WITH WATER
REACTION WITH WATER
Let us see how metals and non-metals react with water.
Sodium metal is very reactive. It reacts vigorously with oxygen and water. A lot of heat is generated in the reaction. It is, therefore, stored in kerosene.
Activity 4.5:
(To be demonstrated by the teacher. During demonstration special care should be taken that the size of the sodium metal piece is roughly the size of a wheat grain. It should be held with a pair of tongs.)
* Take a 250 mL beaker/glass tumbler. Fill half of it with water.
* Now carefully cut a small piece of sodium metal.
* Dry it using filter paper and wrap it in a small piece of cotton.
* Put the sodium piece wrapped in cotton into the beaker.
* Observe carefully. (During observation keep away from the beaker).
* When reaction stops touch the beaker.
* What do you feel? Has the beaker become hot?
* Test the solution with red and blue litmus papers. Is the solution acidic or basic?
Figure 4.5: Reaction of sodium with water
You observed that sodium reacts vigorously with water. Some other metals do not do so. For example, iron reacts with water slowly.
Generally, non-metals do not react with water though they may be very reactive in air. Such non-metals are stored in water. For example, phosphorus is a very reactive non-metal. It catches fire if exposed to air. To prevent the contact of phosphorus with atmospheric oxygen, it is stored in water.
Source: This topic is taken from NCERT TEXTBOOK
-
REACTION WITH ACIDS
REACTION WITH ACIDS
Let us see how metals and non-metals behave with acids.
Activity 4.6:
(Warning : Keep the mouth of the test tube away from your face. Use test tube holder to hold the test tube.)
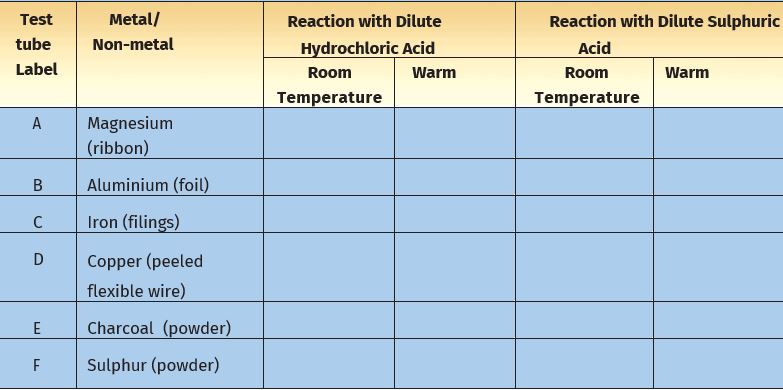
* Take samples of metals and non-metals listed in Table 4.5 in separate test tubes and label them as A, B, C, D, E, and F.
* With the help of a dropper add 5 mL of dilute hydrochloric acid to each test tube one by one.
* Observe the reactions carefully.
* If no reaction occurs in the cold solution, warm the test tube gently.
* Bring a burning matchstick near the mouth of each test tube.
* Repeat the same activity using dilute sulphuric acid instead of the dilute hydrochloric acid.
* Record your observations in Table 4.5.
Table 4.5: Reaction of Metals and Non-metals with Acids
Is there a difference in the way metals and non-metals react with acids? What could the ‘pop’ sound in some cases be due to when a burning match stick is brought near the mouth of the test tubes?
You must have found that non- metals generally do not react with acids but metals react with acids and produce hydrogen gas that burns with a ‘pop’ sound. You must have noticed that copper does not react with dilute hydrochloric acid even on heating but it reacts with sulphuric acid.
Source: This topic is taken from NCERT TEXTBOOK
-
REACTION WITH BASES
REACTION WITH BASES
Activity 4.7:
(To be demonstrated by the teacher. During the preparation of sodium hydroxide solution, care should be taken that pellets of sodium hydroxide are handled with a plastic spatula).
* Prepare a fresh solution of sodium hydroxide in a test tube by dissolving 3-4 pellets of it in 5 mL of water.
* Drop a piece of aluminium foil into it.
* Bring a burning match stick near the mouth of the test tube.
* Observe carefully.
* What does the ‘pop’ sound indicate? As before, the ‘pop’ sound indicates the presence of hydrogen gas.
* Metals react with sodium hydroxide to produce hydrogen gas.
* Reactions of non-metals with bases are complex.
Source: This topic is taken from NCERT TEXTBOOK
-
DISPLACEMENT REACTIONS
DISPLACEMENT REACTIONS
Activity 4.8:
* Recall the activity of the reaction between copper sulphate and iron that you performed in Class VII.
* Let us observe some more reactions of that kind.
* Take five 100 mL beakers and label them A, B, C, D and E.
* Take about 50 mL of water in each beaker.
* Dissolve in each beaker a teaspoonful of each substance as indicated in Fig. 4.6 (a).
* Keep the beakers undisturbed for some time.
* Record your observations in your note book.
Figure 4.6 (a) & (b): Displacement reactions
Beaker A:
Copper sulphate (CuSO4) + Zinc granule (Zn)
Beaker B:
Copper sulphate (CuSO4) + Iron nail (Fe)
Beaker C:
Zinc sulphate (ZnSO4) + Copper turnings (Cu)
Beaker D:
Iron sulphate (FeSO4) + Copper turnings (Cu)
Beaker E:
Zinc sulphate (ZnSO4) + Iron nail (Fe)
What changes do you observe in the various beakers? You have read that one metal displaces another metal from its compound in aqueous solution. In beaker ‘A’ zinc(Zn) replaces copper(Cu) from copper sulphate(CuSO4). That is why the blue colour of copper sulphate disappears and a powdery red mass of copper is deposited at the bottom of the beaker. The reaction can be represented as follows:
Copper Sulphate (CuSO4) + Zinc (Zn) \(\rightarrow\) Zinc Sulphate (ZnSO4 ) + Copper (Cu)
(Blue) (Colourless) (Red)
You can write down the reaction taking place in beaker ‘B’ in a similar manner.
There could have been displacement of zinc by copper in beaker ‘C’ and by iron in beaker ‘E’. Similarly iron could be displaced by copper in beaker ‘D’.
Since we do not see any change in beaker C, we can infer that copper is not able to replace zinc from zinc sulphate. But why? When zinc can replace copper in beaker ‘A’ why cannot copper replace zinc in beaker ‘C’? Remember that science is not arbitrary. It follows definite rules based on facts. And the rule here is that zinc is more reactive than copper and iron. A more reactive metal can replace a less reactive metal, but a less reactive one cannot replace a more reactive metal. Now you can understand why there are no displacement reactions in beakers D and E also. Can you guess the sequence of metals from more reactive to less reactive among zinc, iron and copper?
Source: This topic is taken from NCERT TEXTBOOK