-
WHAT IS COMBUSTION
WHAT IS COMBUSTION
Recall the activity of burning of magnesium ribbon performed in Class VII. We learnt that magnesium burns to form magnesium oxide and produces heat and light (Fig. 6.1). We can perform a similar activity with a piece of charcoal. Hold the piece with a pair of tongs and bring it near the flame of a candle or a Bunsen burner. What do you observe? We find that charcoal burns in air. We know that coal, too, burns in air producing carbon dioxide, heat and light.
Figure 6.1: Burning of magnesium
A chemical process in which a substance reacts with oxygen to give off heat is called combustion. The substance that undergoes combustion is said to be combustible. It is also called a fuel. The fuel may be solid, liquid or gas. Sometimes, light is also given off during combustion, either as a flame or as a glow.
Source: This topic is taken from NCERT TEXTBOOK
-
COMBUSTIBLE AND NONCOMBUSTIBLE SUBSTANCES
COMBUSTIBLE AND NONCOMBUSTIBLE SUBSTANCES
In the reactions mentioned above magnesium and charcoal are combustible substances.
Activity 6.1:
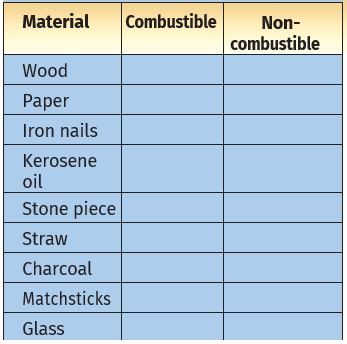
Collect some materials like straw, matchsticks, kerosene oil, paper, iron nails, stone pieces, glass etc.
Under the supervision of your teacher try to burn each of these materials one by one.
If combustion takes place mark the material combustible, otherwise mark it non-combustible (Table 6.1).
Table 6.1: Combustible and Non-combustible Substances
Can you name some more substances which are combustible? You can add those to Table 6.1.
Let us investigate conditions under which combustion takes place.
Activity 6.2:
(Caution : Be careful while handling burning candle).
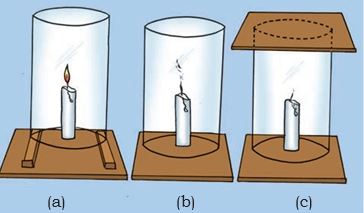
* Fix a lighted candle on a table. Put a glass chimney over the candle and rest it on a few wooden blocks in such a way that air can enter the chimney [Fig. 6.2(a)].
Figure 6.2: Experiment to show that air is essential for burning
* Observe what happens to the flame. Now remove the blocks and let the chimney rest on the table [Fig. 6.2(b)].
* Again observe the flame.
* Finally, put a glass plate over the chimney [Fig. 6.2(c)].
* Watch the flame again.
* What happens in the three cases? Does the flame flicker off?
* Does it flicker and give smoke? Does it burn unaffected?
* Can you infer anything at all about the role played by air in the process of burning?
We find that for combustion, air is necessary. The candle burns freely in case (a) when air can enter the chimney from below. In case (b), when air does not enter the chimney from below, the flame flickers and produces smoke. In case (c), the flame finally goes off because the air is not available.
Activity 6.3:
* Place a piece of burning wood or charcoal on an iron plate or Tawa.
* Cover it with a glass jar or a tumbler, or a transparent plastic jar.
* Observe what happens.
* Does charcoal stop burning after sometime?
* Can you think of the reason why it stops burning?
You might have heard that when the clothes of a person catch fire, the person is covered with a blanket to extinguish fire (Fig. 6.3). Can you guess why?
Figure 6.3: Blanket wrapped around a person whose clothes caught fire
Source: This topic is taken from NCERT TEXTBOOK
-
IGNITION TEMPERATURE
IGNITION TEMPERATURE
* Now recall some of your experiences. Does a matchstick burn by itself? How does it burn?
* You must have had an experience of burning a piece of paper. Does it burn when a burning matchstick is brought near it?
* Can you burn a piece of wood by bringing a lighted matchstick near it?
* Why do you have to use paper or kerosene oil to start fire in wood or coal?
* Have you heard of forest fires?
During extreme heat of summer, at some places dry grass catches fire. From the grass, it spreads to trees, and very soon the whole forest is on fire (Fig. 6.4). It is very difficult to control such fires.
Figure 6.4: Forest fire
Do these experiences tell you that different substances catch fire at different temperatures?
The lowest temperature at which a substance catches fire is called its ignition temperature.
Can you tell now why a matchstick does not catch fire on its own at room temperature?
Why does the matchstick start burning on rubbing it on the side of the matchbox?
We find that a combustible substance cannot catch fire or burn as long as its temperature is lower than its ignition temperature. Have you ever seen cooking oil catching fire when a frying pan is kept for long on a burning stove? Kerosene oil and wood do not catch fire on their own at room temperature. But, if kerosene oil is heated a little, it will catch fire. But if wood is heated a little, it would still not catch fire. Does it mean that ignition temperature of kerosene oil is lower than that of wood? Does it mean that we need to take special care in storing kerosene oil? The following activity shows that it is essential for a substance to reach ignition temperature to burn.
Activity 6.4:
(Caution : Be careful while handling burning candle).
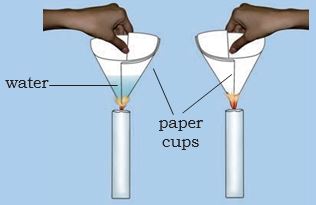
* Make two paper cups by folding a sheet of paper.
* Pour about 50 mL of water in one of the cups.
* Heat both the cups separately with a candle (Fig. 6.5).
* What do you observe?
Figure 6.5: Heating water in a paper cup
What happens to the empty paper cup? What happens to the paper cup with water? Does water in this cup become hot?
If we continue heating the cup, we can even boil water in the paper cup.
Can you think of an explanation for this phenomenon?
The heat supplied to the paper cup is transferred to water by conduction. So, in the presence of water, the ignition temperature of paper is not reached. Hence, it does not burn. The substances which have very low ignition temperature and can easily catch fire with a flame are called inflammable substances. Examples of inflammable substances are petrol, alcohol, Liquified Petroleum Gas (LPG) etc. Can you list some more inflammable substances?
Source: This topic is taken from NCERT TEXTBOOK
-
HISTORY OF MATCHSTICK
HISTORY OF MATCHSTICK
The history of the matchstick is very old. More than five thousand years ago small pieces of pinewood dipped in sulphur were used as matches in ancient Egypt. The modern safety match was developed only about two hundred years ago.
A mixture of antimony trisulphide, potassium chlorate and white phosphorus with some glue and starch was applied on the head of a match made of suitable wood. When struck against a rough surface, white phosphorus got ignited due to the heat of friction. This started the combustion of the match. However, white phosphorus proved to be dangerous both for the workers involved in the manufacturing of matches and for the users.
These days the head of the safety match contains only antimony trisulphide and potassium chlorate. The rubbing surface has powdered glass and a little red phosphorus (which is much less dangerous). When the match is struck against the rubbing surface, some red phosphorus gets converted into white phosphorus. This immediately reacts with potassium chlorate in the matchstick head to produce enough heat to ignite antimony trisulphide and start the combustion.
Source: This topic is taken from NCERT TEXTBOOK