-
OZONE LAYER - FORMATION, USES AND HARMFUL EFFECTS
OZONE LAYER - FORMATION, USES AND HARMFUL EFFECTS
Elemental oxygen is normally found in the form of a diatomic molecule. However, in the upper reaches of the atmosphere, a molecule containing three atoms of oxygen is found. This would mean a formula of O3 and this is called ozone. Unlike the normal diatomic molecule of oxygen, ozone is poisonous and we are lucky that it is not stable nearer to the Earth’s surface. But it performs an essential function where it is found. It absorbs harmful radiations from the Sun. This prevents those harmful radiations from reaching the surface of the Earth where they may damage many forms of life.
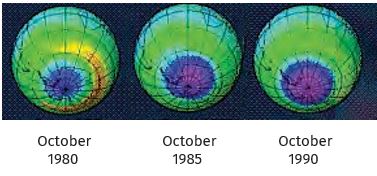
Recently it was discovered that this ozone layer was getting depleted. Various man-made compounds like CFCs (carbon compounds having both fluorine and chlorine which are very stable and not degraded by any biological process) were found to persist in the atmosphere. Once they reached the ozone layer, they would react with the ozone molecules. This resulted in a reduction of the ozone layer and recently they have discovered a hole in the ozone layer above the Antartica. It is difficult to imagine the consequences for life on Earth if the ozone layer dwindles further, but many people think that it would be better not to take chances. These people advocate working towards stopping all further damage to the ozone layer.
Figure 14.9: Satellite picture showing the hole (magenta colour) in the ozone layer over Antartica
Activity 14.13:
* Find out which other molecules are thought to damage the ozone layer.
* Newspaper reports often talk about the hole in the ozone layer.
* Find out whether the size of this hole is changing and in what manner scientists think this would affect life on Earth (Fig. 14.9).
Questions
1. What are the different states in which water is found during the water cycle?
2. Name two biologically important compounds that contain both oxygen and nitrogen.
3. List any three human activities which would lead to an increase in the carbon dioxide content of air.
4. What is the greenhouse effect?
5. What are the two forms of oxygen found in the atmosphere?
Source: This topic is taken from NCERT TEXTBOOK