-
EXPLANATION OF CHEMICAL EFFECTS OF ELECTRIC CURRENT
EXPLANATION OF CHEMICAL EFFECTS OF ELECTRIC CURRENT
In Class VII, we have learnt some effects of electric current. Can you list these effects? What effect does the current produce when it flows through a conducting solution? Let us find out.
Activity 14.6:
* Take out carbon rods carefully from two discarded cells.
* Clean their metal caps with sandpaper.
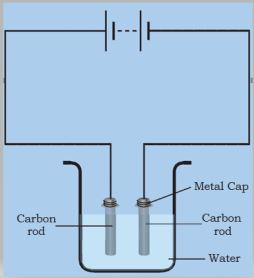
* Wrap copper wires around the metal caps of the carbon rods and join them to a battery (Fig. 14.5).
* We call these two rods electrodes .
(Instead of carbon rods, you may take two iron nails about 6 cm long.)
Figure 14.5: Passing current through water
* Pour a cupful of water in a glass/plastic bowl.
* Add a teaspoonful of salt or a few drops of lemon juice to water to make it more conducting.
* Now immerse the electrodes in this solution.
* Make sure that the metal caps of the carbon rods are outside the water.
* Wait for 3-4 minutes. Observe the electrodes carefully.
* Do you notice any gas bubbles near the electrodes?
* Can we call the change taking place in the solution a chemical change?
* Recall the definition of a chemical change that you learnt in Class VII.
The passage of an electric current through a conducting solution causes chemical reactions. As a result, bubbles of a gas may be formed on the electrodes. Deposits of metal may be seen on electrodes. Changes of colour of solutions may occur. The reaction would depend on what solution and electrodes are used. These are some of chemical effects of electric current.
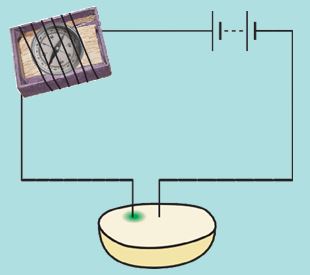
Boojho decided to test whether some fruits and vegetables also conduct electricity or not. He cut a potato into two halves and inserted the copper wires of a tester into it. Just then his mother called him and he forgot to take out the wires of the tester inserted into the potato. When he came back after half an hour, he noticed that there was a greenish-blue spot on the potato around one wire whereas there was no such spot around the other wire (Fig. 14.6).
He was surprised with this observation and along with Paheli repeated this activity many times. They found that it was always the wire connected to the positive terminal, which had a greenish-blue spot around it. They felt that this discovery was very useful because it could be used for identifying the positive terminal of a battery concealed in a box. They decided to report their finding to a children’s magazine.
Figure 14.6: Testing potato
Remember that Boojho set out to test whether potato conducted electricity or not. What he found was that current produced a chemical effect in the potato. To him, this was very exciting. In fact, this is how science sometimes works. You are looking for something and you discover something else. Many important discoveries have been made in this manner.
In 1800, a British chemist, William Nicholson (1753–1815), had shown that if electrodes were immersed in water, and a current was passed, bubbles of oxygen and hydrogen were produced. Oxygen bubbles formed on the electrode connected to the positive terminal of the battery and hydrogen bubbles formed on the other electrode.
Source: This topic is taken from NCERT TEXTBOOK
-
ELECTROPLATING
ELECTROPLATING
You know that a brand new bicycle has shiny handlebar and wheel rims. However, if these are accidentally scratched, the shiny coating comes off revealing a not so shiny surface beneath. You might have also seen women using ornaments, which appear to be made of gold. However, with repeated use, the gold coating wears off, revealing silver or some other metal beneath.
In both these cases, metal has a coating of another metal. Do you wonder how a layer of one metal can be deposited on top of another? Well, let us try doing it ourselves.
Activity 14.7:
* We will need copper sulphate and two copper plates of size around 10 cm × 4 cm.
* Take 250 mL of distilled water in a clean and dry beaker.
* Dissolve two teaspoonfuls of copper sulphate in it.
* Add a few drops of dilute sulphuric acid to the copper sulphate solution to make it more conducting.
* Clean copper plates with sandpaper.
* Now rinse them with water and dry them.
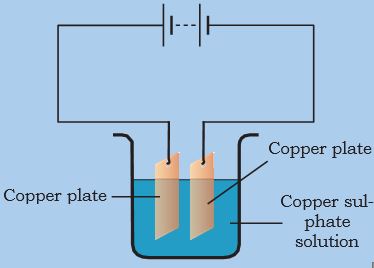
* Connect the copper plates to the terminals of a battery and immerse them in copper sulphate solution (Fig. 14.7).
Figure 14.7: A simple circuit showing electroplating
* Allow the current to pass for about 15 minutes.
* Now remove the electrodes from the solution and look at them carefully.
* Do you find any difference in any one of them? Do you find a coating over it? What colour is the coating?
* Note down the terminal of the battery with which this electrode is connected.
After doing the electroplating activity, Paheli interchanged the electrodes and repeated the activity. What do you think she would observe this time?
When electric current is passed through the copper sulphate solution, copper sulphate dissociates into copper and sulphate. The free copper gets drawn to the electrode connected to the negative terminal of the battery and gets deposited on it. But what about the loss of copper from the solution?
From the other electrode, a copper plate, an equal amount of copper gets dissolved in the solution. Thus, the loss of copper from the solution is restored and the process continues. This means that copper gets transferred from one electrode to the other.
Boojho could get only one copper plate. So he performed Activity 14.7 by connecting a carbon rod in place of the copper plate which was connected to the negative terminal of the battery. He succeeded in obtaining a coating of copper on carbon rod.
The process of depositing a layer of any desired metal on another material by means of electricity is called electroplating. It is one of the most common applications of the chemical effects of electric current.
Electroplating is a very useful process. It is widely used in industry for coating metal objects with a thin layer of a different metal (Fig.14.8). The layer of metal deposited has some desired property, which the metal of the object lacks. For example, chromium plating is done on many objects such as car parts, bath taps, kitchen gas burners, bicycle handlebars, wheel rims, and many others.
Chromium has a shiny appearance. It does not corrode. It resists scratches.
Figure 14.8: Some electroplated objects
However, chromium is expensive and it may not be economical to make the whole object out of chromium. So the object is made from a cheaper metal and only a coating of chromium over it is deposited. Jewellery makers electroplate silver and gold on less expensive metals. These ornaments have the appearance of silver or gold but are much less expensive.
Tin cans, used for storing food, are made by electroplating tin onto the iron. Tin is less reactive than iron. Thus, food does not come into contact with iron and is protected from getting spoilt.
Iron is used in bridges and automobiles to provide strength. However, iron tends to corrode and rust. So, a coating of zinc is deposited on iron to protect it from corrosion and the formation of rust.
In the electroplating factories, the disposal of the used conducting solution is a major concern. It is a polluting waste and there are specific disposal guidelines to protect the environment.
Source: This topic is taken from NCERT TEXTBOOK