-
Origin of Atomic Structure
The existence of atoms has been proposed since the time of early Indian and Greek philosophers (400 B.C.) who were of the view that atoms are the fundamental building blocks of matter. According to them, the continued subdivisions of matter would ultimately yield atoms that would not be further divisible. The word ‘atom’ has been derived from the Greek word ‘a-tomio’ which means ‘uncut-able’ or ‘non-divisible’. These earlier ideas were mere speculations and there was no way to test them experimentally. These ideas remained dormant for a very long time and were revived again by scientists in the nineteenth century.
The atomic theory of matter was first proposed on a firm scientific basis by John Dalton, a British school teacher in 1808. His theory called Dalton’s atomic theory regarded the atom as the ultimate particle of matter (Unit 1). Dalton’s atomic theory was able to explain the law of conservation of mass, law of constant composition, and law of multiple proportions very successfully. However, it failed to explain the results of many experiments, for example, it was known that substances like glass or ebonite when rubbed with silk or fur get electrically charged.
In this unit, we start with the experimental observations made by scientists towards the end of the nineteenth and beginning of the twentieth century. These established that atoms are made of sub-atomic particles, i.e., electrons, protons, and neutrons — a concept very different from that of Dalton.
-
Discovery of Sub-atomic Particle - Electron
DISCOVERY OF SUB-ATOMIC PARTICLES
An insight into the structure of atoms was obtained from the experiments on electrical discharge through gases. Before we discuss these results we need to keep in mind a basic rule regarding the behaviour of charged particles: “Like charges repel each other and unlike charges attract each other”.
Discovery of Electron
In 1830, Michael Faraday showed that if electricity is passed through a solution of an electrolyte, chemical reactions occurred at the electrodes, which resulted in the liberation and deposition of matter at the electrodes. He formulated certain laws which you will study in class XII. These results suggested the particulate nature of electricity.
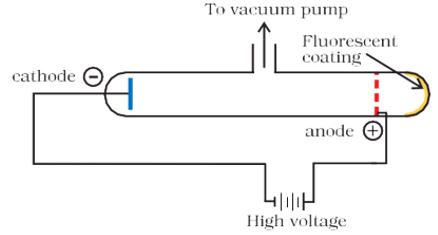
In mid-1850s many scientists mainly Faraday began to study electrical discharge in partially evacuated tubes, known as cathode ray discharge tubes. It is depicted in Fig. 2.1.
Fig. 2.1(a) A cathode ray discharge tube
A cathode ray tube is made of glass containing two thin pieces of metal, called electrodes, sealed in it. The electrical discharge through the gases could be observed only at very low pressures and at very high voltages. The pressure of different gases could be adjusted by evacuation of the glass tubes. When sufficiently high voltage is applied across the electrodes, current starts flowing through a stream of particles moving in the tube from the negative electrode (cathode) to the positive electrode (anode). These were called cathode rays or cathode ray particles. The flow of current from cathode to anode was further checked by making a hole in the anode and coating the tube behind anode with phosphorescent material zinc sulphide. When these rays, after passing through the anode, strike the zinc sulphide coating, a bright spot is developed on the coating [Fig. 2.1(b)].


Fig. 2.1(b) A cathode ray discharge tube with a perforated anode
The results of these experiments are summarised below.
Properties of Cathode rays
The cathode rays start from the cathode and move towards the anode.
These rays themselves are not visible but their behaviour can be observed with the help of certain kind of materials (fluorescent or phosphorescent) which glow when hit by them. Television picture tubes are cathode ray tubes and television pictures result due to fluorescence on the television screen coated with certain fluorescent or phosphorescent materials.
In the absence of electrical or magnetic field, these rays travel in straight lines (Fig. 2.2).
In the presence of the electrical or magnetic field, the behaviour of cathode rays is similar to that expected from negatively charged particles, suggesting that the cathode rays consist of negatively charged particles, called electrons.
The characteristics of cathode rays (electrons) do not depend upon the material of electrodes and the nature of the gas present in the cathode ray tube.
Thus, we can conclude that electrons are the basic constituent of all the atoms.
-
Charge to mass ratio of Electron
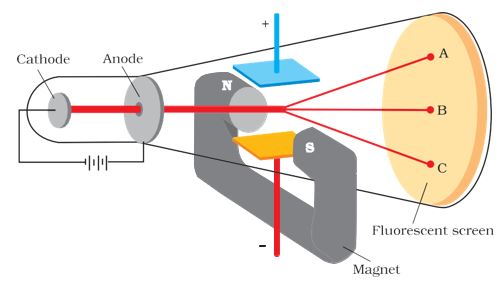
In 1897, British physicist J.J. Thomson measured the ratio of electrical charge (e-) to the mass of the electron (me ) by using cathode ray tube and applying electrical and magnetic field perpendicular to each other as well as to the path of electrons. When only an electric field is applied, the electrons deviate from their path and hit the cathode ray tube at point B. Similarly, when the only magnetic field is applied, electron strikes the cathode ray tube at point C. By carefully balancing the electrical and magnetic field strength, it is possible to bring back the electron to the path which is followed in the absence of electric or magnetic field and they hit the screen at point B. Thomson argued that the amount of deviation of the particles from their path in the presence of electrical or magnetic field depends upon:
(i) the magnitude of the negative charge on the particle, greater the magnitude of the charge on the particle, greater is the interaction with the electric or magnetic field, and thus greater is the deflection.
(ii) the mass of the particle — lighter the particle, greater the deflection.
(iii) the strength of the electrical or magnetic field — the deflection of electrons from its original path increases with the increase in the voltage across the electrodes, or the strength of the magnetic field.
By carrying out accurate measurements on the amount of deflections observed by the electrons on the electrical or magnetic field strength, Thomson was able to determine the value of e/me as:
\(\frac{e}{{{m}_{e}}}\,=\,1.758820\text{ }\times \text{ }{{10}^{11}}\,C\,k{{g}^{-1}}\)
Where me is the mass of the electron in kg and
e is the magnitude of the charge on the electron
in coulomb (C). Since electrons are negatively charged, the charge on an electron is –e.
-
Charge of Electron
R.A. Millikan (1868-1953) devised a method known as the oil drop experiment (1906-14), to determine the charge on the electrons. He found the charge on the electron to be– 1.6 × 10–19 C. The presently accepted value of the electrical charge is – 1.602176 × 10–19 C. The mass of the electron (me) was determined by combining these results with Thomson’s value of e/me ratio.
\({{m}_{e}}\,=\,\frac{e}{{}^{e}/{}_{{{m}_{e}}}}\,=\,\frac{1.602176\,\,\times \,{{10}^{-19}}\,C\,\,\,}{1.758820\text{ }\times \text{ }{{10}^{11}}\,C\,k{{g}^{-1}}}\)
= 9.1094×10–31 kg
-
Discovery of proton and Neutron
Discovery of Proton
Electrical discharge carried out in the modified cathode ray tube led to the discovery of canal rays carrying positively charged particles. The characteristics of these positively charged particles are listed below.
- Unlike cathode rays, the mass of positively charged particles depends upon the nature of gas present in the cathode ray tube. These are simply the positively charged gaseous ions.
- The charge to mass ratio of the particles depends on the gas from which these originate.
- Some of the positively charged particles carry a multiple of the fundamental unit of electrical charge.
- The behavior of these particles in the magnetic or electrical field is opposite to that observed for electron or cathode rays.
The smallest and lightest positive ion was obtained from hydrogen and was called a proton. This positively charged particle was characterised in 1919.
Discovery of Neutron
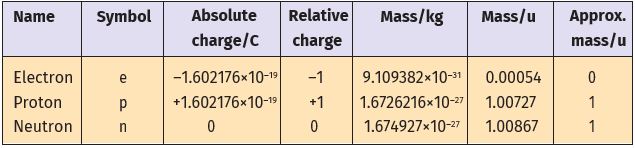
Later, a need was felt for the presence of electrically neutral particles as one of the constituents of atoms. These particles were discovered by Chadwick (1932) by bombarding a thin sheet of beryllium by \(\alpha\)-particles. When electrically neutral particles having a mass slightly greater than that of protons were emitted. He named these particles neutrons. The important properties of all these fundamental particles are given in Table 2.1.