-
Introduction to Atomic Models
Observations obtained from the experiments mentioned in the previous sections have suggested that Dalton’s indivisible atom is composed of sub-atomic particles carrying positive and negative charges. The major problems before the scientists after the discovery of sub-atomic particles were:
- to account for the stability of atom,
- to compare the behaviour of elements in terms of both physical and chemical properties,
- to explain the formation of different kinds of molecules by the combination of different atoms and,
- to understand the origin and nature of the characteristics of electromagnetic radiation absorbed or emitted by atoms.
Different atomic models were proposed to explain the distributions of these charged particles in an atom. Although some of these models were not able to explain the stability of atoms, two of these models, one proposed by J.J. Thomson and the other proposed by Ernest Rutherford are discussed below.
-
J.J. Thomson atomic model
J.J. Thomson atomic model
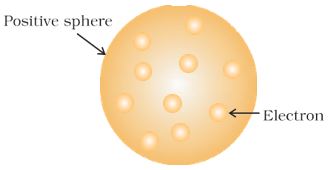
J. J. Thomson, in 1898, proposed that an atom possesses a spherical shape (radius approximately 10–10 m) in which the positive charge is uniformly distributed. The electrons are embedded into it in such a manner as to give the most stable electrostatic arrangement (Fig. 2.4).
Many different names are given to this model, for example, plum pudding, raisin pudding, or watermelon. This model can be visualised as a pudding or watermelon of positive charge with plums or seeds (electrons) embedded into it. An important feature of this model is that the mass of the atom is assumed to be uniformly distributed over the atom.
Although this model was able to explain the overall neutrality of the atom but was not consistent with the results of later experiments. Thomson was awarded Nobel Prize for physics in 1906, for his theoretical and experimental investigations on the conduction of electricity by gases.
-
Rutherford's Atomic Model
rutherford's atomic model
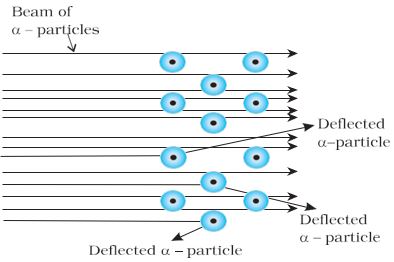
Rutherford and his students (Hans Geiger and Ernest Marsden) bombarded very thin gold foil with \(\alpha\)–particles. Rutherford’s famous \(\alpha\)–particle scattering experiment is represented in Fig. 2.5.
rutherford's scattering experiment
Fig-2.5: Schematic view of Rutherford’s scattering experiment.
When a beam of alpha (\(\alpha\)) particles is “shot” at a thin gold foil,
most of them pass through without much effect. Some, however, are deflected.
experiment and observation by rutherford
A stream of high energy \(\alpha\)–particles from a radioactive source was directed at a thin foil (thickness \(\alpha\) 100 nm) of gold metal. The thin gold foil had a circular fluorescent zinc sulphide screen around it. Whenever \(\alpha\)–particles struck the screen, a tiny flash of light was produced at that point.
The results of the scattering experiment were quite unexpected. According to thomson model of an atom, the mass of each gold atom in the foil should have been spread evenly over the entire atom, and a– particles had enough energy to pass directly through such a uniform distribution of mass. It was expected that the particles would slow down and change directions only by a small angle as they passed through the foil. It was observed that:
- most of the \(\alpha\)–particles passed through the gold foil undeflected.
- a small fraction of the \(\alpha\)–particles were deflected by small angles.
- a very few \(\alpha\)–particles (~1 in 20,000) bounced back, that is, were deflected by nearly 180°.
Conclusions of Rutherford's atomic model
On the basis of the observations, Rutherford drew the following conclusions regarding the structure of the atom:
- Most of the space in the atom is empty as most of the \(\alpha\)–particles passed through the foil undeflected.
- A few positively charged \(\alpha\)–particles were deflected. The deflection must be due to enormous repulsive force showing that the positive charge of the atom is not spread throughout the atom as Thomson had presumed. The positive charge has to be concentrated in a very small volume that repelled and deflected the positively charged \(\alpha\)–particles.
- Calculations by Rutherford showed that the volume occupied by the nucleus is negligibly small as compared to the total volume of the atom. The radius of the atom is about 10–10 m, while that of the nucleus is 10–15 m. One can appreciate this difference in size by realizing that if a cricket ball represents a nucleus, then the radius of atom would be about 5 km.
On the basis of the above observations and conclusions, Rutherford proposed the nuclear model of the atom. According to this model:
- The positive charge and most of the mass of the atom were densely concentrated in an extremely small region. This very small portion of the atom was called the nucleus by Rutherford.
- The nucleus is surrounded by electrons that move around the nucleus at a very high speed in circular paths called orbits. Thus, Rutherford’s model of atom resembles the solar system in which the nucleus plays the role of the sun and the electrons that of revolving planets.
- Electrons and the nucleus are held together by electrostatic forces of attraction.
-
Drawbacks of Rutherford Atomic Model
Drawbacks of Rutherford atomic model:
As you have learned above, Rutherford nuclear model of an atom is like a small-scale solar system with the nucleus playing the role of the massive sun and the electrons being similar to the lighter planets. When classical mechanics is applied to the solar system, it shows that the planets describe well-defined orbits around the sun. The gravitational force between the planets is given by the expression.
\(\left( G.\frac{{{m}_{1}}{{m}_{2}}}{{{r}^{2}}} \right)\)
Where and are the masses, r is the distance of separation of the masses and G is the gravitational constant. The theory can also calculate precisely the planetary orbits and these are in agreement with the experimental measurements.
The similarity between the solar system and nuclear model suggests that electrons should move around the nucleus in well-defined orbits. Further, the coulomb force (kq1q2/r2 where q1 and q2 are the charges, r is the distance of separation of the charges and k is the proportionality constant) between the electron and the nucleus is mathematically similar to the gravitational force. However, when a body is moving in an orbit, it undergoes acceleration even if it is moving at a constant speed in an orbit because of changing direction. So an electron in the nuclear model describing planet-like orbits is under acceleration. According to the electromagnetic theory of Maxwell, charged particles when accelerated should emit electromagnetic radiation (This feature does not exist for planets since they are uncharged). Therefore, an electron in an orbit will emit radiation, the energy carried by radiation comes from electronic motion. The orbit will thus continue to shrink. Calculations show that it should take an electron only 10–8 s to spiral into the nucleus. But this does not happen. Thus, the Rutherford model cannot explain the stability of an atom.
If the motion of an electron is described on the basis of the classical mechanics and electromagnetic theory, you may ask that since the motion of electrons in orbits is leading to the instability of the atom, then why not consider electrons as stationary around the nucleus. If the electrons were stationary, electrostatic attraction between the dense nucleus and the electrons would pull the electrons toward the nucleus to form a miniature version of Thomson’s model of an atom.
Another serious drawback of the Rutherford model is that it says nothing about the distribution of the electrons around the nucleus and the energies of these electrons.