-
Atomic Number and Mass Number
Atomic Number and Mass Number
Atomic number (Z)
The presence of a positive charge on the nucleus is due to the protons in the nucleus. As established earlier, the charge on the proton is equal but opposite to that of an electron. The number of protons present in the nucleus is equal to the atomic number (Z ). For example, the number of protons in the hydrogen nucleus is 1, in the sodium atom, it is 11, therefore their atomic numbers are 1 and 11 respectively. In order to keep the electrical neutrality, the number of electrons in an atom is equal to the number of protons (atomic number, Z ). For example, the number of electrons in the hydrogen atom and sodium atom are 1 and 11 respectively.
Atomic number (Z) = number of protons in the nucleus of an atom = number of electrons in a neutral atom
Mass number (A)
While the positive charge of the nucleus is due to protons, the mass of the nucleus, due to protons and neutrons. As discussed earlier protons and neutrons present in the nucleus are collectively known as nucleons. The total number of nucleons is termed as mass number (A) of the atom.
mass number (A) = number of protons (Z ) + number of neutrons (n)
-
Isotopes and Isobars
Representation of an Atom
The composition of any atom can be represented by using the normal element symbol (X) with super-script on the left-hand side as the atomic mass number (A) and subscript (Z ) on the left-hand side as the atomic number (i.e., \({}_{Z}^{A}X\)).
Isotopes
Atoms with identical atomic numbers but a different atomic mass number are known as Isotopes. In other words (according to equation 2.4), it is evident that the difference between the isotopes is due to the presence of the different number of neutrons present in the nucleus. For example, considering of hydrogen atom again, 99.985% of hydrogen atoms contain only one proton
This isotope is called protium ( \(_{1}^{1}H\)). The rest of the percentage of hydrogen atom contains two other isotopes, the one containing 1 proton and 1 neutron is called deuterium (\(_{1}^{2}D\), 0.015%) and the other one possessing 1 proton and 2 neutrons is called tritium ( \(_{1}^{3}T\) ). The latter isotope is found in trace amounts on the earth.
Other examples of commonly occurring isotopes are: carbon atoms containing 6, 7, and 8 neutrons besides 6 protons ( \(_{6}^{12}C\), \(_{6}^{13}C\), \(_{6}^{14}C\)); chlorine atoms containing 18 and 20 neutrons besides 17 protons ( \(_{17}^{35}Cl\), \(_{17}^{37}Cl\) ).
Lastly, an important point to mention regarding isotopes is that the chemical properties of atoms are controlled by the number of electrons, which are determined by the number of protons in the nucleus. The number of neutrons present in the nucleus has very little effect on the chemical properties of an element. Therefore, all the isotopes of a given element show the same chemical behaviour.
Isobars
Isobars are the atoms with the same mass number but the different atomic number for example, \(_{6}^{14}C\) and \(_{7}^{14}N\)..
-
Origin of Bohr's Atomic Model
Historically, results observed from the studies of interactions of radiations with the matter have provided immense information regarding the structure of atoms and molecules. Neils Bohr utilized these results to improve upon the model proposed by Rutherford. Two developments played a major role in the formulation of Bohr’s model of an atom. These were:
- The dual character of electromagnetic radiation means that radiations possess both wave-like and particle-like properties, and
- Experimental results regarding atomic spectra.
First, we will discuss the dual nature of electromagnetic radiation. Experimental results regarding atomic spectra will be discussed in Section 2.4.
-
Wave Nature of Electromagnetic Radiation
In the mid-nineteenth century, physicists actively studied the absorption and emission of radiation by heated objects. These are called thermal radiations. They tried to find out what the thermal radiation is made. It is now a well-known fact that thermal radiations consist of electromagnetic waves of various frequencies or wavelengths. It is based on a number of modern concepts, which were unknown in the mid-nineteenth century. First, active study of thermal radiation laws occurred in the 1850s, and the theory of electromagnetic waves and the emission of such waves by accelerating charged particles was developed
in the early 1870s by James Clerk Maxwell, which was experimentally confirmed later by Heinrich Hertz. Here, we will learn some facts about electromagnetic radiation.
James Maxwell (1870) was the first to give a comprehensive explanation about the interaction between the charged bodies and the behavior of electrical and magnetic fields on the macroscopic level. He suggested that when an electrically charged particle moves under acceleration, alternating electrical and magnetic fields are produced and transmitted. These fields are transmitted in the form of waves called electromagnetic waves or electromagnetic radiation.
Light is the form of radiation known from early days and speculation about its nature dates back to remote ancient times. In earlier days (Newton) light was supposed to be made of particles (corpuscles). It was only in the 19th century when the wave nature of light was established.
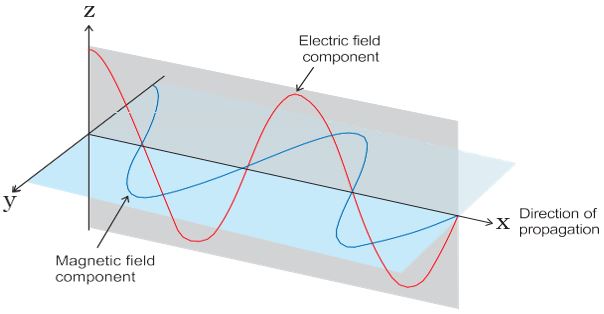
Maxwell was again the first to reveal that light waves are associated with an oscillating electric and magnetic character (Fig. 2.6).
Fig.2.6 The electric and magnetic field components of an electromagnetic wave. These components have the same wavelength, frequency, speed, and amplitude, but they vibrate in two mutually perpendicular planes.
Although electromagnetic wave motion is complex in nature, we will consider here only a few simple properties.
- The oscillating electric and magnetic fields produced by oscillating charged particles are perpendicular to each other and both are perpendicular to the direction of propagation of the wave. A simplified picture of an electromagnetic wave is shown in Fig. 2.6.
- Unlike sound waves or waves produced in water, electromagnetic waves do not require a medium and can move in a vacuum.
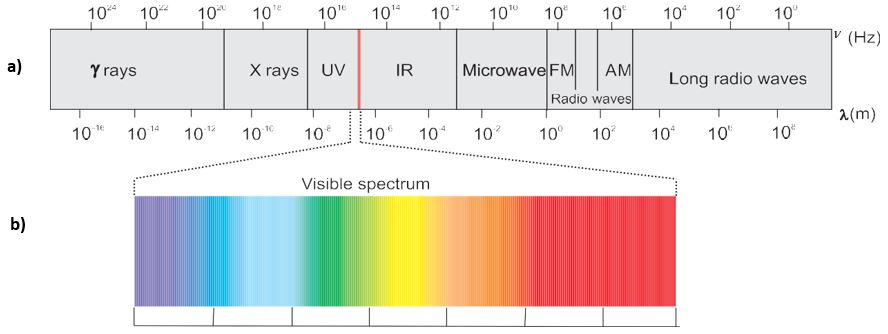
- It is now well established that there are many types of electromagnetic radiations, which differ from one another in wavelength (or frequency). These constitute what is called the electromagnetic spectrum (Fig. 2.7). Different regions of the spectrum are identified by different names. Some examples are radiofrequency region around 106 Hz, used for broadcasting; microwave region around 1010 Hz used for radar; the infrared region around 1013 Hz used for heating; the ultraviolet region around 1016Hz a component of sun’s radiation. The small portion around 1015 Hz, is what is ordinarily called visible light. It is only this part which our eyes can see (or detect). Special instruments are required to detect non-visible radiation.
- Different kinds of units are used to represent electromagnetic radiation.
Fig. 2.7 (a) The spectrum of electromagnetic radiation.
(b) Visible spectrum. The visible region is only a small part of the entire spectrum.
These radiations are characterized by the properties, namely, frequency (n ) and wavelength (l).
The SI unit for frequency (n ) is hertz (Hz, s–1), after Heinrich Hertz. It is defined as the number of waves that pass a given point in one second.
The wavelength should have the units of length and as you know that the SI units of length is meter (m). Since electromagnetic radiation consists of different kinds of waves of much smaller wavelengths, smaller units are used. Fig.2.7 shows various types of electromagnetic radiations which differ from one another in wavelengths and frequencies.
In a vacuum all types of electromagnetic radiations, regardless of wavelength, travel at the same speed, i.e., 3.0 × 108 m s–1 (2.997925
× 108 ms–1, to be precise). This is called the speed of light and is given the symbol ‘c‘. The frequency (n ), wavelength (l), and velocity of light (c) are related by the equation (2.5).
c = n l (2.5)
The other commonly used quantity specially in spectroscopy, is the wavenumber (
).
It is defined as the number of wavelengths per unit length. Its units are reciprocal of wavelength unit, i.e., m–1. However the commonly used unit is cm–1 (not SI unit).
-
Planck's Quantum Theory
Some of the experimental phenomena such as diffraction and interference can be explained by the wave nature of the electromagnetic radiation. However, the following are some of the observations which could not be explained with the help of even the electromagnetic theory of 19th-century physics (known as classical physics):
- the nature of emission of radiation from hot bodies (black -body radiation)
- ejection of electrons from a metal surface when radiation strikes it (photoelectric effect)
- variation of heat capacity of solids as a function of temperature
- Line spectra of atoms with special reference to hydrogen.
These phenomena indicate that the system can take energy only in discrete amounts. All possible energies cannot be taken up or radiated.
It is noteworthy that the first concrete explanation for the phenomenon of the black body radiation mentioned above was given by Max Planck in 1900. Let us first try to understand this phenomenon, which is given below:
Hot objects emit electromagnetic radiation over a wide range of wavelengths. At high temperatures, an appreciable proportion of radiation is in the visible region of the spectrum. As the temperature is raised, a higher proportion of short wavelength (blue light) is generated. For example, when an iron rod is heated in a furnace, it first turns to dull red and then progressively becomes more and more red as the temperature increases. As this is heated further, the radiation emitted becomes white and then becomes blue as the temperature becomes very high. This means that red radiation is most intense at a particular temperature and blue radiation is more intense at another temperature. This means intensities of radiations of different wavelengths emitted by a hot body depend upon its temperature. By the late 1850’s it was known that objects made of different material and kept at different temperatures emit different amounts of radiation. Also, when the surface of an object is irradiated with light (electromagnetic radiation), a part of radiant energy is generally reflected as such, a part is absorbed and a part of it is transmitted. The reason for incomplete absorption is that ordinary objects are as a rule imperfect absorbers of radiation. An ideal body, which emits and absorbs radiations of all frequencies uniformly, is called a black body, and the radiation emitted by such a body is called black body radiation. In practice, no such body exists. Carbon black approximates fairly closely to the black body. A good physical approximation to a black body is a cavity with a tiny hole, which has no other opening. Any ray entering the hole will be reflected by the cavity walls and will be eventually absorbed by the walls. A black body is also a perfect radiator of radiant energy. Furthermore, a black body is in thermal equilibrium with its surroundings. It radiates the same amount of energy per unit area as it absorbs from its surrounding at any given time. The amount of light emitted (intensity of radiation) from a black body and its spectral distribution depends only on its temperature. At a given temperature, the intensity of radiation emitted increases with the increase of wavelength reaches a maximum value at a given wavelength, and then starts decreasing with the further increase of wavelength, as shown in Fig. 2.8. Also, as the temperature increases, the maxima of the curve shifts to a short wavelength. Several attempts were made to predict the intensity of radiation as a function of wavelength.
Fig. 2.8 Wavelength-intensity relationship
Fig. 2.8(a) Black body
But the results of the above experiment could not be explained satisfactorily on the basis of the wave theory of light. Max Planck arrived at a satisfactory relationship by making an assumption that absorption and emission of radiation arise from oscillator i.e., atoms in the wall of the black body. Their frequency of oscillation is changed by interaction with oscillators of electromagnetic radiation. Planck assumed that radiation could be sub-divided into discrete chunks of energy. He suggested that atoms and molecules could emit or absorb energy only in discrete quantities and not in a continuous manner. He gave the name quantum to the smallest quantity of energy that can be emitted or absorbed in the form of electromagnetic radiation. The energy (E) of a quantum of radiation is proportional to its frequency (n ) and is expressed by equation (2.6).
E = hu (2.6)
The proportionality constant, ‘h’ is known as Planck’s constant and has the value 6.626×10–34 J s.
With this theory, Planck was able to explain the distribution of intensity in the radiation from the black body as a function of frequency or wavelength at different temperatures.
Quantization has been compared to standing on a staircase. A person can stand on any step of a staircase, but it is not possible for him/her to stand in between the two steps. The energy can take any one of the values from the following set, but cannot take on any values between them.
E = 0, hu, 2hu, 3hu....nhu.....
-
Photoelectric Effect
Photoelectric Effect
In 1887, H. Hertz performed a very interesting experiment in which electrons (or electric current) were ejected when certain metals (for example potassium, rubidium, cesium, etc.) were exposed to a beam of light as shown in Fig.2.9. The phenomenon is called the Photoelectric effect. The results observed in this experiment were:
- The electrons are ejected from the metal surface as soon as the beam of light strikes the surface, i.e., there is no time lag between the striking of the light beam and the ejection of electrons from the metal surface.
- The number of electrons ejected is proportional to the intensity or brightness of the light.
For each metal, there is a characteristic minimum frequency,n0 (also known as threshold frequency) below which photoelectric effect is not observed. At a frequency n >n0, the ejected electrons come out with certain kinetic energy. The kinetic energies of these electrons increase with the increase of frequency of the light used.
All the above results could not be explained on the basis of the laws of classical physics. According to the latter, the energy content of the beam of light depends upon the brightness of the light. In other words, the number of electrons ejected and kinetic energy associated with them should depend on the brightness of the light. It has been observed that though the number of electrons ejected does depend upon the brightness of light, the kinetic energy of the ejected electrons does not. For example, red light [n = (4.3 to 4.6) × 1014 Hz] of any brightness (intensity) may shine on a piece of potassium metal for hours but no photoelectrons are ejected. But, as soon as even a very weak yellow light (n = 5.1–5.2 × 1014 Hz) shines on the potassium metal, the photoelectric effect is observed. The threshold frequency (n0) for potassium metal is 5.0×1014 Hz.
Einstein (1905) was able to explain the photoelectric effect using Planck’s quantum theory of electromagnetic radiation as a starting point.
Shining a beam of light onto a metal surface can, therefore, be viewed as shooting a beam of particles, the photons. When a photon of sufficient energy strikes an electron in the atom of the metal, it transfers its energy instantaneously to the electron during the collision and the electron is ejected without any time lag or delay. The greater the energy possessed by the photon, the greater will be the transfer of energy to the electron and the greater the kinetic energy of the ejected electron. In other words, the kinetic energy of the ejected electron is proportional to the frequency of the electromagnetic radiation. Since the striking photon has an energy equal to hn and the minimum energy required to eject the electron is hn0 (also called work function, W0; Table 2.2), then the difference in energy (hn – hn0 ) is transferred as the kinetic energy of the photoelectron. Following the conservation of energy principle, the kinetic energy of the ejected electron is given by equation 2.7.
\(hv\,=\,h{{v}_{0}}\,+\,\frac{1}{2}{{m}_{e}}{{v}^{2}}\)where me is the mass of the electron and v is the velocity associated with the ejected electron. Lastly, a more intense beam of light consists
of the larger number of photons, consequently, the number of electrons ejected is also larger as compared to that in an experiment in which a beam of the weaker intensity of light is employed.