-
Thermal Properties of Matter
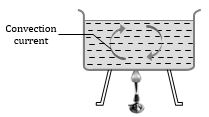
Convection
Mode of transfer of heat by means of migration of material particles of medium is called convection. It is of two types.
(1) Natural convection: This arise due to difference of densities at two places and is a consequence of gravity because on account of gravity the hot light particles rise up and cold heavy particles try setting down. It mostly occurs on heating a liquid/fluid.
(2) Forced convection: If a fluid is forced to move to take up heat from a hot body then the convection process is called forced convection. In this case Newton's law of cooling holds good. According to which rate of loss of heat from a hot body due to moving fluid is directly proportional to the surface area of body and excess temperature of body over its surroundings i.e.
\(\frac{Q}{t}\propto A(T-{{T}_{0}})\Rightarrow\frac{Q}{t}=h\,A(T-{{T}_{0}})\)
where h = Constant of proportionality called convection coefficient, T = Temperature of body and T0 = Temperature of surrounding
Convection coefficient (h) depends on properties of fluid such as density, viscosity, specific heat and thermal conductivity.
(3) Natural convection takes place from bottom to top while forced convection in any direction.
(4) In case of natural convection, convection currents move warm air upwards and cool air downwards. That is why heating is done from base, while cooling from the top.
(5) Natural convection plays an important role in ventilation, in changing climate and weather and in forming land and sea breezes and trade winds.
(6) Natural convection is not possible in a gravity free region such as a free falling lift or an orbiting satellite.
(7) The force of blood in our body by heart helps in keeping the temperature of body constant.
(8) If liquids and gases are heated from the top (so that convection is not possible) they transfer heat (from top to bottom) by conduction.
(9) Mercury though a liquid is heated by conduction and not by convection.
Heat Transfer by Convection
(1) The process of the transfer of heat from one place to another place without heating the intervening medium is called radiation.
(2) Precisely it is electromagnetic energy transfer in the form of electromagnetic wave through any medium. It is possible even in vacuum e.g. the heat from the sun reaches the earth through radiation.
(3) The wavelength of thermal radiations ranges from \(7.8\times {{10}^{-7}}\,m\) to \(4\times {{10}^{-4}}\,m\). They belong to infra-red region of the electromagnetic spectrum. That is why thermal radiations are also called infra-red radiations.
(4) Medium is not required for the propagation of these radiations.
(5) They produce sensation of warmth in us but we can’t see them.
(6) Everybody whose temperature is above zero Kelvin emits thermal radiation.
(7) Their speed is equal to that of light i.e. \((=3\times {{10}^{8}}\,m/s)\).
(8) Their intensity is inversely proportional to the square of distance of point of observation from the source (i.e. \(I\propto 1/{{d}^{2}}\)).
(9) Just as light waves, they follow laws of reflection, refraction, interference, diffraction and polarization.
(10) When these radiations fall on a surface then exert pressure on that surface which is known as radiation pressure.
(11) While travelling these radiations travel just like photons of other electromagnetic waves. They manifest themselves as heat only when they are absorbed by a substance.
(12) Spectrum of these radiations cannot be obtained with the help of glass prism because it absorbs heat radiations. It is obtained by quartz or rock salt prism because these materials do not have free electrons and interatomic vibrational frequency is greater than the radiation frequency, hence they do not absorb heat radiations.
(13) Diathermanous Medium: A medium which allows heat radiations to pass through it without absorbing them is called diathermanous medium. Thus, the temperature of a diathermanous medium does not increase irrespective of the amount of the thermal radiations passing through it e.g., dry air, \(S{{O}_{2}}\), rock salt (NaCl).
(i) Dry air does not get heated in summers by absorbing heat radiations from sun. It gets heated through convection by receiving heat from the surface of earth.
(ii) In winters heat from sun is directly absorbed by human flesh while the surrounding air being diathermanous is still cool. This is the reason that sun’s warmth in winter season appears very satisfying to us.
(14) Athermanous medium: A medium which partly absorbs heat rays is called a thermos medium. As a result, temperature of an athermanous medium increases when heat radiations pass through it e.g., wood, metal, moist air, simple glass, human flesh etc.
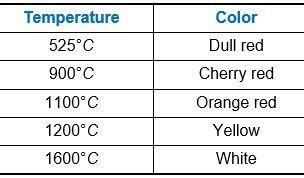
Color of Heated Object
When a body is heated, all radiations having wavelengths from zero to infinity are emitted.
(1) Radiations of longer wavelengths are predominant at lower temperature.
(2) The wavelength corresponding to maximum emission of radiations shifts from longer wavelength to shorter wavelength as the temperature increases. Due to this the colour of a body appears to be changing.
(3) A blue flame is at a higher temperature than a yellow flame.
Table: Variation of color of a body on heating
Interaction of Radiation with Matter
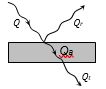
When thermal radiations (Q) fall on a body, they are partly reflected, partly absorbed and partly transmitted.
(1) \(Q={{Q}_{a}}+{{Q}_{r}}+{{Q}_{t}}\)
(2) \(\frac{{{Q}_{a}}}{Q}+\frac{{{Q}_{r}}}{Q}+\frac{{{Q}_{t}}}{Q}=a+r+t=1\)
(3) \(a=\frac{{{Q}_{a}}}{Q}\)= Absorptance or absorbing power
\(r=\frac{{{Q}_{r}}}{Q}\) = Reflectance or reflecting power
\(t=\frac{{{Q}_{t}}}{Q}\)= Transmittance or transmitting power
(4) r, a and t all are the pure ratios so they have no unit and dimension.
(5) Different bodies
(i) If a = t = 0 and r = 1 \(\rightarrow\) body is perfect reflector
(ii) If r = t = 0 and a = 1 \(\rightarrow\) body is perfectly black body
(iii) If, a = r = 0 and t = 1 \(\rightarrow\) body is perfect transmitter
(iv) If t = 0 \(\Rightarrow\) \(r+a=1\,\,\text{or}\,\,a=1-r\) i.e. good reflectors are bad absorbers.
Emissive Power, Absorptive Power and Emissivity
If temperature of a body is more than it's surrounding then body emits thermal radiation
(1) Monochromatic Emittance or Spectral emissive power (e\(\lambda\)) : For a given surface it is defined as the radiant energy emitted per sec per unit area of the surface with in a unit wavelength around \(\lambda\) i.e. lying between \(\left( \lambda -\frac{1}{2} \right)\) to \(\left( \lambda +\frac{1}{2} \right)\).
Spectral emissive power\(({{e}_{\lambda }})=\frac{\text{Energy}}{\text{Area}\times \text{time}\times \text{wavelength}}\)
Unit :\(\frac{Joule}{{{m}^{2}}\times sec\times {A ^0}}\) and Dimension :\([M{{L}^{-1}}{{T}^{-3}}]\)
(2) Total emittance or total emissive power (e) : It is defined as the total amount of thermal energy emitted per unit time, per unit area of the body for all possible wavelengths. \(e=\int_{\,0}^{\,\infty }{\,\,{{e}_{\lambda }}d\lambda }\)
Unit : \(\frac{Joule}{{{m}^{2}}\times \sec }\) or \(\frac{Watt}{{{m}^{2}}}\) and Dimension :\([M{{T}^{-3}}]\)
(3) Monochromatic absorptance or spectral absorptive power (a\(\lambda\)) : It is defined as the ratio of the amount of the energy absorbed in a certain time to the total heat energy incident upon it in the same time, both in the unit wavelength interval. It is dimensionless and unit less quantity. It is represented by a\(\lambda\).
(4) Total absorptance or total absorpting power (a): It is defined as the total amount of thermal energy absorbed per unit time, per unit area of the body for all possible wavelengths.
\(a=\int_{\,0}^{\,\infty }{{{a}_{\lambda }}d\lambda }\)
(5) Emissivity (\(\varepsilon \)) : Emissivity of a body at a given temperature is defined as the ratio of the total emissive power of the body (e) to the total emissive power of a perfect black body (E) at that temperature i.e\(\varepsilon =\frac{e}{E}\) (\(\varepsilon \) \(\rightarrow\) read as epsilon)
(i) For perfectly black body \(\varepsilon \) = 1
(ii) For highly polished body \(\varepsilon \) = 0
(iii) But for practical bodies emissivity (\(\varepsilon \)) lies between zero and one (0 <\(\varepsilon \) < 1).
Perfectly Black Body
(1) A perfectly black body is that which absorbs completely the radiations of all wavelengths incident on it.
(2) As a perfectly black body neither reflects nor transmits any radiation, therefore the absorptance of a perfectly black body is unity i.e. t = 0 and r = 0 \(\Rightarrow\) a = 1.
(3) We know that the color of an opaque body is the color (wavelength) of radiation reflected by it. As a black body reflects no wavelength so, it appears black, whatever be the color of radiations incident on it.
(4) When perfectly black body is heated to a suitable high temperature, it emits radiation of all possible wavelengths. For example, temperature of the sun is very high (6000 K approx.) it emits all possible radiation so it is an example of black body.
(5) Ferry’s black body : A perfectly black body can’t be realized in practice. The nearest example of an ideal black body is the Ferry’s black body. It is a doubled walled evacuated spherical cavity whose inner wall is blackened. The space between the wall is evacuated to prevent the loss of heat by conduction and radiation. There is a fine hole in it. All the radiations incident upon this hole are absorbed by this black body. If this black body is heated to high temperature, then it emits radiations of all wavelengths. It is the hole which is to be regarded as a black body and not the total enclosure.
(6) A perfectly black body cannot be realized in practice but materials like Platinum black or Lamp black come close to being ideal black bodies. Such materials absorb 96% to 85% of the incident radiations.
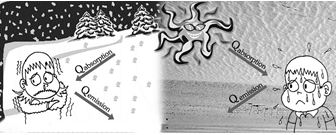
Prevost Theory of Heat Exchange
(1) Everybody emits heat radiations at all finite temperature (Except 0 K) as well as it absorbs radiations from the surroundings.
(2) Exchange of energy along various bodies takes place via radiation.
(3) The process of heat exchange among various bodies is a continuous phenomenon.
(4) At absolute zero temperature (0 K or – 273°C) this law is not applicable because at this temperature the heat exchange among various bodies ceases.
(5) If Qemission > Qabsorbed \(\rightarrow\) temperature of body decreases and consequently the body appears colder.
If Qemission < Qabsorbed\(\rightarrow\) temperature of body increases and it appears hotter.
If Qemission = Qabsorbed \(\rightarrow\) temperature of body remains constant (thermal equilibrium)
Kirchoff's Law
According to this law the ratio of emissive power to absorptive power is same for all surfaces at the same temperature and is equal to the emissive power of a perfectly black body at that temperature. Hence \(\frac{{{e}_{1}}}{{{a}_{1}}}=\frac{{{e}_{2}}}{{{a}_{2}}}=...\,\,{{\left( \frac{E}{A} \right)}_{\text{Perfectly black body}}}\)
But for perfectly black body A = 1 i.e. \(\frac{e}{a}=E\)
If emissive and absorptive powers are considered for a particular wavelength \(\lambda\),\(\left( \frac{{{e}_{\lambda }}}{{{a}_{\lambda }}} \right)={{({{E}_{\lambda }})}_{\text{black}}}\)
Now since (E\(\lambda\))black is constant at a given temperature, according to this law if a surface is a good absorber of a particular wavelength it is also a good emitter of that wavelength.This in turn implies that a good absorber is a good emitter (or radiator)
Applications of Kirchoff's Law
(1) Sand is rough black, so it is a good absorber and hence in deserts, days (when radiation from the sun is incident on sand) will be very hot. Now in accordance with Kirchoff’s law, good absorber is a good emitter so nights (when sand emits radiation) will be cold. This is why days are hot and nights are cold in desert.
(2) Sodium vapors, on heating, emit two bright yellow lines. These are called D1, D2 lines of sodium. When continuous white light from an arc lamp is made to pass through sodium vapors at low temperature, the continuous spectrum is intercepted by two dark lines exactly in the same places as D1 and D2 lines. Hence sodium vapors when cold, absorbs the same wavelength, as they emit while hot. This is in accordance with Kirchoff's law.
(3) When a shining metal ball having some black spots on its surface is heated to a high temperature and is seen in dark, the black spots shine brightly, and the shining ball becomes dull or invisible. The reason is that the black spots on heating absorb radiation and so emit these in dark while the polished shining part reflects radiations and absorb nothing and so does not emit radiations and becomes invisible in the dark.
(4) When a green glass is heated in furnace and taken out, it is found to glow with red light. This is because red and green are complimentary colors. At ordinary temperatures, a green glass appears green, because it transmits green color and absorb red color strongly. According to Kirchoff's law, this green glass, on heating must emit the red color, which is absorbed strongly. Similarly, when a red glass is heated to a high temperature it will glow with green light.
(5) A person with black skin experiences more heat and colder as compared to a person of white skin because when the outside temperature is greater, the person with black skin absorbs more heat and when the outside temperature is less the person with black skin radiates more energy.
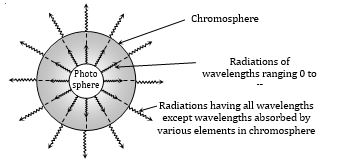
(6) Kirchoff' law also explains the Fraunhoffer lines:
(i) Sun's inner most part (photosphere) emits radiation of all wavelength at high temperature.
(ii) When these radiations enter in outer part (chromosphere) of sun, few wavelength are absorbed by some terrestrial elements (present in vapor form at lower temperature)
(iii) These absorbed wavelengths, which are missing appear as dark lines in the spectrum of the sun called Fraunhoffer lines.
(iv) During total solar eclipse these lines appear bright because the gases and vapour present in the chromosphere start emitting those radiation which they had absorbed.
Stefan's Law
According to it the radiant energy emitted by a perfectly black body per unit area per sec (i.e. emissive power of black body) is directly proportional to the fourth power of its absolute temperature, i.e. \(E\propto {{T}^{4}}\) \(\Rightarrow\) E = \(\sigma\)T4
where s is a constant called Stefan’s constant having dimension \([M{{T}^{-3}}{{\theta }^{-4}}]\) and value \(5.67\times {{10}^{-8}}W/{{m}^{2}}{{K}^{4}}\).
(i) For ordinary body : e = \(\varepsilon\)E \(=\varepsilon \sigma {{T}^{4}}\)
(ii) Radiant energy : If Q is the total energy radiated by the ordinary body then \(e=\frac{Q}{A\times t}=\varepsilon \sigma {{T}^{4}}\) \(\Rightarrow\)\(Q=A\,\varepsilon \sigma {{T}^{4}}t\)
(iii) Radiant power (P) : It is defined as energy radiated per unit area i.e. \(P=\frac{Q}{t}=A\varepsilon \sigma {{T}^{4}}\).
(iv) If an ordinary body at temperature T is surrounded by a body at temperature T0, then Stefan's law may be put as
\(e=\varepsilon \,\sigma \,({{T}^{4}}-T_{0}^{4})\)
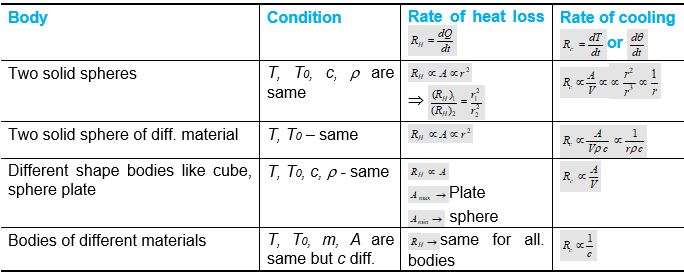
Rate of Loss of Heat (RH) and Rate of Cooling (RC)
(1) Rate of loss of heat (or initial rate of loss of heat) : If an ordinary body at temperature T is placed in an environment of temperature T0 (T0 < T) then heat loss by radiation is given by
\(\Delta Q={{Q}_{\text{emission}}}-{{Q}_{\text{absorption}}}=A\varepsilon \,\sigma ({{T}^{4}}-T_{0}^{4})\)
(2) Rate of loss of heat \(({{R}_{H}})=\frac{dQ}{dt}=A\varepsilon \,\sigma ({{T}^{4}}-T_{0}^{4})\)
If two bodies are made of same material, have same surface finish and are at the same initial temperature then \(\frac{dQ}{dt}\propto A\Rightarrow\frac{{{\left( \frac{dQ}{dt} \right)}_{1}}}{{{\left( \frac{dQ}{dt} \right)}_{2}}}=\frac{{{A}_{1}}}{{{A}_{2}}}\)
(3) Initial rate of fall in temperature (Rate of cooling): If m is the body and c is the specific heat then
\(\frac{dQ}{dt}=mc.\frac{dT}{dt}=mc\frac{d\theta }{dt} \,\,\, (\because \ Q=mc\,\Delta T\ \,\,and \,\,dT=d\theta )\)
(i)Rate of cooling \(({{R}_{c}})=\frac{d\theta }{dt}=\frac{(dQ/dt)}{mc}=\frac{A\varepsilon \,\sigma }{mc}({{T}^{4}}-T_{0}^{4})\)
\(=\frac{A\varepsilon \,\sigma }{V\rho \,c}({{T}^{4}}-T_{0}^{4})\); where m = density (\(\rho\)) \(\times\) volume (V)
(ii)For two bodies of the same material under identical environments, the ratio of their rate of cooling is\(\frac{{{({{R}_{c}})}_{1}}}{{{({{R}_{c}})}_{2}}}=\frac{{{A}_{1}}}{{{A}_{2}}}.\frac{{{V}_{2}}}{{{V}_{1}}}\)
(4) Dependence of rate of cooling : When a body cools by radiation the rate of cooling depends on
(i) Nature of radiating surface i.e. greater the emissivity, faster will be the cooling.
(ii) Area of radiating surface, i.e. greater the area of radiating surface, faster will be the cooling.
(iii) Mass of radiating body i.e. greater the mass of radiating body slower will be the cooling.
(iv) Specific heat of radiating body i.e. greater the specific heat of radiating body slower will be cooling.
(v) Temperature of radiating body i.e. greater the temperature of body faster will be cooling.
(vi) Temperature of surrounding i.e. greater the temperature of surrounding slower will be cooling.
Comparison of rate of heat loss (RH) and rate of cooling (Rc) for different bodies
Newton's Law of Cooling
When the temperature difference between the body and its surrounding is not very large i.e. T – T0 = \(\Delta\)T then \({{T}^{4}}-T_{0}^{4}\) may be approximated as\(4T_{0}^{3}\Delta T\)
By Stefan’s law, \(\frac{dT}{dt}=\frac{A\varepsilon \sigma }{mc}[{{T}^{4}}-T_{0}^{4}]\)
Hence \(\frac{dT}{dt}=\frac{A\varepsilon \sigma }{mc}4T_{0}^{3}\Delta T\Rightarrow\frac{dT}{dt}\propto \Delta T\) or \(\frac{d\theta }{dt}\propto \theta -{{\theta }_{0}}\)
i.e., if the temperature of body is not very different from surrounding, rate of cooling is proportional to temperature difference between the body and its surrounding. This law is called Newton’s law of cooling.
(1) Greater the temperature difference between body and its surrounding greater will be the rate of cooling.
(2) If\(\theta ={{\theta }_{0}}\), \(\frac{d\theta }{dt}=0\) i.e. a body can never be cooled to a temperature lesser than its surrounding by radiation.
(3) If a body cools by radiation from \(\theta _{1}^{o}C\) to\(\theta _{2}^{o}C\) in time t, then \(\frac{d\theta }{dt}=\frac{{{\theta }_{1}}-{{\theta }_{2}}}{t}\) and \(\theta ={{\theta }_{av}}=\frac{{{\theta }_{1}}+{{\theta }_{2}}}{2}\). The Newton’s law of cooling becomes \(\left[ \frac{{{\theta }_{1}}-{{\theta }_{2}}}{t} \right]=K\left[ \frac{{{\theta }_{1}}+{{\theta }_{2}}}{2}-{{\theta }_{0}} \right]\).This form of law helps in solving numericals.
(4) Practical examples
(i) Hot water loses heat in smaller duration as compared to moderate warm water.
(ii) Adding milk in hot tea reduces the rate of cooling.
Cooling Curves
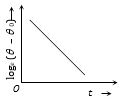
(1) Curve between log(\(\theta\) – \(\theta\)0) and time
As \(\frac{d\theta }{dt}\propto -(\theta -{{\theta }_{0}})\Rightarrow \,\frac{d\theta }{(\theta -{{\theta }_{0}})}=-Kdt\)
Integrating \({{\log }_{e}}(\theta -{{\theta }_{0}})=-Kt+C\)
\({{\log }_{e}}(\theta -{{\theta }_{0}})=-Kt+{{\log }_{e}}A\)
This is a straight line with negative slope
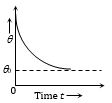
(2) Curve between temperature of body and time
As \({{\log }_{e}}(\theta -{{\theta }_{0}})=-Kt+{{\log }_{e}}A\Rightarrow{{\log }_{e}}\frac{\theta -{{\theta }_{0}}}{A}=-Kt\)
Þ \(\theta -{{\theta }_{0}}=A{{e}^{-kt}}\)
which indicates temperature decreases exponentially with increasing time.
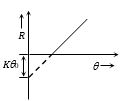
(3) Curve between the rate of cooling
(R) and body temperature (\(\theta\)).
\(R=K(\theta -{{\theta }_{0}})=K\theta -K{{\theta }_{0}}\)
This is a straight line intercept R-axis at \(-K{{\theta }_{0}}\)
(4) Curve between rate of cooling (R)
and temperature difference between body (\(\theta\)) and surrounding (\(\theta\)0)
\(R\propto (\theta -{{\theta }_{0}})\). This is a straight line passing through origin.
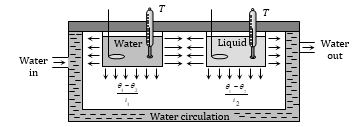
Determination of Specific Heat of Liquid
If volume, radiating surface area, nature of surface, initial temperature and surrounding of water and given liquid are equal and they are allowed to cool down (by radiation) then rate of loss of heat and fall in temperature of both will be same
i.e. \({{\left( \frac{dQ}{dt} \right)}_{\text{water}}}={{\left( \frac{dQ}{dt} \right)}_{\text{liquid}}}\)
\(({{m}_{W}}{{c}_{W}}+W)\frac{({{\theta }_{1}}-{{\theta }_{2}})}{{{t}_{1}}}=({{m}_{l}}{{c}_{l}}+W)\frac{({{\theta }_{1}}-{{\theta }_{2}})}{{{t}_{2}}}\)
or \(\left[ \frac{{{m}_{W}}{{c}_{W}}+W}{{{t}_{1}}} \right]=\left[ \frac{{{m}_{l}}{{c}_{l}}+W}{{{t}_{2}}} \right]\)
W = mccc = Water equivalent of calorimeter, where mc and cc are mass and specific heat of calorimeter.
If density of water and liquid is \(\rho\) and \(\rho\)¢ respectively then \({{m}_{W}}=V{{\rho }_{W}}\) and \({{m}_{l}}=V\rho {{\,}_{l}}\)
Specific heat of liquid \({{c}_{l}}=\frac{1}{{{m}_{l}}}\left[ \frac{{{t}_{l}}}{{{t}_{W}}}({{m}_{W}}{{c}_{W}}+W)-W \right]\)
Distribution of Energy in the Spectrum of Black Body
A perfectly black body emits radiation of all possible wavelength.
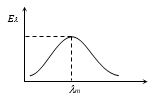
Langley and later on Lummer and Pringsheim investigated the distribution of energy amongst the different wavelengths in the thermal spectrum of a black body radiation. The results obtained are shown in figure. From these curves it is clear that
(1) At a given temperature energy is not uniformly distributed among different wavelengths.
(2) At a given temperature intensity of heat radiation increases with wavelength, reaches a maximum at a particular wavelength and with further increase in wavelength it decreases.
(3) For all wavelengths an increase in temperature causes an increase in intensity.
(4) The area under the curve will represent the total intensity of radiation at a particular temperature i.e. Area = E \(=\int{{{E}_{\lambda }}d\lambda }\)
From Stefan's law E = \(\sigma\)T4 \(\Rightarrow\) Area under E\(\lambda\) - \(\lambda\) curve (A) \(\propto\) T4
(5) The energy (Emax) emitted corresponding to the wavelength of maximum emission (\(\lambda\)m) increases with fifth power of the absolute temperature of the black body i.e., \({{E}_{\max }}\propto {{T}^{5}}\)
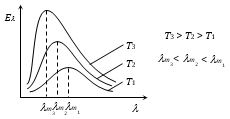
Wien's Displacement Law
According to Wien's law the product of wavelength corresponding to maximum intensity of radiation and temperature of body (in Kelvin) is constant, i.e.\({{\lambda }_{m}}T=b=\text{constant}\) where b is Wien's constant and has value \(2.89\times {{10}^{-3}}m\text{-}K\).
As the temperature of the body increases, the wavelength at which the spectral intensity (E\(\lambda\)) is maximum shifts towards left. Therefore, it is also called Wien's displacement law.
This law is of great importance in ‘Astrophysics’ as through the analysis of radiations coming from a distant star, by finding \({{\lambda }_{m}}\) the temperature of the star \(T(=b/{{\lambda }_{m}})\) is determined.
Law of Distribution of Energy (Plank's Hypothesis)
(1) The theoretical explanation of black body radiation was done by Planck.
(2) According to Plank's atoms of the walls of a uniform temperature enclosure behave as oscillators, each with a characteristic frequency of oscillation.
(3) These oscillations emits electromagnetic radiations in the form of photons (The radiation coming out from a small hole in the enclosure are called black body radiation). The energy of each photon is hn. Where n is the frequency of oscillator and h is the Plank's constant. Thus emitted energies may be hn, 2hn, 3hn ... nhn but not in between.
According to Planck's law \({{E}_{\lambda }}d\lambda =\frac{8\pi hc}{{{\lambda }^{5}}}\,\frac{1}{[{{e}^{hc/\lambda KT}}-1]}\,d\lambda\)
where c = speed of light and k = Boltzmann's constant. This equation is known as Plank's radiation law. It is correct and complete law of radiation
(4) This law is valid for radiations of all wavelengths ranging from zero to infinite.
(5) For radiations of short wavelength \(\left( \lambda <<\frac{hc}{KT} \right)\) Planck's law reduces to Wien's energy distribution law\({{E}_{\lambda }}d\lambda =\frac{A}{{{\lambda }^{5}}}{{e}^{-\,B/\lambda T}}d\lambda \)
(6) For radiations of long wavelength \(\left( \lambda >>\frac{hc}{KT} \right)\) Planck's law reduces to Rayleigh-Jeans energy distribution law \({{E}_{\lambda }}d\lambda =\frac{8\pi KT}{{{\lambda }^{4}}}d\lambda \)
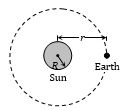
Temperature of the Sun and Solar Constant
If R is the radius of the sun and T its temperature, then the energy emitted by the sun per sec through radiation in accordance with Stefan’s law will be given by
\(P=A\sigma {{T}^{4}}=4\pi {{R}^{2}}\sigma {{T}^{4}}\)
In reaching earth this energy will spread over a sphere of radius r (= average distance between sun and earth); so the intensity of solar radiation at the surface of earth (called solar constant S) will be given by
\(S=\frac{P}{4\pi {{r}^{2}}}=\frac{4\pi {{R}^{2}}\sigma {{T}^{4}}}{4\pi {{r}^{2}}}\)
i.e\(T={{\left[ {{\left( \frac{r}{R} \right)}^{2}}\frac{S}{\sigma } \right]}^{1/4}}\) \(={{\left[ {{\left( \frac{1.5\times {{10}^{8}}}{7\times {{10}^{5}}} \right)}^{2}}\times \frac{1.4\times {{10}^{3}}}{5.67\times {{10}^{-8}}} \right]}^{1/4}}\tilde{-}5800\,K\)
As \(r=1.5\times {{10}^{8}}\)km, \(R=7\times {{10}^{5}}\)km,
\(S=2\frac{cal}{c{{m}^{2}}min}=1.4\frac{kW}{{{m}^{2}}}\) and\(\sigma =5.67\times {{10}^{-8}}\frac{W}{{{m}^{2}}{{K}^{4}}}\)
This result is in good agreement with the experimental value of temperature of sun, i.e., 6000 K.