-
Explanation of Hybridisation
Explanation of Hybridisation
In order to explain the characteristic geometrical shapes of polyatomic molecules like CH4, NH3 and H2O etc., Pauling introduced the concept of hybridisation. According to him the atomic orbitals combine to form new set of equivalent orbitals known as hybrid orbitals. Unlike pure orbitals, the hybrid orbitals are used in bond formation.The phenomenon is known as hybridisation which can be defined as the process of intermixing of the orbitals of slightly different energies so as to redistribute their energies, resulting in the formation of new set of orbitals of equivalent energies and shape. For example when one 2s and three 2p-orbitals of carbon hybridise, there is the formation of four new sp3 hybrid orbitals.
Salient features of hybridisation: The main features of hybridisation are as under :
1. The number of hybrid orbitals is equal to the number of the atomic orbitals that get hybridised.
2. The hybridised orbitals are always equivalent in energy and shape.
3. The hybrid orbitals are more effective in forming stable bonds than the pure atomic orbitals.
4. These hybrid orbitals are directed in space in some preferred direction to have minimum repulsion between electron pairs and thus a stable arrangement. Therefore, the type of hybridisation indicates the geometry of the molecules.Important conditions for hybridisation
1. The orbitals present in the valence shell of the atom are hybridised.
2. The orbitals undergoing hybridisation should have almost equal energy.
3. Promotion of electron is not essential condition prior to hybridisation.
4. It is not necessary that only half filled orbitals participate in hybridisation.In some cases, even filled orbitals of valence shell take part in hybridisation.
-
Types of Hybridisation - sp hybridisation
Types of Hybridisation - sp hybridisation
There are various types of hybridisation involving s, p and d orbitals.The different types of hybridisation are as under:
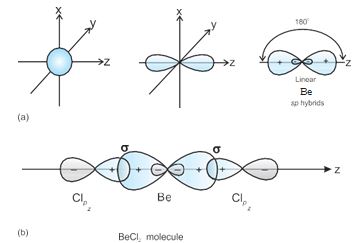
1. sp hybridisation: This type of hybridisation involves the mixing of one s and one p orbital resulting in the formation of two equivalent sp hybrid orbitals.The suitable orbitals for sp hybridisation are s and pz, if the hybrid orbitals are to lie along the z-axis.
Each sp hybrid orbitals has 50% s-character and 50% p-character. Such a molecule in which the central atom is sp-hybridised and linked directly to two other central atoms possesses linear geometry. This type of hybridisation is also known as diagonal hybridisation.
The two sp hybrids point in the opposite direction along the z-axis with projecting positive lobes and very small negative lobes, which provides more effective overlapping resulting in the formation of stronger bonds.
Example of molecule having sp hybridisation
BeCl2: The ground state electronic configuration of Be is 1s22s2. In the exited state one of the 2s-electrons is promoted to vacant 2p orbital to account for its bivalency. One 2s and one 2p-orbital gets hybridised to form two sp hybridised orbitals.These two sp hybrid orbitals are oriented in opposite direction forming an angle of 180°.Each of the sp hybridised orbital overlaps with the 2p-orbital of chlorine axially and form two Be-Cl sigma bonds. This is shown in Fig. 4.10.
Fig.4.10 (a) Formation of sp hybrids from s and p orbitals; (b) Formation of the linear BeCl2 molecule.
-
Types of Hybridisation - sp2 hybridisation
Types of Hybridisation - sp2 hybridisation
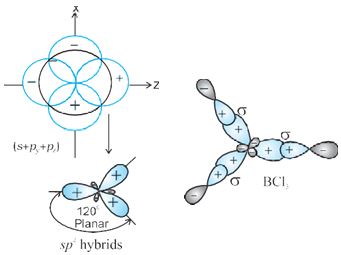
sp2 hybridisation : In this hybridisation there is involvement of one s and two p-orbitals in order to form three equivalent sp2 hybridised orbitals. For example,in BCl3 molecule,the ground state electronic configuration of central boron atom is 1s22s22p1.In the excited state, one of the 2s electrons is promoted to vacant 2p orbital as
Fig.4.11 Formation of sp2 hybrids and the BCl3 molecule
a result boron has three unpaired electrons. These three orbitals (one 2s and two 2p) hybridise to form three sp2 hybrid orbitals. The three hybrid orbitals so formed are oriented in a trigonal planar arrangement and overlap with 2p orbitals of chlorine to form three B-Cl bonds. Therefore, in BCl3 (Fig. 4.11), the geometry is trigonal planar with ClBCl bond angle of 120°.
-
Types of Hybridisation - sp3 hybridisation
Types of Hybridisation - sp3 hybridisation
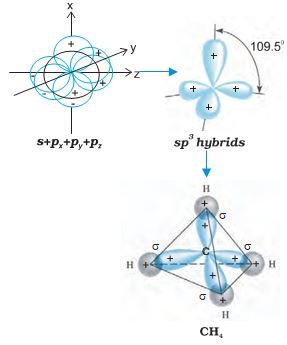
sp3 hybridisation: This type of hybridisation can be explained by taking the example of CH4 molecule in which there is mixing of one s-orbital and three p-orbitals of the valence shell to form four sp3 hybrid orbital of equivalent energies and shape. There is 25% s-character and 75% p-character in each sp3 hybrid orbital.The four sp3 hybrid orbitals so formed are directed towards the four corners of the tetrahedron. The angle between sp3 hybrid orbital is 109.5° as shown in Fig. 4.12.
Fig.4.12 Formation of sp3 hybrids by the combination of s , px , py and pz atomic orbitals of carbon and the formation of CH4 molecule
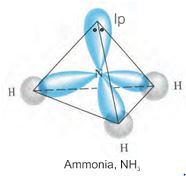
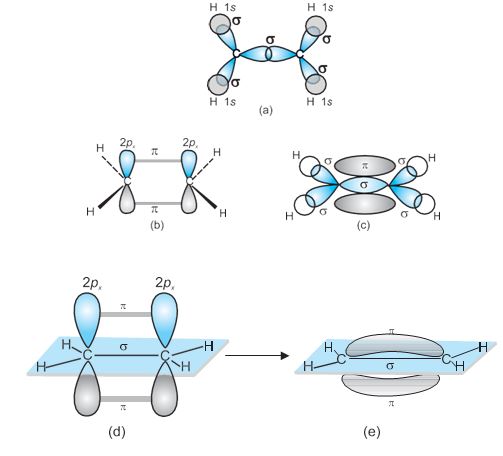
The structure of NH3 and H2O molecules can also be explained with the help of sp3 hybridisation. In NH3, the valence shell (outer) electronic configuration of nitrogen in the ground state is 2S2\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaaGOmaiaadc % hadaqhaaWcbaGaamiEaaqaaiaaigdaaaGccaaIYaGaamiCamaaDaaa % leaacaWG5baabaGaaGymaaaakiaaikdacaWGWbWaa0baaSqaaiaadQ % haaeaacaaIXaaaaaaa!40CF! 2p_x^12p_y^12p_z^1\) having three unpaired electrons in the sp3 hybrid orbitals and a lone pair of electrons is present in the fourth one. These three hybrid orbitals overlap with 1s orbitals of hydrogen atoms to form three N–H sigma bonds. We know that the force of repulsion between a lone pair and a bond pair is more than the force of repulsion between two bond pairs of electrons. The molecule thus gets distorted and the bond angle is reduced to 107° from 109.5°. The geometry of such a molecule will be pyramidal as shown in Fig. 4.13.
Fig.4.13 Formation of NH3 molecule
In case of H2O molecule, the four oxygen orbitals (one 2s and three 2p) undergo sp3 hybridisation forming four sp3 hybrid orbitals out of which two contain one electron each and the other two contain a pair of electrons.These four sp3 hybrid orbitals acquire a tetrahedral geometry, with two corners occupied by hydrogen atoms while the other two by the lone pairs.The bond angle in this case is reduced to 104.5° from 109.5° (Fig. 4.14) and the molecule thus acquires a V-shape or angular geometry.
Fig.4.14 Formation of H2O molecule.
-
Other Examples of sp3,sp2 and sp Hybridisation
Other Examples of sp3,sp2 and sp Hybridisation
sp3 Hybridisation in C2H6 molecule: In ethane molecule both the carbon atoms assume sp3 hybrid state. One of the four sp3 hybrid orbitals of carbon atom overlaps axially with similar orbitals of other atom to form sp3-sp3 sigma bond while the other three hybrid orbitals of each carbon atom are used in forming sp3–s sigma bonds with hydrogen atoms as discussed in section 4.6.1(iii). Therefore in ethane C–C bond length is 154 pm and each C–H bond length is 109 pm.
sp2 Hybridisation in C2H6 : In the formation of ethene molecule, one of the sp2 hybrid orbitals of carbon atom overlaps axially with sp2 hybridised orbital of another carbon atom to form C–C sigma bond. While the other two sp2 hybrid orbitals of each carbon atom are used for making sp2–s sigma bond with two hydrogen atoms. The unhybridised orbital (2p or 2py ) of one carbon atom overlaps sidewise with the similar orbital of the other carbon atom to form weak \(\pi\) bond, which consists of two equal electron clouds distributed above and below the plane of carbon and hydrogen atoms.
Thus, in ethene molecule, the carbon- carbon bond consists of one sp2–sp2 sigma bond and one pi (\(\pi\)) bond between p orbitals which are not used in the hybridisation and are perpendicular to the plane of molecule;the bond length 134 pm. The C–H bond is sp2–s sigma with bond length 108 pm.The H–C–H bond angle is 117.6° while the H–C–C angle is 121°. The formation of sigma and pi bonds in ethene is shown in Fig. 4.15.
Fig. 4.15 Formation of sigma and pi bonds in ethene
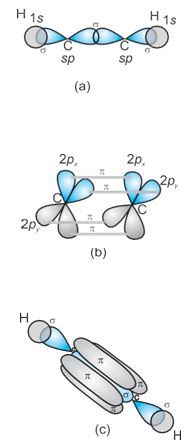
sp Hybridisation in C2H2: In the formation of ethyne molecule, both the carbon atoms undergo sp-hybridisation having two unhybridised orbital i.e., 2py and 2px.
One sp hybrid orbital of one carbon atom overlaps axially with sp hybrid orbital of the other carbon atom to form C–C sigma bond, while the other hybridised orbital of each carbon atom overlaps axially with the half filled s orbital of hydrogen atoms forming \(\sigma\) bonds. Each of the two unhybridised p orbitals of both the carbon atoms overlaps sidewise to form two \(\pi\) bonds between the carbon atoms. So the triple bond between the carbon atoms. So the triple bond between the two carbon atoms is made up of one sigma and two pi bonds as shown in Fig. 4.16.
Fig.4.16 Formation of sigma and pi bonds in ethyne.
-
Hybridisation of Elements involving d Orbitals
Hybridisation Of Elements involving d Oebitals
The elements present in the third period contain d orbitals in addition to s and p orbitals.The energy of the 3d orbitals are comparable to the energy of the 3s and 3p orbitals. The energy of 3d orbitals are also comparable to those of 4s and 4p orbitals. As a consequence the hybridisation involving either 3s, 3p and 3d or 3d, 4s and 4p is possible. However,since the difference in energies of 3p and 4s orbitals is significant, no hybridisation involving 3p, 3d and 4s orbitals is possible.
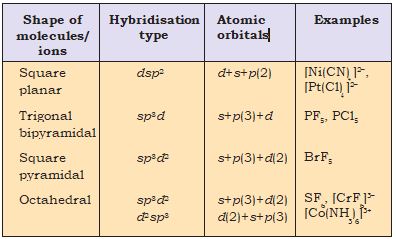
The important hybridisation schemes involving s, p and d orbitals are summarised below
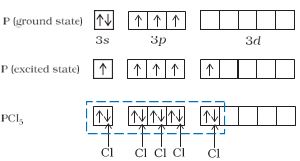
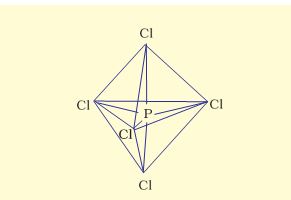
(1) Formation of PCl5(sp3d hybridisation):
The ground state and the excited state outer electronic configurations of phosphorus (Z=15) are represented below.
sp3d hybrid orbitals filled by electron pairs donated by five Cl atoms
Now the five orbitals (i.e., one s, three p and one d orbitals) are available for hybridisation to yield a set of five sp3d hybrid orbitals which are directed towards the five corners of a trigonal bipyramidal as depicted in the Fig. 4.17.
Fig. 4.17 Trigonal bipyramidal geometry of PCl5 molecule
It should be noted that all the bond angles in trigonal bipyramidal geometry are not equivalent.In PCl5 the five sp3d orbitals of phosphorus overlap with the singly occupied p orbitals of chlorine atoms to form five P–Cl sigma bonds. Three P–Cl bond lie in one plane and make an angle of 120° with each other; these bonds are termed as equatorial bonds. The remaining two P–Cl bonds–one lying above and the other lying below the equatorial plane, make an angle of 90° with the plane. These bonds are called axial bonds. As the axial bond pairs suffer more repulsive interaction from the equatorial bond pairs, therefore axial bonds have been found to be slightly longer and hence slightly weaker than the equatorial bonds; which makes PCl5 molecule more reactive.
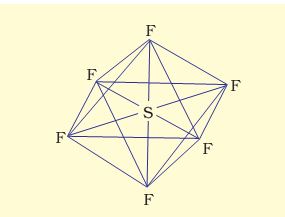
(2) Formation of SF6 (sp3d2 hybridisation): In SF6 the central sulphur atom has the ground state outer electronic configuration 3s23p4.In the exited state the available six orbitals i.e., one s, three p and two d are singly occupied by electrons.These orbitals hybridise to form six new sp3d2 hybrid orbitals, which are projected towards the six corners of a regular octahedron in SF.These six sp3d2 hybrid orbitals overlap with singly occupied orbitals of fluorine atoms to form six S–F sigma bonds. Thus SF6 molecule has a regular octahedral geometry as shown in Fig. 4.18.
sp3d2 hybridisation
Fig. 4.18 Octahedral geometry of SF6 molecule.