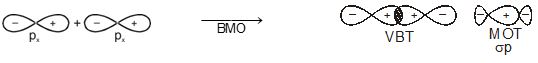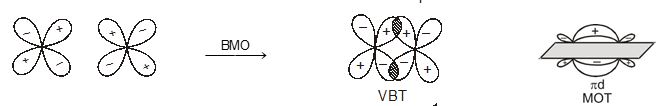-
Chemical bonding and Molecular structure
Molecular Orbital Theory
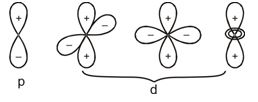
Shapes of Molecular Orbitals
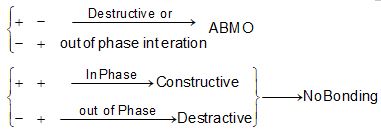
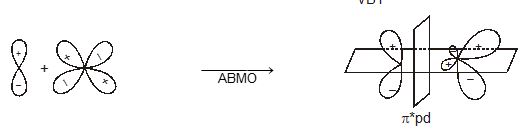
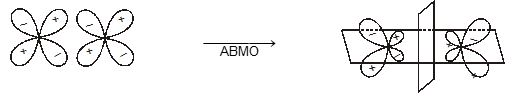
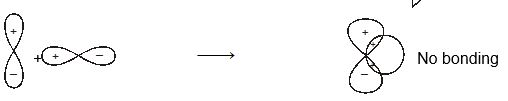
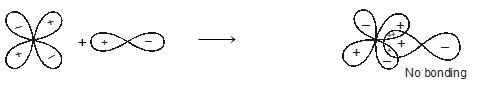
When atomic orbital overlaps in phase then B-MO are formed and if overlaps out of phase then ABMO are formed. And both process occurs simultaneously. Non bonding MO are formed
\(\left\{ \begin{matrix} + & + & \xrightarrow{{}}\,\,constructive \\ - & - & \xrightarrow{{}}constructive \\ \end{matrix} \right\}\xrightarrow{{}}BMO\)
For two lobe or two interaction.
\(\left\{ \begin{matrix} + & + \\ - & - \\ \end{matrix} \right\}\xrightarrow{in\,\,\,phase\,\,\,\operatorname{int}eraction}BMO\)\(\left\{ \begin{matrix} + & - & \xrightarrow{Destructive\,\,(or)\,\,out\,\,of\,\,phase} \\ {} & {} & \,\operatorname{int}eration \\ \end{matrix}ABMO \right.\)
Symmetry of Molecular Orbital :
It is specified by using term gerade and ungerade (No symmetry odd).
Those molecular orbitals which are symmetric about internuclear axis and axis bisecting internuclear axis perpendicularly when rotated by 1800 are called as gerade molecular orbital. Otherwise ungerade. Mathematically concept of symmetry is used to analyse a spectroscopic phenomena called as even pairity and odd pairity.
for M.O. to be gerade
f (x,y,z) = f(–x, –y, –z) gerade
f (x,y,z) = –f(–x,–y, –z) ungerade
Relative Energy of Molecular Orbital
for \({{O}_{2}}\,\,and\,\,{{F}_{2}}\) and their ions s1s < s*1s < s2s < s*2s < s2pz < [p 2px = p 2py] < [p* 2px = p*2py] < s*2pz
for \({{B}_{2}},\,\,{{C}_{2}},\,\,{{N}_{2}}\) etc. and their ions. 1s < s* 1s < s2s < s*2s < [p 2px = p 2py] < s2pz < [p*2px = p*2py] < s*2pzWhy \(H{{e}_{2}}\) molecule does not exist and why \({{O}_{2}}\) is paramagnetic? These questions cannot be explained by valence bond theory. In 1932 F. Hund and R.S. Mulliken put forward a theory known as Molecular Orbital Theory to explain above questions and many others. According to this theory, as the electrons of an atom are present in various atomic orbitals, electrons of a molecule are present in various molecular orbitals. Molecular orbitals are formed by the combination of atomic orbitals of comparable energy and proportional symmetry. While an electron in atomic orbital is influenced by one nucleus, in a molecular orbital, it is influenced by two or more nuclei depending upon the number of atoms in the molecule. Thus, an atomic orbital is monocentric while a molecular orbital is polycentric. The number of molecular orbitals formed is equal to the number of combining atomic orbitals. When two atomic orbitals combine, two molecular orbitals are formed. One is known as bonding molecular orbital (BMO) whereas other is anti-bonding molecular orbital (ABMO). BMO has lower energy and hence greater stability than the corresponding ABMO. First BMO are filled, then ABMO starts filling because BMO has lower energy than that of ABMO.
Comparision between BMO and ABMOBMOABMO 01. These are formed by addition of the orbitals of the same phase \(\Psi ={{\Psi }_{A}}+{{\Psi }_{B}}\)01. These are formed by the addition of the orbitals of different phase \(\Psi ={{\Psi }_{A}}-{{\Psi }_{B}}\).02. They have lower energy than atomic orbitals involved.02. They have higher energy than atomic orbitals.Molecular orbitals like the atomic orbitals are filled in accordance with the Aufbau Principle obeying the Pauli’s Principle and the Hund’s rule.
Order of energy of various molecular orbitals is as follows:
For O2 and higher molecules
\(\sigma \)1s,\(\sigma \)*1s, \(\sigma \)2s, \(\sigma \)*2s, \(\sigma \)2px, [\(\pi \)2py = \(\pi \)2pz], [\(\pi \)*2py =\(\pi \)*2pz], \(\sigma \)*2px
For N2 and lower molecules
\(\sigma \)1s, \(\sigma \)*1s, \(\sigma \)2s, \(\sigma \)*2s, [\(\pi \)2py = \(\pi \)2pz], \(\sigma \)2px, [\(\pi \)*2py = \(\pi \)*2pz], \(\sigma \)*2px
-
Explanation of Molecular Orbital Theory
Explanation of Molecular Orbital Theory
Molecular orbital (MO) theory was developed by F. Hund and R.S. Mulliken in 1932.The salient features of this theory are :
1. The electrons in a molecule are present in the various molecular orbitals as the electrons of atoms are present in the various atomic orbitals.
2. The atomic orbitals of comparable energies and proper symmetry combine to form molecular orbitals.
3. While an electron in an atomic orbital is influenced by one nucleus, in a molecular orbital it is influenced by two or more nuclei depending upon the number of atoms in the molecule. Thus,an atomic orbital is monocentric while a molecular orbital is polycentric.
4. The number of molecular orbital formed is equal to the number of combining atomic orbitals.When two atomic orbitals combine, two molecular orbitals are formed. One is known as bonding molecular orbital while the other is called antibonding molecular orbital.
5. The bonding molecular orbital has lower energy and hence greater stability than the corresponding antibonding molecular orbital.
6. Just as the electron probability distribution around a nucleus in an atom is given by an atomic orbital, the electron probability distribution around a group of nuclei in a molecule is given by a molecular orbital.
7. The molecular orbitals like atomic orbitals are filled in accordance with the aufbau principle obeying the Pauli’s exclusion principle and the Hund’s rule.
-
Linear Combination of Atomic Orbitals (LCAO) and Conditions for the Combination
Linear Combination of Atomic Orbitals (LCAO) and Conditions for the Combination
According to wave mechanics, the atomic orbitals can be expressed by wave functions (\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeqiYdKhaaa!37C4! \psi \)’s) which represent the amplitude of the electron waves. These are obtained from the solution of Schrödinger wave equation.However, since it cannot be solved for any system containing more than one electron, molecular orbitals which are one electron wave functions for molecules are difficult to obtain directly from the solution of Schrödinger wave equation. To overcome this problem, an approximate method known as linear combination of atomic orbitals (LCAO) has been adopted.
Let us apply this method to the homonuclear diatomic hydrogen molecule. Consider the hydrogen molecule consisting of two atoms A and B. Each hydrogen atom in the ground state has one electron in 1s orbital.The atomic orbitals of these atoms may be represented by the wave functions \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeqiYdKhaaa!37C4! \psi \)A and \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeqiYdKhaaa!37C4! \psi \)B.
Mathematically, the formation of molecular orbitals may be described by the linear combination of atomic orbitals that can take place by addition and by subtraction of wave functions of individual atomic orbitals as shown below :
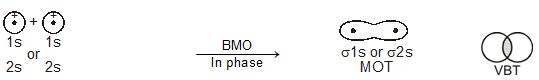
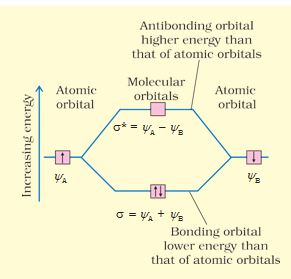
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbiqaaG3acqaHip % qEdaWgaaWcbaGaamytaiaad+eaaeqaaOGaeyypa0JaeqiYdK3aaSba % aSqaaiaadgeaaeqaaOGaeyySaeRaeqiYdK3aaSbaaSqaaiaadkeaae % qaaaaa!429D! {\psi _{MO}} = {\psi _A} \pm {\psi _B}\)
Therefore, the two molecular orbitals \(\sigma\) and \(\sigma\)* are formed as :
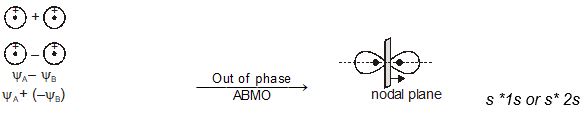
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGceiqabeaa4nqaai % abeo8aZjabg2da9iabeI8a5naaBaaaleaacaWGbbaabeaakiabgUca % RiabeI8a5naaBaaaleaacaWGcbaabeaaaOqaaiabeo8aZbbaaaaaaa % aapeGaaiOka8aacqGH9aqpcqaHipqEdaWgaaWcbaGaamyqaaqabaGc % cqGHsislcqaHipqEdaWgaaWcbaGaamOqaaqabaaaaaa!49D9! \begin{array}{l} \sigma = {\psi _A} + {\psi _B}\\ \sigma * = {\psi _A} - {\psi _B} \end{array}\)
The molecular orbital \(\sigma\) formed by the addition of atomic orbitals is called the bonding molecular orbital while the molecular orbital \(\sigma\)* formed by the subtraction of atomic orbital is called antibonding molecular orbital as depicted in Fig. 4.19.
Fig.4.19 Formation of bonding (\(\sigma\)) and antibonding (\(\sigma\)*) molecular orbitals by the linear combination of atomic orbitals \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeqiYdKhaaa!37C4! \psi \) and \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeqiYdKhaaa!37C4! \psi \)B centered on two atoms A and B respectively.
Qualitatively, the formation of molecular orbitals can be understood in terms of the constructive or destructive interference of the electron waves of the combining atoms. In the formation of bonding molecular orbital, the two electron waves of the bonding atoms reinforce each other due to constructive interference while in the formation of antibonding molecular orbital, the electron waves cancel each other due to destructive interference. As a result, the electron density in a bonding molecular orbital is located between the nuclei of the bonded atoms because of which the repulsion between the nuclei is very less while in case of an antibonding molecular orbital, most of the electron density is located away from the space between the nuclei. Infact, there is a nodal plane (on which the electron density is zero) between the nuclei and hence the repulsion between the nuclei is high. Electrons placed in a bonding molecular orbital tend to hold the nuclei together and stabilise a molecule. Therefore, a bonding molecular orbital always possesses lower energy than either of the atomic orbitals that have combined to form it. In contrast, the electrons placed in the antibonding molecular orbital destabilise the molecule. This is because the mutual repulsion of the electrons in this orbital is more than the attraction between the electrons and the nuclei, which causes a net increase in energy.
It may be noted that the energy of the antibonding orbital is raised above the energy of the parent atomic orbitals that have combined and the energy of the bonding orbital has been lowered than the parent orbitals. The total energy of two molecular orbitals, however, remains the same as that of two original atomic orbitals.
Conditions for the Combination of Atomic Orbitals
The linear combination of atomic orbitals to form molecular orbitals takes place only if the following conditions are satisfied:
1. The combining atomic orbitals must have the same or nearly the same energy. This means that 1s orbital can combine with another 1s orbital but not with 2s orbital because the energy of 2s orbital is appreciably higher than that of 1s orbital. This is not true if the atoms are very different.
2. The combining atomic orbitals must have the same symmetry about the molecular axis.By convention z-axis is taken as the molecular axis. It is important to note that atomic orbitals having same or nearly the same energy will not combine if they do not have the same symmetry.For example, 2pz orbital of one atom can combine with 2pz orbital of the other atom but not with the 2px or 2py orbitals
because of their different symmetries.
3. The combining atomic orbitals must overlap to the maximum extent. Greater the extent of overlap, the greater will be the electron-density between the nuclei of a molecular orbital.
-
Types of Molecular Orbitals
Types of Molecular Orbitals
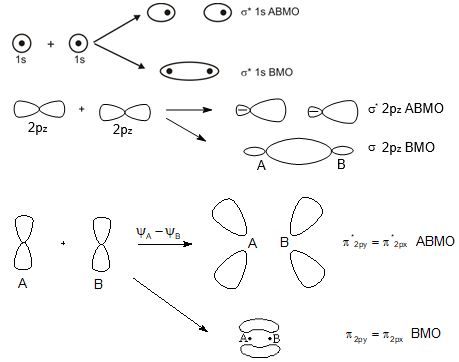
Molecular orbitals of diatomic molecules are designated as \(\sigma\) (sigma), \(\pi\) (pi), \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbiqaaG3aqaaaaa % aaaaWdbiabes7aKbaa!3839! \delta \) (delta), etc.
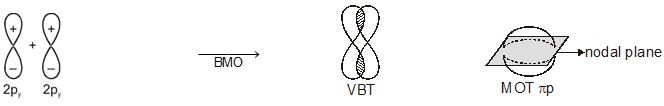
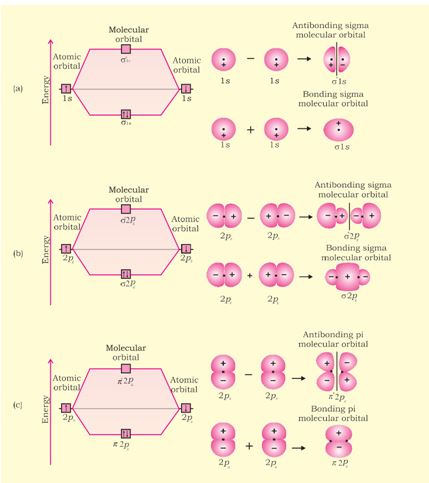
In this nomenclature, the sigma (\(\sigma\)) molecular orbitals are symmetrical around the bond-axis while pi (\(\pi\)) molecular orbitals are not symmetrical. For example, the linear combination of 1s orbitals centered on two nuclei produces two molecular orbitals which are symmetrical around the bond-axis. Such molecular orbitals are of the \(\sigma\) type and are designated as \(\sigma\)1s and \(\sigma\)*1s [Fig. 4.20(a),page 124]. If internuclear axis is taken to be in the z-direction, it can be seen that a linear combination of 2pz- orbitals of two atoms also produces two sigma molecular orbitals designated as \(\sigma\)2p and \(\sigma\)*2p . [Fig. 4.20(b)].
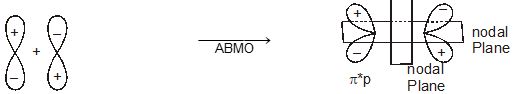
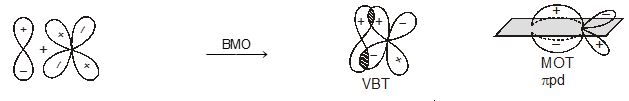
Molecular orbitals obtained from 2px and 2py orbitals are not symmetrical around the bond axis because of the presence of positive lobes above and negative lobes below the molecular plane. Such molecular orbitals, are labelled as \(\pi\) and \(\pi\) * [Fig. 4.20(c)]. A \(\pi\) bonding MO has larger electron density above and below the inter -nuclear axis.The \(\pi\)* antibonding MO has a node between the nuclei.
-
Energy Level Diagram for Molecular Orbitals
Energy Level Diagram for Molecular Orbitals
We have seen that 1s atomic orbitals on two atoms form two molecular orbitals designated as \(\sigma\)1s and \(\sigma\)*1s. In the same manner, the 2s and 2p atomic orbitals (eight atomic orbitals on two atoms) give rise to the following eight molecular orbitals:
Fig. 4.20 Contours and energies of bonding and antibonding molecular orbitals formed through combinations of (a) 1s atomic orbitals; (b) 2pz atomic orbitals and (c) 2px atomic orbitals.
Antibonding MOs \(\sigma\)*2s \(\sigma\)*2pz π *2px π*2py
Bonding MOs \(\sigma\)2s \(\sigma\)2pz π2px π2py
The energy levels of these molecular orbitals have been determined experimentally from spectroscopic data for homonuclear diatomic molecules of second row elements of the periodic table. The increasing order of energies of various molecular orbitals for O2 and F2 is given below:
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbiqaaG3aqaaaaa % aaaaWdbiabeo8aZjaaigdacaWGZbGaeyipaWJaeq4Wdm3aaWbaaSqa % beaacaGGQaaaaOGaaGymaiaadohacqGH8aapcqaHdpWCcaaIYaGaam % 4CaiabgYda8iabeo8aZnaaCaaaleqabaGaaiOkaaaakiaaikdacaWG % ZbGaeyipaWJaeq4WdmNaaGOmaiaadchadaWgaaWcbaGaamOEaaqaba % GccqGH8aapdaqadaqaaiabec8aWjaaikdacaWGWbWaaSbaaSqaaiaa % dIhaaeqaaOGaeyypa0JaeqiWdaNaaGOmaiaadchadaWgaaWcbaGaam % yEaaqabaaakiaawIcacaGLPaaacqGH8aapdaqadaqaaiabec8aWnaa % CaaaleqabaGaaiOkaaaakiaaikdacaWGWbWaaSbaaSqaaiaadIhaae % qaaOGaeyypa0JaeqiWdaNaaiOkaiaaikdacaWGWbWaaSbaaSqaaiaa % dMhaaeqaaaGccaGLOaGaayzkaaGaeyipaWJaeq4Wdm3aaWbaaSqabe % aacaGGQaaaaOGaaGOmaiaadchadaWgaaWcbaGaamOEaaqabaaaaa!70B8! \sigma 1s < {\sigma ^*}1s < \sigma 2s < {\sigma ^*}2s < \sigma 2{p_z} < \left( {\pi 2{p_x} = \pi 2{p_y}} \right) < \left( {{\pi ^*}2{p_x} = \pi *2{p_y}} \right) < {\sigma ^*}2{p_z}\)
However, this sequence of energy levels of molecular orbitals is not correct for the remaining molecules Li2, Be2, B2, C2, N2. For instance, it has been observed experimentally that for molecules such as B2, C2, N2, etc. the
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbiqaaG3aqaaaaa % aaaaWdbiabeo8aZjaaigdacaWGZbGaeyipaWJaeq4Wdm3aaWbaaSqa % beaacaGGQaaaaOGaaGymaiaadohacqGH8aapcqaHdpWCcaaIYaGaam % 4CaiabgYda8iabeo8aZnaaCaaaleqabaGaaiOkaaaakiaaikdacaWG % ZbGaeyipaWZaaeWaaeaacqaHapaCcaaIYaGaamiCamaaBaaaleaaca % WG4baabeaakiabg2da9iabec8aWjaaikdacaWGWbWaaSbaaSqaaiaa % dMhaaeqaaaGccaGLOaGaayzkaaGaeyipaWJaeq4Wdm3aaWbaaSqabe % aacaGGQaaaaOGaaGOmaiaadchadaWgaaWcbaGaamOEaaqabaGccqGH % 8aapdaqadaqaaiabec8aWnaaCaaaleqabaGaaiOkaaaakiaaikdaca % WGWbWaaSbaaSqaaiaadIhaaeqaaOGaeyypa0JaeqiWda3aaWbaaSqa % beaacaGGQaaaaOGaaGOmaiaadchadaWgaaWcbaGaamyEaaqabaaaki % aawIcacaGLPaaacqGH8aapcqaHdpWCdaahaaWcbeqaaiaacQcaaaGc % caaIYaGaamiCamaaBaaaleaacaWG6baabeaaaaa!71D4! \sigma 1s < {\sigma ^*}1s < \sigma 2s < {\sigma ^*}2s < \left( {\pi 2{p_x} = \pi 2{p_y}} \right) < {\sigma ^*}2{p_z} < \left( {{\pi ^*}2{p_x} = {\pi ^*}2{p_y}} \right) < {\sigma ^*}2{p_z}\)
The important characteristic feature of this order is that the energy of \(\sigma\)2p molecular orbital is higher than that of π 2px and π 2py molecular orbitals.
-
Electronic Configuration and Molecular Behaviour
Electronic Configuration and Molecular Behaviour
The distribution of electrons among various molecular orbitals is called the electronic configuration of the molecule.From the electronic configuration of the molecule, it is possible to get important information about the molecule as discussed below.
Stability of Molecules: If Nb is the number of electrons occupying bonding orbitals and Na the number occupying the antibonding orbitals, then
1. the molecule is stable if Nb is greater than Na, and
2. the molecule is unstable if Nb is less than Na.
In (i) more bonding orbitals are occupied and so the bonding influence is stronger and a stable molecule results. In (ii) the antibonding influence is stronger and therefore the molecule is unstable.