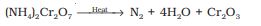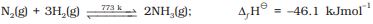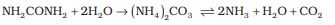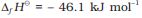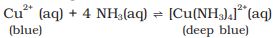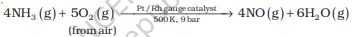-
All topics
In Class XI, you have learnt that the p-block elements are placed in groups 13 to 18 of the periodic table. Their valence shell electronic configuration is ns2np1–6 (except He which has 1s2 configuration). The properties of p-block elements like that of others are greatly influenced by atomic sizes, ionisation enthalpy, electron gain enthalpy and electronegativity. The absence of d- orbitals in second period and presence of d or d and f orbitals in heavier elements (starting from third period onwards) have significant effects on the properties of elements. In addition, the presence of all the three types of elements; metals, metalloids and non-metals bring diversification in chemistry of these elements.
Having learnt the chemistry of elements of Groups 13 and 14 of the p-block of periodic table in Class XI, you will learn the chemistry of the elements of subsequent groups in this Unit.
Group 15 ELEMENTS
Group 15 includes nitrogen, phosphorus, arsenic, antimony, bismuth and moscovium. As we go down the group, there is a shift from non- metallic to metallic through metalloidic character. Nitrogen and phosphorus are non-metals, arsenic and antimony metalloids, bismuth and moscovium are typical metals.
Occurrence
Molecular nitrogen comprises 78% by volume of the atmosphere. In the earth’s crust, it occurs as sodium nitrate, NaNO3 (called Chile saltpetre) and potassium nitrate (Indian saltpetre). It is found in the form of proteins in plants and animals. Phosphorus occurs in minerals of the apatite family, Ca9(PO4)6. CaX2 (X = F, Cl or OH) (e.g., fluorapatite Ca9 (PO4)6. CaF2) which are the main components of phosphate rocks. Phosphorus is an essential constituent of animal and plant matter. It is present in bones as well as in living cells. Phosphoproteins are present in milk and eggs. Arsenic, antimony and bismuth are found mainly as sulphide minerals. Moscovium is a synthetic radioactive element. Its symbol is Mc, atomic number 115, atomic mass 289 and electronic configuration [Rn] 5f 146d107s27p3. Due to very short half life and availability in very little amount, its chemistry is yet to be established.
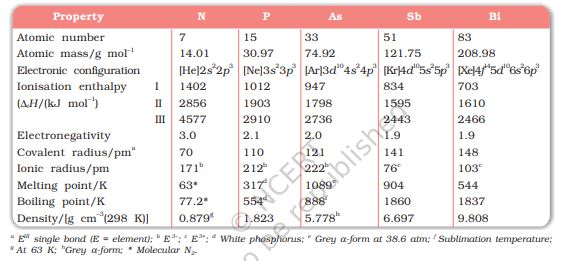
Here, except for moscovium, important atomic and physical properties of other elements of this group along with their electronic configurations are given in Table 7.1.
Table 7.1: Atomic and Physical Properties of Group 15 Elements
Trends of some of the atomic, physical and chemical properties of the group are discussed below.
Electronic ConfiguratioN
The valence shell electronic configuration of these elements is ns2np3. The s orbital in these elements is completely filled and p orbitals are half-filled, making their electronic configuration extra stable.
Atomic AND Ionic Radii
Covalent and ionic (in a particular state) radii increase in size down the group. There is a considerable increase in covalent radius from N to P. However, from As to Bi only a small increase in covalent radius is observed. This is due to the presence of completely filled d and/or f orbitals in heavier members.
Ionisation Enthalpy
Ionisation enthalpy decreases down the group due to gradual increase in atomic size. Because of the extra stable half-filled p orbitals electronic configuration and smaller size, the ionisation enthalpy of the group 15 elements is much greater than that of group 14 elements in the corresponding periods. The order of successive ionisation enthalpies, as expected is \(\Delta\)iH1 < \(\Delta\)iH2 < \(\Delta\)iH3 (Table 7.1).
Electronegativity
The electronegativity value, in general, decreases down the group with increasing atomic size. However, amongst the heavier elements, the difference is not that much pronounced.
PHYSICAL Properties
All the elements of this group are polyatomic. Dinitrogen is a diatomic gas while all others are solids. Metallic character increases down the group. Nitrogen and phosphorus are non-metals, arsenic and antimony metalloids and bismuth is a metal. This is due to decrease in ionisation enthalpy and increase in atomic size. The boiling points, in general, increase from top to bottom in the group but the melting point increases upto arsenic and then decreases upto bismuth. Except nitrogen, all the elements show allotropy.
Chemical Properties
Oxidation states and trends in chemical reactivity
The common oxidation states of these elements are –3, +3 and +5. The tendency to exhibit –3 oxidation state decreases down the group due to increase in size and metallic character. In fact last member of the group, bismuth hardly forms any compound in –3 oxidation state. The stability of +5 oxidation state decreases down the group. The only well characterised Bi (V) compound is BiF5. The stability of +5 oxidation state decreases and that of +3 state increases (due to inert pair effect) down the group. Besides +5 oxidation state, nitrogen exhibits + 1, + 2, + 4 oxidation states also when it reacts with oxygen. However, it does not form compounds in +5 oxidation state with halogens as nitrogen does not have d-orbitals to accommodate electrons from other elements to form bonds. Phosphorus also shows +1 and +4 oxidation states in some oxoacids. In the case of nitrogen, all oxidation states from +1 to +4 tend to disproportionate in acid solution. For example,
3HNO2 \(\to\) HNO3 + H2O + 2NO
Similarly, in case of phosphorus nearly all intermediate oxidation states disproportionate into +5 and –3 both in alkali and acid. However
+3 oxidation state in case of arsenic, antimony and bismuth becomes increasingly stable with respect to disproportionation.
Nitrogen is restricted to a maximum covalency of 4 since only four (one s and three p) orbitals are available for bonding. The heavier elements have vacant d orbitals in the outermost shell which can be used for bonding (covalency) and hence, expand their covalence as in PF\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaiAdaaeaacqGHsislaaaaaa!37D0! _6^ - \).
Anomalous properties of nitrogen
Nitrogen differs from the rest of the members of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d orbitals. Nitrogen has unique ability to form p\(\pi\) -p\(\pi\) multiple bonds with itself and with other elements having small size and high electronegativity (e.g., C, O). Heavier elements of this group do not form p\(\pi\) -p\(\pi\) bonds as their atomic orbitals are so large and diffuse that they cannot have effective overlapping. Thus, nitrogen exists as a diatomic molecule with a triple bond (one s and two p) between the two atoms. Consequently, its bond enthalpy (941.4 kJ mol–1) is very high. On the contrary, phosphorus, arsenic and antimony form single bonds as P–P, As–As and Sb–Sb while bismuth forms metallic bonds in elemental state. However, the single N–N bond is weaker than the single P–P bond because of high interelectronic repulsion of the non-bonding electrons, owing to the small bond length. As a result the catenation tendency is weaker in nitrogen. Another factor which affects the chemistry of nitrogen is the absence of d orbitals in its valence shell. Besides restricting its covalency to four, nitrogen cannot form d\(\pi\) –p\(\pi\) bond as the heavier elements can e.g., R3P = O or R3P = CH2 (R = alkyl group). Phosphorus and arsenic can form d\(\pi\) –d\(\pi\) bond also with transition metals when their compounds like P(C2H5)3 and As(C6H5)3 act as ligands.
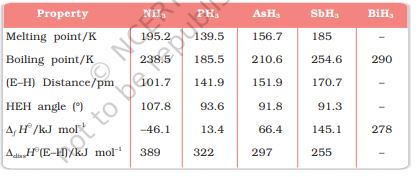
(1) Reactivity towards hydrogen: All the elements of Group 15 form hydrides of the type EH3 where E = N, P, As, Sb or Bi. Some of the properties of these hydrides are shown in Table 7.2. The hydrides show regular gradation in their properties.
Table 7.2: Properties of Hydrides of Group 15 Elements
The stability of hydrides decreases from NH3 to BiH3 which can be observed from their bond dissociation enthalpy. Consequently, the reducing character of the hydrides increases. Ammonia is only a mild reducing agent while BiH3 is the strongest reducing agent amongst all the hydrides. Basicity also decreases in the order NH3 > PH3 > AsH3 > SbH3 > BiH3. Due to high electronegativity and small size of nitrogen, NH3 exhibits hydrogen bonding in solid as well as liquid state. Because of this, it has higher melting and boiling points than that of PH3.
(2) Reactivity towards oxygen: All these elements form two types of oxides: E2O3 and E2O5. The oxide in the higher oxidation state of the element is more acidic than that of lower oxidation state. Their acidic character decreases down the group. The oxides of the type E2O3 of nitrogen and phosphorus are purely acidic, that of arsenic and antimony amphoteric and those of bismuth predominantly basic.
(3) Reactivity towards halogens: These elements react to form two series of halides: EX3 and EX5. Nitrogen does not form pentahalide due to non-availability of the d orbitals in its valence shell. Pentahalides are more covalent than trihalides. This is due to the fact that in pentahalides +5 oxidation state exists while in the case of trihalides +3 oxidation state exists. Since elements in +5 oxidation state will have more polarising power than in +3 oxidation state, the covalent character of bonds is more in pentahalides. All the trihalides of these elements except those of nitrogen are stable. In case of nitrogen, only NF3 is known to be stable. Trihalides except BiF3 are predominantly covalent in nature.
(4) Reactivity towards metals: All these elements react with metals to form their binary compounds exhibiting –3 oxidation state, such as, Ca3N2 (calcium nitride) Ca3P2 (calcium phosphide), Na3As (sodium arsenide), Zn3Sb2 (zinc antimonide) and Mg3Bi2 (magnesium bismuthide).
EXAMPLE 1
Though nitrogen exhibits +5 oxidation state, it does not form pentahalide. Give reason.
SOLUTION
Nitrogen with n = 2, has s and p orbitals only. It does not have d orbitals to expand its covalence beyond four. That is why it does not form pentahalide.
EXAMPLE 2
PH3 has lower boiling point than NH3. Why?
SOLUTION
Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3.
INTEXT QUESTION
Why are pentahalides of P, As, Sb and Bi more covalent than their trihalides?
Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements ?
Dinitrogen
Preparation
Dinitrogen is produced commercially by the liquefaction and fractional distillation of air. Liquid dinitrogen (b.p. 77.2 K) distils out first leaving behind liquid oxygen (b.p. 90 K).
In the laboratory, dinitrogen is prepared by treating an aqueous solution of ammonium chloride with sodium nitrite.
NH4CI(aq) + NaNO2(aq) \(\to\) N2(g) + 2H2O(l) + NaCl (aq)
Small amounts of NO and HNO3 are also formed in this reaction; these impurities can be removed by passing the gas through aqueous sulphuric acid containing potassium dichromate. It can also be obtained by the thermal decomposition of ammonium dichromate.
Very pure nitrogen can be obtained by the thermal decomposition of sodium or barium azide.
Ba(N3)2 \(\to\) Ba + 3N2
Properties
Dinitrogen is a colourless, odourless, tasteless and non-toxic gas. Nitrogen atom has two stable isotopes: 14N and 15N. It has a very low solubility in water (23.2 cm3 per litre of water at 273 K and 1 bar pressure) and low freezing and boiling points (Table 7.1).
Dinitrogen is rather inert at room temperature because of the high bond enthalpy of N \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeyyyIOlaaa!37BF! \equiv \) N bond. Reactivity, however, increases rapidly with rise in temperature. At higher temperatures, it directly combines with some metals to form predominantly ionic nitrides and with non-metals, covalent nitrides. A few typical reactions are:
It combines with hydrogen at about 773 K in the presence of a catalyst (Haber’s Process) to form ammonia:
Dinitrogen combines with dioxygen only at very high temperature (at about 2000 K) to form nitric oxide, NO.
Uses: The main use of dinitrogen is in the manufacture of ammonia and other industrial chemicals containing nitrogen, (e.g., calcium cyanamide). It also finds use where an inert atmosphere is required (e.g., in iron and steel industry, inert diluent for reactive chemicals). Liquid dinitrogen is used as a refrigerant to preserve biological materials, food items and in cryosurgery.
EXAMPLE 3
Write the reaction of thermal decomposition of sodium azide.
SOLUTION Thermal decomposition of sodium azide gives dinitrogen gas.
2NaN3 \(\to\) 2Na + 3N2
INTEXT QUESTION
Why is N2 less reactive at room temperature?
Ammonia
Preparation
Ammonia is present in small quantities in air and soil where it is formed by the decay of nitrogenous organic matter e.g., urea.
On a small scale ammonia is obtained from ammonium salts which decompose when treated with caustic soda or calcium hydroxide.
2NH4Cl + Ca(OH)2 \(\to\) 2NH3 + 2H2O + CaCl2
(NH4)2 SO4 + 2NaOH \(\to\) 2NH3 + 2H2O + Na2SO4
On a large scale, ammonia is manufactured by Haber’s process.
N2 (g) + 3H2 (g) Ö 2NH3 (g);
In accordance with Le Chatelier’s principle, high pressure would favour the formation of ammonia. The optimum conditions for the production of ammonia are a pressure of 200 × 105 Pa (about 200 atm), a temperature of ~ 700 K and the use of a catalyst such as iron oxide with small amounts of K2O and Al2O3 to increase the rate of attainment of equilibrium. The flow chart for the production of ammonia is shown in Fig. 7.1. Earlier, iron was used as a catalyst with molybdenum as a promoter.
Fig. 7.1. Flow chart for the manufacture of ammonia
Properties
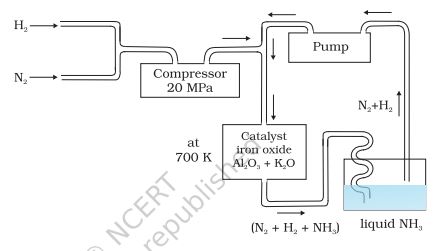
Ammonia is a colourless gas with a pungent odour. Its freezing and boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated through hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis of its molecular mass. The ammonia molecule is trigonal pyramidal with the nitrogen atom at the apex. It has three bond pairs and one lone pair of electrons as shown in the structure.
Ammonia gas is highly soluble in water. Its aqueous solution is weakly basic due to the formation of OH– ions.
It forms ammonium salts with acids, e.g., NH4Cl, (NH4)2 SO4, etc. As a weak base, it precipitates the hydroxides (hydrated oxides in case of some metals) of many metals from their salt solutions. For example,
ZnSO4 (aq) + 2NH4OH (aq ) \(\to\) Zn (OH)2 (s) + (NH4) SO4 (aq)
( white ppt )
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGceaqabeaacaWGgb % GaamyzaiaadoeacaWGSbWaaSbaaSqaaiaaiodaaeqaaOWaaeWaaeaa % caWGHbGaamyCaaGaayjkaiaawMcaaiabgUcaRiaad6eacaWGibWaaS % baaSqaaiaaisdaaeqaaOGaam4taiaadIeadaqadaqaaiaadggacaWG % XbaacaGLOaGaayzkaaGaeyOKH4QaamOraiaadwgadaWgaaWcbaGaaG % OmaaqabaGccaWGpbWaaSbaaSqaaiaaiodaaeqaaOGaaiOlaiaadIha % caWGibWaaSbaaSqaaiaaikdaaeqaaOGaam4tamaabmaabaGaam4Caa % GaayjkaiaawMcaaiabgUcaRiaad6eacaWGibWaaSbaaSqaaiaaisda % aeqaaOGaam4qaiaadYgadaqadaqaaiaadggacaWGXbaacaGLOaGaay % zkaaaabaGaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPa % VlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8 % UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7 % caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVl % aaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8Ua % aGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7ca % aMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaa % ykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaG % PaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7daqa % daqaaiaadkgacaWGYbGaam4BaiaadEhacaWGUbGaaGPaVlaaykW7ca % WGWbGaamiCaiaadshaaiaawIcacaGLPaaaaaaa!D5C1! \begin{array}{l} FeC{l_3}\left( {aq} \right) + N{H_4}OH\left( {aq} \right) \to F{e_2}{O_3}.x{H_2}O\left( s \right) + N{H_4}Cl\left( {aq} \right)\\ \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\left( {brown\,\,ppt} \right) \end{array}\)
The presence of a lone pair of electrons on the nitrogen atom of the ammonia molecule makes it a Lewis base. It donates the electron pair and forms linkage with metal ions and the formation of such complex compounds finds applications in detection of metal ions such as Cu2+, Ag+:
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamyqaiaadE % gadaahaaWcbeqaaiabgUcaRaaakmaabmaabaGaamyyaiaadghaaiaa % wIcacaGLPaaacqGHRaWkcaWGdbGaamiBamaaCaaaleqabaGaeyOeI0 % caaOWaaeWaaeaacaWGHbGaamyCaaGaayjkaiaawMcaaiabgkziUkaa % dgeacaWGNbGaam4qaiaadYgadaqadaqaaiaadohaaiaawIcacaGLPa % aaaaa!4B23! A{g^ + }\left( {aq} \right) + C{l^ - }\left( {aq} \right) \to AgCl\left( s \right)\)
(colourless) (white ppt)
AgCl (s) + 2NH3 (aq) \(\to\) [Ag (NH3)2 ] Cl (aq)
(white ppt) (colourless)
Uses: Ammonia is used to produce various nitrogenous fertilisers (ammonium nitrate, urea, ammonium phosphate and ammonium sulphate) and in the manufacture of some inorganic nitrogen compounds, the most important one being nitric acid. Liquid ammonia is also used as a refrigerant.
EXAMPLE 4
Why does NH3 act as a Lewis base ?
SOLUTION
Nitrogen atom in NH3 has one lone pair of electrons which is available for donation. Therefore, it acts as a Lewis base.
INTEXT QUESTION
Mention the conditions required to maximise the yield of ammonia.
How does ammonia react with a solution of Cu2+?
Oxides of Nitrogen
Nitrogen forms a number of oxides in different oxidation states. The names, formulas, preparation and physical appearance of these oxides are given in Table 7.3.
Table 7.3: Oxides of Nitrogen
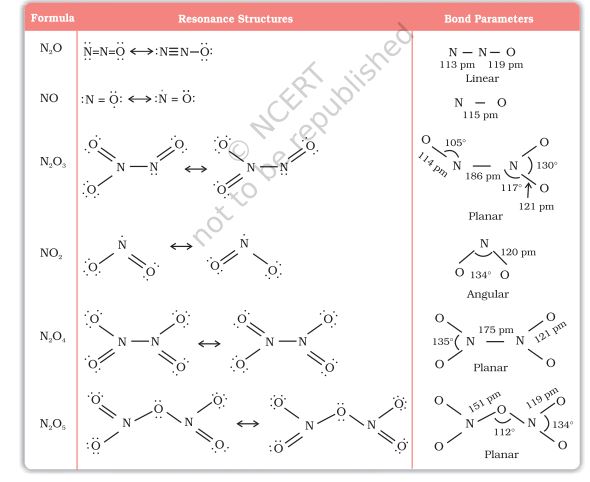
Lewis dot main resonance structures and bond parameters of oxides are given in Table 7.4.
Table 7.4: Structures of Oxides of Nitrogen
EXAMPLE 5
Why does NO2 dimerise ?
SOLUTION
NO2 contains odd number of valence electrons. It behaves as a typical odd molecule. On dimerisation, it is converted to stable N2O4 molecule with even number of electrons.
INTEXT QUESTION
What is the covalence of nitrogen in N2O5 ?
Nitric Acid
Nitrogen forms oxoacids such as H2N2O2 (hyponitrous acid), HNO2
(nitrous acid) and HNO3 (nitric acid). Amongst them HNO3 is the most important.
Preparation
In the laboratory, nitric acid is prepared by heating KNO3 or NaNO3 and concentrated H2SO4 in a glass retort.
NaNO3 + H2SO4 \(\to\) NaHSO4 + HNO3
On a large scale it is prepared mainly by Ostwald’s process.
This method is based upon catalytic oxidation of NH3 by atmospheric oxygen.
Nitric oxide thus formed combines with oxygen giving NO2.
2NO (g ) + O2 (g) 2NO2 (g)
Nitrogen dioxide so formed, dissolves in water to give HNO3.
3NO2 (g) \(\to\) H2O (l) \(\to\) 2HNO3 (aq) + NO (g)
NO thus formed is recycled and the aqueous HNO3 can be concentrated by distillation upto ~ 68% by mass. Further concentration to 98% can be achieved by dehydration with concentrated H2SO4.
Properties
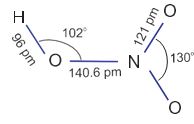
It is a colourless liquid (f.p. 231.4 K and b.p. 355.6 K). Laboratory grade nitric acid contains ~ 68% of the HNO3 by mass and has a specific gravity of 1.504.
In the gaseous state, HNO3 exists as a planar molecule with the structure as shown.
In aqueous solution, nitric acid behaves as a strong acid giving hydronium and nitrate ions.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamisaiaad6 % eacaWGpbWaaSbaaSqaaiaaiodaaeqaaOGaaiikaiaadggacaWGXbGa % aiykaiabgUcaRiaadIeadaWgaaWcbaGaaGOmaaqabaGccaWGpbGaai % ikaiaaigdacaGGPaGaeyOKH4QaamisamaaBaaaleaacaaIZaaabeaa % kiaad+eadaahaaWcbeqaaiabgUcaRaaakiaacIcacaWGHbGaamyCai % aacMcacqGHRaWkcaWGobGaam4tamaaDaaaleaacaaIZaaabaGaeyOe % I0caaOWaaeWaaeaacaWGHbGaamyCaaGaayjkaiaawMcaaaaa!52B9! HN{O_3}(aq) + {H_2}O(1) \to {H_3}{O^ + }(aq) + NO_3^ - \left( {aq} \right)\)
Concentrated nitric acid is a strong oxidising agent and attacks most metals except noble metals such as gold and platinum. The products of oxidation depend upon the concentration of the acid, temperature and the nature of the material undergoing oxidation.
3Cu + 8 HNO3(dilute) \(\to\) 3Cu(NO3)2 + 2NO + 4H2O
Cu + 4HNO3(conc.)\(\to\)Cu(NO3)2 + 2NO2 + 2H2O
Zinc reacts with dilute nitric acid to give N2O and with concentrated acid to give NO2.
4Zn + 10HNO3(dilute) \(\to\)4 Zn (NO3)2 + 5H2O + N2O
Zn + 4HNO3(conc.) \(\to\)Zn (NO3)2 + 2H2O + 2NO2
Some metals (e.g., Cr, Al) do not dissolve in concentrated nitric acid because of the formation of a passive film of oxide on the surface. Concentrated nitric acid also oxidises non–metals and their compounds. Iodine is oxidised to iodic acid, carbon to carbon dioxide,
sulphur to H2SO4, and phosphorus to phosphoric acid.
I2 + 10HNO3 \(\to\)2HIO3 + 10NO2 + 4H2O
C + 4HNO3 \(\to\)CO2 + 2H2O + 4NO2
S8 + 48HNO3 \(\to\) 8H2SO4 + 48NO2 + 16H2O
P4 + 20HNO3 \(\to\)4H3PO4 + 20NO2 + 4H2O
Brown Ring Test: The familiar brown ring test for nitrates depends on the ability of Fe2+ to reduce nitrates to nitric oxide, which reacts with Fe2+ to form a brown coloured complex. The test is usually carried out by adding dilute ferrous sulphate solution to an aqueous solution containing nitrate ion, and then carefully adding concentrated sulphuric acid along the sides of the test tube. A brown ring at the interface between the solution and sulphuric acid layers indicates the presence of nitrate ion in solution.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGceaqabeaacaWGob % Gaam4tamaaDaaaleaacaaIZaaabaGaeyOeI0caaOGaey4kaSIaaG4m % aiaadAeacaWGLbWaaWbaaSqabeaacaaIYaGaey4kaScaaOGaey4kaS % IaaGinaiaadIeadaahaaWcbeqaaiabgUcaRaaakiabgkziUkaad6ea % caWGpbGaey4kaSIaaG4maiaadAeacaWGLbWaaWbaaSqabeaacaaIZa % Gaey4kaScaaOGaaGOmaiaadIeadaWgaaWcbaGaaGOmaaqabaGccaWG % pbaabaWaamWaaeaacaWGgbGaamyzamaabmaabaGaamisamaaBaaale % aacaaIYaaabeaakiaad+eaaiaawIcacaGLPaaadaWgaaWcbaGaaGOn % aaqabaaakiaawUfacaGLDbaadaahaaWcbeqaaiaaikdacqGHRaWkaa % GccqGHRaWkcaWGobGaam4taiabgkziUoaadmaabaGaamOraiaadwga % daqadaqaaiaadIeadaWgaaWcbaGaaGOmaaqabaGccaWGpbaacaGLOa % GaayzkaaWaaSbaaSqaaiaaiwdaaeqaaOWaaeWaaeaacaWGobGaam4t % aaGaayjkaiaawMcaaaGaay5waiaaw2faamaaCaaaleqabaGaaGOmai % abgUcaRaaakiabgUcaRiaadIeadaWgaaWcbaGaaGOmaaqabaGccaWG % pbaabaGaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVl % aaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8Ua % aGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7ca % aMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaa % ykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaG % PaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaM % c8UaaGPaVlaaykW7caaMc8UaaGPaVlaaykW7caaMc8UaaGPaVlaayk % W7caaMc8+aaeWaaeaacaWGIbGaamOCaiaad+gacaWG3bGaamOBaaGa % ayjkaiaawMcaaaaaaa!CCB5! \begin{array}{l} NO_3^ - + 3F{e^{2 + }} + 4{H^ + } \to NO + 3F{e^{3 + }}2{H_2}O\\ {\left[ {Fe{{\left( {{H_2}O} \right)}_6}} \right]^{2 + }} + NO \to {\left[ {Fe{{\left( {{H_2}O} \right)}_5}\left( {NO} \right)} \right]^{2 + }} + {H_2}O\\ \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\left( {brown} \right) \end{array}\)
USES:
The major use of nitric acid is in the manufacture of ammonium nitrate for fertilisers and other nitrates for use in explosives and pyrotechnics. It is also used for the preparation of nitroglycerin, trinitrotoluene and other organic nitro compounds. Other major uses are in the pickling of stainless steel, etching of metals and as an oxidiser in rocket fuels.
Phosphorus — Allotropic Forms
Phosphorus is found in many allotropic forms, the important ones being white, red and black.
White phosphorus is a translucent white waxy solid. It is poisonous, insoluble in water but soluble in carbon disulphide and glows in dark (chemiluminescence). It dissolves in boiling NaOH solution in an inert atmosphere giving PH3.
P4 + 3NaOH + 3H2O \(\to\) PH3 + 3NaH2PO2
(sodium hypophosphite)
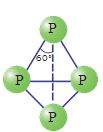
White phosphorus is less stable and therefore, more reactive than the other solid phases under normal conditions because of angular strain in the P4 molecule where the angles are only 60°. It readily catches fire in air to give dense white fumes of P4O10.
P4 + 5O2 \(\to\) P4O10
It consists of discrete tetrahedral P4 molecule as shown in Fig. 7.2.
Fig. 7.2 White phosphorus
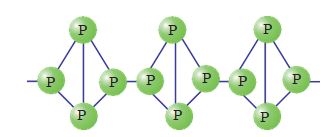
Red phosphorus is obtained by heating white phosphorus at 573K in an inert atmosphere for several days. When red phosphorus is heated under high pressure, a series of phases of black phosphorus is formed. Red phosphorus possesses iron grey lustre. It is odourless, non- poisonous and insoluble in water as well as in carbon disulphide. Chemically, red phosphorus is much less reactive than white phosphorus. It does not glow in the dark.
It is polymeric, consisting of chains of P4 tetrahedra linked together in the manner as shown in Fig. 7.3.
Fig.7.3: Red phosphorus
Black phosphorus has two forms \(\alpha\)-black phosphorus and \(\beta\)-black phosphorus. \(\alpha\)-Black phosphorus is formed when red phosphorus is heated in a sealed tube at 803K. It can be sublimed in air and has opaque monoclinic or rhombohedral crystals. It does not oxidise in air. \(\beta\)-Black phosphorus is prepared by heating white phosphorus at 473 K under high pressure. It does not burn in air upto 673 K.
Phosphine
Preparation
Phosphine is prepared by the reaction of calcium phosphide with water or dilute HCl.
Ca3P2 + 6H2O \(\to\) 3Ca(OH)2 + 2PH3
Ca3P2 + 6HCl \(\to\) 3CaCl2 + 2PH3
In the laboratory, it is prepared by heating white phosphorus with concentrated NaOH solution in an inert atmosphere of CO2.
P4 + 3NaOH + 3H2O \(\to\) PH3 + 3NaH2PO2
(sodium hypophosphite)
When pure, it is non inflammable but becomes inflammable owing to the presence of P2H4 or P4 vapours. To purify it from the impurities, it is absorbed in HI to form phosphonium iodide (PH4I) which on treating with KOH gives off phosphine.
PH4I + KOH \(\to\) KI + H2O + PH3
properties
It is a colourless gas with rotten fish smell and is highly poisonous. It explodes in contact with traces of oxidising agents like HNO3, Cl2 and Br2 vapours.
It is slightly soluble in water. The solution of PH3 in water decomposes in presence of light giving red phosphorus and H2. When absorbed in copper sulphate or mercuric chloride solution, the corresponding phosphides are obtained.
3CuSO4 + 2PH3 \(\to\) Cu3P2 + 3H2SO4
3HgCl2 + 2PH3 \(\to\) Hg3P2 + 6HCl
Phosphine is weakly basic and like ammonia, gives phosphonium compounds with acids e.g.,
PH3 + HBr \(\to\) PH4Br
USES: The spontaneous combustion of phosphine is technically used in Holme’s signals. Containers containing calcium carbide and calcium phosphide are pierced and thrown in the sea when the gases evolved burn and serve as a signal. It is also used in smoke screens.
EXAMPLE 6
In what way can it be proved that PH3 is basic in nature?
SOLUTION
PH3 reacts with acids like HI to form PH4I which shows that it is basic in nature.
PH3 + HI \(\to\) PH4I
Due to lone pair on phosphorus atom, PH3 is acting as a Lewis base in the above reaction.
INTEXT QUESTION
(a) Bond angle in PH\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaisdaaeaacqGHRaWkaaaaaa!37C3! _4^ + \) is higher than that in PH3. Why?
(b) What is formed when PH3 reacts with an acid?
What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2 ?
Phosphorus Halides
Phosphorus forms two types of halides, PX3 (X = F, Cl, Br, I) and PX5 (X = F, Cl, Br).
Phosphorus Trichloride
Preparation
It is obtained by passing dry chlorine over heated white phosphorus.
P4 + 6Cl2 \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaisdaaeaacqGHRaWkaaaaaa!37C3! _4^ + \) 4PCl3
It is also obtained by the action of thionyl chloride with white phosphorus.
P4 + 8SOCl2 \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaisdaaeaacqGHRaWkaaaaaa!37C3! _4^ + \) 4PCl3 + 4SO2 + 2S2Cl2
Properties
It is a colourless oily liquid and hydrolyses in the presence of moisture.
PCl3 + 3H2O \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaisdaaeaacqGHRaWkaaaaaa!37C3! _4^ + \) H3PO3 + 3HCl
It reacts with organic compounds containing –OH group such as CH3COOH, C2H5OH.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGceaqabeaacaaIZa % Gaam4qaiaadIeadaWgaaWcbaGaaG4maaqabaGccaWGdbGaam4taiaa % d+eacaWGibGaey4kaSIaamiuaiaadoeacaWGSbWaaSbaaSqaaiaaio % daaeqaaOGaeyOKH4QaaG4maiaadoeacaWGibWaaSbaaSqaaiaaioda % aeqaaOGaam4qaiaad+eacaWGdbGaamiBaiabgUcaRiaadIeadaWgaa % WcbaGaaG4maaqabaGccaWGqbGaam4tamaaBaaaleaacaaIZaaabeaa % aOqaaiaaiodacaWGdbWaaSbaaSqaaiaaikdaaeqaaOGaamisamaaBa % aaleaacaaI1aaabeaakiaad+eacaWGibGaey4kaSIaamiuaiaadoea % caWGSbWaaSbaaSqaaiaaiodaaeqaaOGaeyOKH4QaaG4maiaadoeada % WgaaWcbaGaaGOmaaqabaGccaWGibWaaSbaaSqaaiaaiwdaaeqaaOGa % am4qaiaadYgacqGHRaWkcaWGibWaaSbaaSqaaiaaiodaaeqaaOGaam % iuaiaad+eadaWgaaWcbaGaaG4maaqabaaaaaa!65F7! \begin{array}{l} 3C{H_3}COOH + PC{l_3} \to 3C{H_3}COCl + {H_3}P{O_3}\\ 3{C_2}{H_5}OH + PC{l_3} \to 3{C_2}{H_5}Cl + {H_3}P{O_3} \end{array}\)
Phosphorus Pentachloride
It has a pyramidal shape as shown, in which phosphorus is sp3 hybridised.
Preparation
Phosphorus pentachloride is prepared by the reaction of white phosphorus with excess of dry chlorine.
P4 + 10Cl2 \(\to\) 4PCl5
It can also be prepared by the action of SO2Cl2 on phosphorus.
P4 + 10SO2Cl2 \(\to\) 4PCl5 + 10SO2
properties
PCl5 is a yellowish white powder and in moist air, it hydrolyses to POCl3 and finally gets converted to phosphoric acid.
PCl5 + H2O \(\to\) POCl3 + 2HCl
POCl3 + 3H2O \(\to\) H3PO4 + 3HCl
When heated, it sublimes but decomposes on stronger heating.
It reacts with organic compounds containing –OH group converting them to chloro derivatives.
C2H5OH + PCl5 \(\to\) C2H5Cl + POCl3 + HCl
CH3COOH+ PCl5 \(\to\) CH3COCl + POCl3 +HCl
Finely divided metals on heating with PCl5 give corresponding chlorides.
2 Ag + PCl5 \(\to\) 2AgCl + PCl3
Sn + 2PCl5 \(\to\) SnCl4 + 2PCl3
It is used in the synthesis of some organic compounds, e.g., C2H5Cl, CH3COCl.
In gaseous and liquid phases, it has a trigonal bipyramidal structure as shown. The three equatorial P–Cl bonds are equivalent, while the two axial bonds are longer than equatorial bonds. This is due to the fact that the axial bond pairs suffer more repulsion as compared to equatorial bond pairs.
EXAMPLE 7
Why does PCl3 fume in moisture ?
SOLUTION
PCl3 hydrolyses in the presence of moisture giving fumes of HCl.
PCl3 + 3H2O \(\to\) H3PO3 + 3HCl
EXAMPLE 8
Are all the five bonds in PCl5 molecule equivalent? Justify your answer.
SOLUTION
PCl5 has a trigonal bipyramidal structure and the three equatorial P-Cl bonds are equivalent, while the two axial bonds are different and longer than equatorial bonds.
Intext Questions
What happens when PCl5 is heated?
Write a balanced equation for the reaction of PCl5 with water.
Oxoacids of Phosphorus
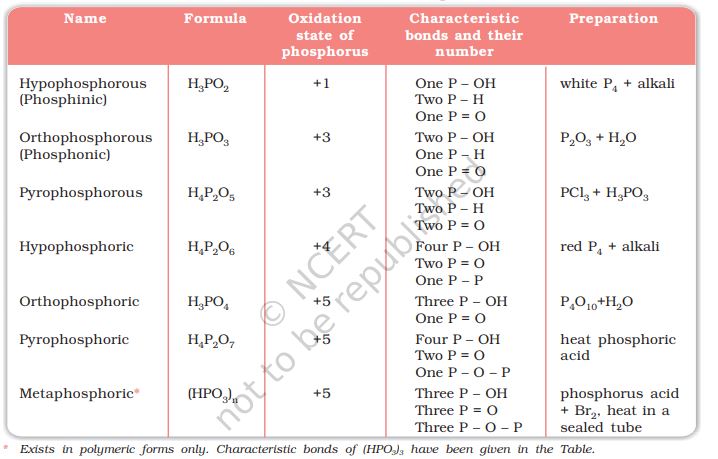
Phosphorus forms a number of oxoacids. The important oxoacids of phosphorus with their formulas, methods of preparation and the presence of some characteristic bonds in their structures are given in Table 7.5.
Table 7.5: Oxoacids of Phosphorus
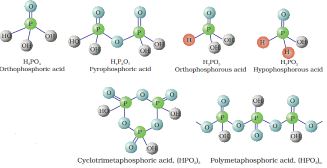
The compositions of the oxoacids are interrelated in terms of loss or gain of H2O molecule or O-atom. The structures of some important oxoacids are given next.
In oxoacids phosphorus is tetrahedrally surrounded by other atoms. All these acids contain at least one P=O bond and one P–OH bond. The oxoacids in which phosphorus has lower oxidation state (less than +5) contain, in addition to P=O and P–OH bonds, either P–P (e.g., in H4P2O6) or P–H (e.g., in H3PO2) bonds but not both. These acids in +3 oxidation state of phosphorus tend to disproportionate to higher and lower oxidation states. For example, orthophophorous acid (or phosphorous acid) on heating disproportionates to give orthophosphoric acid (or phosphoric acid) and phosphine.
4H3PO3 + 3H3PO4 \(\to\) PH3
Fig.7.4. Structures of some important oxoacids of phosphorus
The acids which contain P–H bond have strong reducing properties. Thus, hypophosphorous acid is a good reducing agent as it contains two P–H bonds and reduces, for example, AgNO3 to metallic silver.
4 AgNO3 + 2H2O + H3PO2 \(\to\) 4Ag + 4HNO3 + H3PO4
These P–H bonds are not ionisable to give H+ and do not play any role in basicity. Only those H atoms which are attached with oxygen in P–OH form are ionisable and cause the basicity. Thus, H3PO3 and H3PO4 are dibasic and tribasic, respectively as the structure of H3PO3 has two P–OH bonds and H3PO4 three.
EXAMPLE 9
How do you account for the reducing behaviour of H3PO2 on the basis of its structure ?
SOLUTION
In H3PO2, two H atoms are bonded directly to P atom which imparts reducing character to the acid.
INTEXT QUESTION
What is the basicity of H3PO4?
What happens when H3PO3 is heated?