-
Solids in Liquids
Solids in liquids
We know from our experience that we can dissolve only a limited amount of salt or sugar in a given amount of water at room temperature. If we make a thick sugar syrup solution by dissolving sugar at a higher temperature, sugar crystals separate out if we cool the syrup to the room temperature. We call it a saturated solution when no more of solute can be dissolved in it at a given temperature. The concentration of the solute in a saturated solution depends upon the temperature. In a saturated solution, a dynamic equilibrium exits between the solute molecules in the solid state and in the solution:
Sugar (solution)
Sugar (solid), and the rate of dissolution of sugar = rate of crystallisation of sugar.
Equality of the two rates and dynamic nature of equilibrium has been confirmed with the help of radioactive sugar. If we drop some radioactive sugar into saturated solution of non-radioactive sugar, then after some time radioactivity is observed both in the solution and in the solid sugar. Initially there were no radioactive sugar molecules in the solution but due to dynamic nature of equilibrium, there is exchange between the radioactive and non-radioactive sugar molecules between the two phases. The ratio of the radioactive to non- radioactive molecules in the solution increases till it attains a constant value.
-
Gases in Liquids
Gases in liquids
When a soda water bottle is opened, some of the carbon dioxide gas dissolved in it fizzes out rapidly. The phenomenon arises due to difference in solubility of carbon dioxide at different pressures. There is equilibrium between the molecules in the gaseous state and the molecules dissolved in the liquid under pressure i.e.,
CO2 (gas)
CO2 (in solution)
This equilibrium is governed by Henry’s law, which states that the mass of a gas dissolved in a given mass of a solvent at any temperature is proportional to the pressure of the gas above the solvent. This amount decreases with increase of temperature. The soda water bottle is sealed under pressure of gas when its solubility in water is high. As soon as the bottle is opened, some of the dissolved carbon dioxide gas escapes to reach a new equilibrium condition required for the lower pressure, namely its partial pressure in the atmosphere. This is how the soda water in bottle when left open to the air for some time, turns ‘flat’. It can be generalised that:
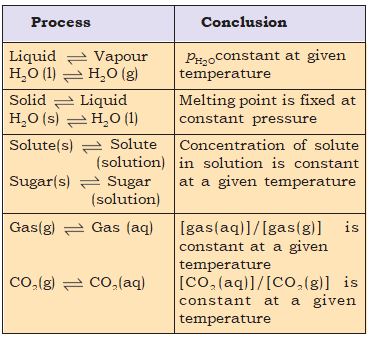
1. For solid
liquid equilibrium, there is only one temperature (melting point) at 1 atm (1.013 bar) at which the two phases can coexist. If there is no exchange of heat with the surroundings, the mass of the two phases remains constant.
2. For liquid
vapour equilibrium, the vapour pressure is constant at a given temperature.
3. For dissolution of solids in liquids, the solubility is constant at a given temperature.
4. For dissolution of gases in liquids, the concentration of a gas in liquid is proportional to the pressure (concentration) of the gas over the liquid. These observations are summarised in Table 7.1
Table 7.1 Some Features of Physical Equilibria
-
General Characteristics of Equilibria Involving Physical Processes
General Characteristics of Equilibria Involving Physical Processes
For the physical processes discussed above, following characteristics are common to the system at equilibrium:
1. Equilibrium is possible only in a closed system at a given temperature.
2. Both the opposing processes occur at the same rate and there is a dynamic but stable condition.
3. All measurable properties of the system remain constant.
4. When equilibrium is attained for a physical process, it is characterised by constant value of one of its parameters at a given temperature. Table 7.1 lists such quantities.
5. The magnitude of such quantities at any stage indicates the extent to which the physical process has proceeded before reaching equilibrium.