-
Law of Chemical Equilibrium and Equilibrium Consant
LAW OF CHEMICAL EQUILIBRIUM AND EQUILIBRIUM CONSTANT
A mixture of reactants and products in the equilibrium state is called an equilibrium mixture. In this section we shall address a number of important questions about the composition of equilibrium mixtures: What is the relationship between the concentrations of reactants and products in an equilibrium mixture? How can we determine equilibrium concentrations from initial concentrations?
What factors can be exploited to alter the composition of an equilibrium mixture? The last question in particular is important when choosing conditions for synthesis of industrial chemicals such as H2, NH3, CaO etc.
To answer these questions, let us consider a general reversible reaction:
A+B
C+D
where A and B are the reactants, C and D are the products in the balanced chemical equation. On the basis of experimental studies of many reversible reactions, the Norwegian chemists Cato Maximillian Guldberg and Peter Waage proposed in 1864 that the concentrations in an equilibrium mixture are related by the following equilibrium equation,
\({K_c} = \,\frac{{\left[ C \right]\left[ D \right]}}{{\left[ A \right]\left[ B \right]}}\)(7.1) where Kc is the equilibrium constant and the expression on the right side is called the equilibrium constant expression.
The equilibrium equation is also known as the law of mass action because in the early days of chemistry, concentration was called “active mass”. In order to appreciate their work better, let us consider reaction between gaseous H2 and I2 carried out in a sealed vessel
at 731K.
H2(g)+I2(g)
2HI(g)
1 mol 1 mol 2 mol
-
Experiment Analysis
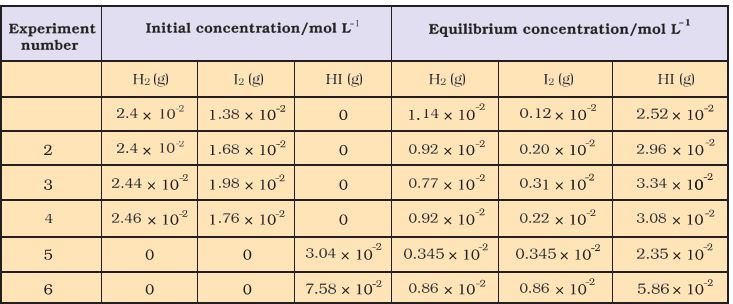
Six sets of experiments with varying initial conditions were performed, starting with only gaseous H2 and I2 in a sealed reaction vessel in first four experiments (1, 2, 3 and 4) and only HI in other two experiments (5 and 6). Experiment 1, 2, 3 and 4 were performed taking different concentrations of H2 and / or I2, and with time it was observed that intensity of the purple colour remained constant and equilibrium was attained. Similarly, for experiments 5 and 6, the equilibrium was attained from the opposite direction.Data obtained from all six sets of experiments are given in Table 7.2.
Table 7.2 Initial and Equilibrium Concentrations of H2, I2 and HI
It is evident from the experiments 1, 2, 3 and 4 that number of moles of dihydrogen reacted = number of moles of iodine reacted =
½ (number of moles of HI formed). Also,experiments 5 and 6 indicate that,
[H2(g)]eq = [I2(g)]eq
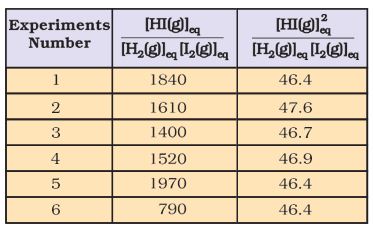
Knowing the above facts, in order to establish a relationship between concentrations of the reactants and products, several combinations can be tried. Let us consider the simple expression,
[HI(g)]eq / [H2(g)]eq [I2(g)]eq
It can be seen from Table 7.3 that if we put the equilibrium concentrations of the reactants and products, the above expression
Table 7.3 Expression Involving the Equilibrium Concentration of Reactants H2(g)+I2(g)
2HI(g) is far from constant. However, if we consider the expression,[HI(g)]eq / [H2(g)]eq [I2(g)]eq
we find that this expression gives constant value (as shown in Table 7.3) in all the six cases. It can be seen that in this expression the power of the concentration for reactants and products are actually the stoichiometric coefficients in the equation for the chemical reaction. Thus, for the reaction H2(g) +I2(g)
2HI(g), following equation 7.1, the equilibrium constant Kc is written as,
\({K_c} = \,\frac{{\left[ {HI\left( g \right)} \right]_{eq}^2}}{{{{\left[ {{H_2}(g)} \right]}_{eq}}{{\left[ {{I_2}(g)} \right]}_{eq}}}}\)\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaam4samaaBa % aaleaacaWGJbaabeaakiabg2da9maadmaabaGaamisaiaadMeacaGG % OaGaam4zaiaacMcaaiaawUfacaGLDbaadaqhaaWcbaGaamyzaiaadg % haaeaacaaIYaaaaOGaai4lamaadmaabaGaamisamaaBaaaleaacaaI % YaaabeaakiaacIcacaWGNbGaaiykaaGaay5waiaaw2faamaaBaaale % aacaWGLbGaamyCaaqabaGcdaWadaqaaiaadMeadaWgaaWcbaGaaGOm % aaqabaGccaGGOaGaam4zaiaacMcaaiaawUfacaGLDbaadaWgaaWcba % Gaamyzaiaadghaaeqaaaaa!5251! {K_c} = \left[ {HI(g)} \right]_{eq}^2/{\left[ {{H_2}(g)} \right]_{eq}}{\left[ {{I_2}(g)} \right]_{eq}}\)(7.2)
Generally the subscript ‘eq’ (used for equilibrium) is omitted from the concentration terms. It is taken for granted that the concentrations in the expression for Kc are equilibrium values. We, therefore, write,
Kc = [HI(g)]2 /[H2 (g)] [I2 (g)](7.3)
The subscript ‘c’ indicates that Kc is expressed in concentrations of mol L–1.
At a given temperature, the product of concentrations of the reaction products raised to the respective stoichiometric coefficient in the balanced chemical equation divided by the product of concentrations of the reactants raised to their individual stoichiometric coefficients has a constant value. This is known as the Equilibrium Law or Law of Chemical Equilibrium.
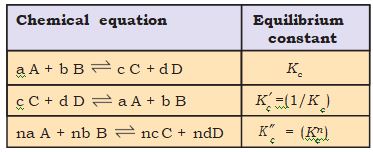
The equilibrium constant for a general reaction,
a A + b B
c C + d D is expressed as,
Kc= [C]c[D]d / [A]a[B]b (7.4)
where [A], [B], [C] and [D] are the equilibrium concentrations of the reactants and products.
Equilibrium constant for the reaction,
4NH3(g) + 5O2(g)
4NO(g) + 6H2O(g) is written as
Kc = [NO]4[H2 O]6 / [N3H ]4 [O2 ]5
Molar concentration of different species is indicated by enclosing these in square bracket and, as mentioned above, it is implied that these are equilibrium concentrations. While writing expression for equilibrium constant, symbol for phases (s, l, g) are generally ignored.
Let us write equilibrium constant for the reaction, H2(g) + I2 (g)
2HI(g) (7.5)
as, Kc = [HI]2 / [H2] [I2] = x (7.6)
The equilibrium constant for the reverse reaction, 2HI(g)
H2(g) + I2(g), at the same temperature is,
Kc = [H2 ] [I2 ] / [HI]2 = 1/ x = 1 / Kc (7.7)
Thus, \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGabm4sayaafa % WaaSbaaSqaaiaadogaaeqaaaaa!37E6! {K'_c}\) = 1 / Kc (7.8)
Equilibrium constant for the reverse reaction is the inverse of the equilibrium constant for the reaction in the forward direction.
If we change the stoichiometric coefficients in a chemical equation by multiplying throughout by a factor then we must make sure that the expression for equilibrium constant also reflects that change. For example, if the reaction (7.5) is written as,
½ H2 (g) + ½ I2 (g)
HI(g) (7.9)
the equilibrium constant for the above reaction is given by
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGabm4sayaaga % WaaSbaaSqaaiaadogaaeqaaOGaeyypa0ZaamWaaeaacaWGibGaamys % aaGaay5waiaaw2faaiaac+cadaWadaqaaiaadIeadaWgaaWcbaGaaG % OmaaqabaaakiaawUfacaGLDbaadaahaaWcbeqaaiaaigdacaGGVaGa % aGOmaaaakmaadmaabaGaamysamaaBaaaleaacaaIYaaabeaaaOGaay % 5waiaaw2faamaaCaaaleqabaGaaGymaiaac+cacaaIYaaaaOGaeyyp % a0ZaaiWaaeaadaWadaqaaiaadIeacaWGjbaacaGLBbGaayzxaaWaaW % baaSqabeaacaaIYaaaaOGaai4lamaadmaabaGaamisamaaBaaaleaa % caaIYaaabeaaaOGaay5waiaaw2faamaadmaabaGaamysamaaBaaale % aacaaIYaaabeaaaOGaay5waiaaw2faaaGaay5Eaiaaw2haamaaCaaa % leqabaGaaGymaiaac+cacaaIYaaaaOGaeyypa0JaamiEamaaCaaale % qabaGaaGymaiaac+cacaaIYaaaaOGaeyypa0Jaam4samaaDaaaleaa % caWGJbaabaGaaGymaiaac+cacaaIYaaaaaaa!6503! {K''_c} = \left[ {HI} \right]/{\left[ {{H_2}} \right]^{1/2}}{\left[ {{I_2}} \right]^{1/2}} = {\left\{ {{{\left[ {HI} \right]}^2}/\left[ {{H_2}} \right]\left[ {{I_2}} \right]} \right\}^{1/2}} = {x^{1/2}} = K_c^{1/2}\)(7.10)
On multiplying the equation (7.5) by n, we get
nH2(g) + nI2(g)D
2nHI(g) (7.11)
Therefore, equilibrium constant for the reaction is equal to \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaam4samaaDa % aaleaacaWGJbaabaGaamOBaaaakiaac6caaaa!398A! K_c^n\).These findings are summarised in Table 7.4. It should be noted that because the equilibrium constants Kc and \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGabm4sayaafa % WaaSbaaSqaaiaadogaaeqaaaaa!37E6! {K'_c}\) have different numerical values, it is important to specify the form of the balanced chemical equation when quoting the value of an equilibrium constant.
Table 7.4 Relations between Equilibrium Constants for a General Reaction and its Multiples.
-
Solved Examples
PROBLEM 1
The following concentrations were obtained for the formation of NH3 from N2 and H2 at equilibrium at 500K.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaamWaaeaaca % WGobWaaSbaaSqaaiaaikdaaeqaaaGccaGLBbGaayzxaaGaeyypa0Ja % aGymaiaac6cacaaI1aGaey41aqRaaGymaiaaicdadaahaaWcbeqaai % abgkHiTiaaikdaaaGccaWGnbGaaiOlamaadmaabaGaamisamaaBaaa % leaacaaIYaaabeaaaOGaay5waiaaw2faaiabg2da9iaaiodacaGGUa % GaaGimaiabgEna0kaaigdacaaIWaWaaWbaaSqabeaacqGHsislcaaI % YaaaaOGaamytaaaa!50ED! \left[ {{N_2}} \right] = 1.5 \times {10^{ - 2}}M.\left[ {{H_2}} \right] = 3.0 \times {10^{ - 2}}M\) and \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaamWaaeaaca % WGobGaamisamaaBaaaleaacaaIZaaabeaaaOGaay5waiaaw2faaiab % g2da9iaaigdacaGGUaGaaGOmaiabgEna0kaaigdacaaIWaWaaWbaaS % qabeaacqGHsislcaaIYaaaaOGaamytaiaac6caaaa!449A! \left[ {N{H_3}} \right] = 1.2 \times {10^{ - 2}}M.\) Calculate equilibrium constant.
SOLUTION
The equilibrium constant for the reaction,
N2(g) + 3H2(g)
2NH3(g) can be written as,
\({K_c} = \,\frac{{{{\left[ {N{H_3}(g)} \right]}^2}}}{{\left[ {{N_2}(g)} \right]{{\left[ {{H_2}(g)} \right]}^3}}}\)
\({K_c} = \,\frac{{{{\left[ {1.2x{{10}^{ - 2}}} \right]}^2}}}{{\left[ {1.5x{{10}^{ - 2}}} \right]{{\left[ {3.0x{{10}^{ - 2}}} \right]}^3}}}\)
= 0.106 x 104 = 1.06 x 103
PROBLEM 2
At equilibrium, the concentrations of N2 =3.0 × 10–3M, O2 = 4.2 × 10–3M and NO= 2.8 × 10–3M in a sealed vessel at 800K. What will be Kc for the reaction N2(g) + O2(g)
2NO(g)
SOLUTION
For the reaction equilibrium constant \({K_c}\) can be written as
\({K_c} = \,\frac{{{{\left[ {NO} \right]}^2}}}{{\left[ {{N_2}} \right]{{\left[ {{O_2}} \right]}^3}}}\)
\({K_c} = \,\frac{{{{\left[ {2.8x{{10}^{ - 3}}M} \right]}^2}}}{{\left[ {3x{{10}^{ - 3}}M} \right]\left[ {4.2x{{10}^{ - 2}}M} \right]}}\)
= 0.622