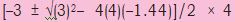-
Homogeneous Equilibria
HOMOGENEOUSEQUILIBRIA
In a homogeneous system, all the reactants and products are in the same phase. For example,in the gaseous reaction,
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamOtamaaBa % aaleaacaaIYaaabeaakmaabmaabaGaam4zaaGaayjkaiaawMcaaiab % gUcaRiaaiodacaWGibWaaSbaaSqaaiaaikdaaeqaaOWaaeWaaeaaca % WGNbaacaGLOaGaayzkaaaaaa!4003! {N_2}\left( g \right) + 3{H_2}\left( g \right)\)
2NH3(g), reactants and products are in the homogeneous phase.
Similarly, for the reactions,
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaam4qaiaadI % eadaWgaaWcbaGaaG4maaqabaGccaWGdbGaam4taiaad+eacaWGdbWa % aSbaaSqaaiaaikdaaeqaaOGaamisamaaBaaaleaacaaI1aaabeaakm % aabmaabaGaamyyaiaadghaaiaawIcacaGLPaaacqGHRaWkcaWGibWa % aSbaaSqaaiaaikdaaeqaaOGaam4tamaabmaabaGaaGymaaGaayjkai % aawMcaaaaa!4788! C{H_3}COO{C_2}{H_5}\left( {aq} \right) + {H_2}O\left( 1 \right)\)
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaam4qaiaadI % eadaWgaaWcbaGaaG4maaqabaGccaWGdbGaam4taiaad+eacaWGibWa % aeWaaeaacaWGHbGaamyCaaGaayjkaiaawMcaaiabgUcaRiaadoeada % WgaaWcbaGaaGOmaaqabaGccaWGibWaaSbaaSqaaiaaiwdaaeqaaOGa % am4taiaadIeadaqadaqaaiaadggacaWGXbaacaGLOaGaayzkaaaaaa!4884! C{H_3}COOH\left( {aq} \right) + {C_2}{H_5}OH\left( {aq} \right)\) and, Fe3+ (aq) + SCN–(aq)
Fe(SCN)2+ (aq) all the reactants and products are in homogeneous solution phase. We shall now consider equilibrium constant for some homogeneous reactions.
Equilibrium Constant in Gaseous System
So far we have expressed equilibrium constant of the reactions in terms of molar concentration of the reactants and products,and used symbol, Kc for it. For reactions involving gases, however, it is usually more convenient to express the equilibrium constant in terms of partial pressure.
The ideal gas equation is written as,
\(\begin{array}{l} pV = nRT\\ \Rightarrow p = \frac{n}{V}RT \end{array}\)
Here, p is the pressure in Pa, n is the number of moles of the gas, V is the volume in m3 and T is the temperature in Kelvin
Therefore,n/V is concentration expressed in mol/m3
If concentration c, is in mol/L or mol/dm3, and p is in bar then p = cRT,
We can also write p = [gas]RT.
Here, R= 0.0831 bar litre/mol K
At constant temperature, the pressure of the gas is proportional to its concentration i.e.,\(p\infty \left[ {gas} \right]\)
For reaction in equilibrium \({H_2}\left( g \right) + {I_2}\left( g \right)\)
2HI(g)
We can write either
\( \begin{array}{l} {K_c} = \frac{{{{\left[ {HI\left( g \right)} \right]}^2}}}{{\left[ {{H_2}\left( g \right)} \right]\left[ {{I_2}\left( g \right)} \right]}}\\ or\,\,\,\,\,{K_c} = \frac{{{{\left( {{p_{HI}}} \right)}^2}}}{{\left( {{p_{{H_2}}}} \right)\left( {{p_{{I_2}}}} \right)}}\,\,\,\,\,\,\left( {7.12} \right) \end{array}\)
Futher,since
\(\begin{array}{l} {p_{HI}} = \left[ {HI\left( g \right)} \right]RT\\ {p_{{I_2}}} = \left[ {{I_2}\left( g \right)} \right]RT\\ {p_{{H_2}}} = \left[ {{H_2}\left( g \right)} \right]RT \end{array}\)
Therefore,
\(\begin{array}{l} {K_p} = \frac{{{{\left( {{p_{HI}}} \right)}^2}}}{{\left( {{p_{{H_2}}}} \right)\left( {{p_{{I_2}}}} \right)}} = \frac{{{{\left[ {HI\left( g \right)} \right]}^2}{{\left[ {RT} \right]}^2}}}{{\left[ {{H_2}\left( g \right)} \right]RT.\left[ {{I_2}\left( g \right)} \right]RT}}\\ = \frac{{{{\left[ {HI\left( g \right)} \right]}^2}}}{{\left[ {{H_2}\left( g \right)} \right]\left[ {{I_2}\left( g \right)} \right]}} = {K_c}\,\,\,\,\,\,\left( {7.13} \right) \end{array}\)
In this example, Kp = Kc i.e., both equilibrium constants are equal.However, this is not always the case. For example in reaction
\({N_2}\left( g \right) + 3{H_2}\left( g \right)\)
2NH3(g)
\( \begin{array}{l} {K_p} = \frac{{{{\left( {{p_{N{H_3}}}} \right)}^2}}}{{\left( {{p_{{N_2}}}} \right){{\left( {{p_{{H_2}}}} \right)}^3}}}\\ = \frac{{{{\left[ {N{H_3}\left( g \right)} \right]}^2}{{\left[ {RT} \right]}^2}}}{{\left[ {{N_2}\left( g \right)} \right]RT.{{\left[ {{H_2}\left( g \right)} \right]}^3}{{\left( {RT} \right)}^3}}}\\ = \frac{{{{\left[ {N{H_3}\left( g \right)} \right]}^2}{{\left[ {RT} \right]}^{ - 2}}}}{{\left[ {{N_2}\left( g \right)} \right]{{\left[ {{H_2}\left( g \right)} \right]}^3}}} = {K_c}{\left( {RT} \right)^{ - 2}}\\ or\,\,\,\,{K_p} = {K_c}{\left( {RT} \right)^{ - 2}}\,\,\,\,\,\,\,\left( {7.14} \right) \end{array}\)
Similarly, for a general reaction
a A + b B
c C + d D
\(\begin{array}{l} {K_p} = \frac{{\left( {p_C^c} \right)\left( {p_D^d} \right)}}{{\left( {p_A^a} \right)\left( {p_B^b} \right)}} = \frac{{{{\left[ C \right]}^c}{{\left[ D \right]}^d}{{\left( {RT} \right)}^{\left( {c + d} \right)}}}}{{{{\left[ A \right]}^a}{{\left[ B \right]}^b}{{\left( {RT} \right)}^{\left( {a + b} \right)}}}}\\ = \frac{{{{\left[ C \right]}^c}{{\left[ D \right]}^d}}}{{{{\left[ A \right]}^a}{{\left[ B \right]}^b}}}{\left( {RT} \right)^{\left( {c + d} \right) - \left( {a + d} \right)}}\\ = \frac{{{{\left[ C \right]}^c}{{\left[ D \right]}^d}}}{{{{\left[ A \right]}^a}{{\left[ B \right]}^b}}}{\left( {RT} \right)^{\Delta n}} = {K_c}{\left( {RT} \right)^{\Delta n}}\,\,\,\,\left( {7.15} \right) \end{array}\)
where \(\Delta\)n=(number of moles of gaseous products) – (number of moles of gaseous reactants) in the balanced chemical equation.It is necessary that while calculating the value of Kp, pressure should be expressed in bar because standard state for pressure is 1 bar.
We know from Unit 1 that :
1pascal, Pa=1Nm–2, and 1bar = 105 Pa
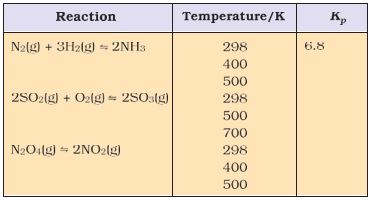
Kp values for a few selected reactions at different temperatures are given in Table 7.5
Table 7.5 Equilibrium Constants, Kp for a Few Selected Reactions
PROBLEM 3
PCl5, PCl3 and Cl2 are at equilibrium at 500 K and having concentration 1.59M PCl3, 1.59M Cl2 and 1.41 M PCl5.Calculate Kc for the reaction,PCl5
PCl3 + Cl2 .
SOLUTION
The equilibrium constant Kc for the above reaction can be written as,\({K_c} = \frac{{\left[ {PC{l_3}} \right]\left[ {C{l_2}} \right]}}{{\left[ {PC{l_5}} \right]}} = \frac{{{{\left( {1.59} \right)}^2}}}{{\left( {1.41} \right)}} = 1.79\)
PROBLEM 4
The value of Kc = 4.24 at 800K for the reaction, CO (g) + H2O (g)
CO2(g)+H2(g).Calculate equilibrium concentrations of CO2, H2, CO and H2O at 800 K, if only CO and H2O are present initially at concentrations of 0.10M each.
SOLUTION
For the reaction,CO (g) +H2O (g)
CO2 (g) + H2 (g)
Initial concentration:
0.1M 0.1M 0 0
Let x mole per litre of each of the product be formed.
At equilibrium:
(0.1-x) M (0.1-x) M x M x M
where x is the amount of CO2 and H2 at equilibrium.
Hence, equilibrium constant can be written as,
Kc = x2/(0.1-x)2 = 4.24
x2 = 4.24(0.01 + x2-0.2x)
x2 = 0.0424 + 4.24x2-0.848x 3.24x2 – 0.848x + 0.0424 = 0
a = 3.24, b = – 0.848, c = 0.0424
(for quadratic equation ax2 + bx + c = 0,
\(\begin{array}{l} x = \frac{{\left( { - b \pm \sqrt {{b^2} - 4ac} } \right)}}{{2a}}\\ x = 0.848 \pm {\left( {0.848} \right)^2} - 4\left( {3.24} \right)\left( {0.0424} \right)/\left( {3.24 \times 2} \right)\\ x = \left( {0.848 \pm 0.4118} \right)/6.48\\ {x_1} = \left( {0.848 - 0.4118} \right)/6.48 = 0.067\\ {x_2} = \left( {0.848 + 0.4118} \right)/6.48 = 0.194 \end{array}\)
the value 0.194 should be neglected because it will give concentration of the reactant which is more than initial concentration.
Hence the equilibrium concentrations are, [CO2] = [H2-] = x = 0.067 M
[CO] = [H2O] = 0.1 – 0.067 = 0.033 M.
PROBLEM 5
For the equilibrium, 2NOCl(g)
2NO(g) + Cl2(g) the value of the equilibrium constant,Kc is 3.75 × 10–6 at 1069 K.Calculate the Kp for the reaction at this temperature?
SOLUTION
We know that,\({K_p} = {K_c}{\left( {RT} \right)^{\Delta n}}\)
For the above reaction,
\(\begin{array}{l} \Delta n = \left( {2 + 1} \right) - 2 = 1\\ {K_p} = 3.75 \times {10^{ - 6}}\left( {0.0831 \times 1069} \right)\\ {K_n} = 0.033 \end{array}\)
-
Heterogeneous Equilibria
HETEROGENEOUS EQUILIBRIA
Equilibrium in a system having more than one phase is called heterogeneous equilibrium.The equilibrium between water vapour and liquid water in a closed container is an example of heterogeneous equilibrium.
\({H_2}O\left( 1 \right)\)
H2O(g)
In this example, there is a gas phase and a liquid phase. In the same way, equilibrium between a solid and its saturated solution,Ca(OH)2(s)
Ca2+(aq)+2OH-(aq) is a heterogeneous equilibrium.
Heterogeneous equilibria often involve pure solids or liquids. We can simplify equilibrium expressions for the heterogeneous equilibria involving a pure liquid or a pure solid, as the molar concentration of a pure solid or liquid is constant (i.e., independent of the amount present). In other words if a substance ‘X’ is involved, then [X(s)] and [X(l)] are constant, whatever the amount of ‘X’ is taken. Contrary to this, [X(g)] and [X(aq)] will vary as the amount of X in a given volume varies. Let us take thermal dissociation of calcium carbonate which is an interesting and important example of heterogeneous chemical equilibrium.
(7.16)
On the basis of the stoichiometric equation,we can write,
\(K{ _c} = \frac{{\left[ {CaO\left( s \right)} \right]\left[ {C{O_2}\left( g \right)} \right]}}{{\left[ {CaC{O_3}\left( s \right)} \right]}}\)
Since [CaCO3(s)] and [CaO(s)] are both constant,therefore modified equilibrium constant for the thermal decomposition of calcium carbonate will be
\({K'_c} = \left[ {C{O_2}\left( g \right)} \right]\)(7.17)
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaam4Baiaadk % hacaaMc8UaaGPaVlaaykW7caaMc8Uaam4samaaBaaaleaacaWGWbaa % beaakiabg2da9iaadchadaWgaaWcbaGaam4qaiaad+eadaWgaaadba % GaaGOmaaqabaaaleqaaaaa!44BF! or\,\,\,\,{K_p} = {p_{C{O_2}}}\)(7.18)
This shows that at a particular temperature, there is a constant concentration or pressure of CO2 in equilibrium with CaO(s) and CaCO3(s). Experimentally it has been found that at 1100 K,the pressure of CO2 in equilibrium with CaCO3(s) and CaO(s),is 2.0 ×105 Pa.Therefore, equilibrium constant at 1100K for the above reaction is:
\({K_p} = {P_{C{O_2}}} = 2 \times {10^5}Pa/{10^5}Pa = 2.00\)
Similarly, in the equilibrium between nickel, carbon monoxide and nickel carbonyl (used in the purification of nickel),
\( Ni\left( s \right) + 4CO(g)\)
Ni(CO)4 (g),
the equilibrium constant is written as \({K_c} = \frac{{\left[ {Ni{{\left( {CO} \right)}_4}} \right]}}{{{{\left[ {CO} \right]}^4}}}\)
It must be remembered that for the existence of heterogeneous equilibrium pure solids or liquids must also be present (however small the amount may be) at equilibrium, but their concentrations or partial pressures do not appear in the expression of the equilibrium constant. In the reaction,
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamyqaiaadE % gadaWgaaWcbaGaaGOmaaqabaGccaWGpbWaaeWaaeaacaWGZbaacaGL % OaGaayzkaaGaey4kaSIaaGOmaiaadIeacaWGobGaam4tamaaBaaale % aacaaIZaaabeaakmaabmaabaGaamyyaiaadghaaiaawIcacaGLPaaa % aaa!4459! A{g_2}O\left( s \right) + 2HN{O_3}\left( {aq} \right)\)
\(2AgN{O_3}\left( {aq} \right) + {H_2}O\left( 1 \right)\)
\({K_c} = \frac{{{{\left[ {AgN{O_3}} \right]}^2}}}{{{{\left[ {HN{O_3}} \right]}^2}}}\)
PROBLEM 7
The value of Kp for the reaction, CO2 (g) + C (s)
2CO (g) is 3.0 at 1000 K. If initially PCO = 0.48 bar and PCO = 0 bar and pure graphite is present, calculate the equilibrium partial pressures of CO and CO2.
SOLUTION
For the reaction,
let ‘x’ be the decrease in pressure of CO2, then CO2 (g) + C (s)
2CO (g)
Initial
pressure: 0.48 bar 0
At equilibrium:
(0.48 – x)bar 2xbar
\(\begin{array}{l} {K_p} = \frac{{p_{CO}^2}}{{{p_{C{O_2}}}}}\\ {K_p} = {\left( {2x} \right)^2}/\left( {0.48 - x} \right) = 3\\ 4{x^2} = 3\left( {0.48 - x} \right)\\ 4{x^2} = 1.44 - x\\ 4{x^2} + 3x - 1.44 = 0\\ a = 4,b = 3,c = - 1.44\\ x = \frac{{\left( { - b \pm \sqrt {{b^2} - 4ac} } \right)}}{{2a}} \end{array}\)
=
= (–3 ± 5.66)/8
= (–3 + 5.66)/ 8 (as value of x cannot be negative hence we neglect that value)
x = 2.66/8 = 0.33
The equilibrium partial pressures are,
pCO2 = 2x = 2 × 0.33 = 0.66 bar
pCO2 = 0.48 – x = 0.48 – 0.33 = 0.15 bar