-
Important features of Equilibrium
APPLICATIONS OF EQUILIBRIUM CONSTANTS
Before considering the applications of equilibrium constants, let us summarise the important features of equilibrium constants as follows:
1. Expression for equilibrium constant is applicable only when concentrations of the reactants and products have attained
constant value at equilibrium state.
2. The value of equilibrium constant is independent of initial concentrations of the reactants and products.3. Equilibrium constant is temperature dependent having one unique value for a particular reaction represented by a balanced equation at a given temperature.
4. The equilibrium constant for the reverse reaction is equal to the inverse of the equilibrium constant for the forward reaction.
5. The equilibrium constant K for a reaction is related to the equilibrium constant of the corresponding reaction, whose equation is obtained by multiplying or dividing the equation for the original reaction by a small integer.
Let us consider applications of equilibrium constant to:
(a) predict the extent of a reaction on the basis of its magnitude,
(b) predict the direction of the reaction, and
(c) calculate equilibrium concentrations.
-
Predicting the Extent of a Reaction
Predicting the Extent of a Reaction
The numerical value of the equilibrium constant for a reaction indicates the extent of the reaction.But it is important to note that an equilibrium constant does not give any information about the rate at which the equilibrium is reached.The magnitude of Kc or Kp is directly proportional to the concentrations of products (as these appear in the numerator of equilibrium constant expression) and inversely proportional to the concentrations of the reactants (these appear in the denominator).This implies that a high value of K is suggestive of a high concentration of products and vice-versa.
We can make the following generalisations concerning the composition of
equilibrium mixtures:
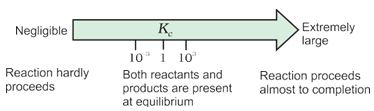
1. If Kc > 103, products predominate over reactants, i.e., if Kc is very large, the reaction proceeds nearly to completion.Consider the following examples:
(a) The reaction of H2 with O2 at 500 K has a very large equilibrium constant ,Kc =2.4 × 1047.
(b) H2 (g) + Cl2 (g)
2HCl(g) at 300K has Kc = 4.0 × 1031.
(c) H2(g) + Br2(g)
2HBr (g) at 300 K,Kc = 5.4 × 1018.
2. If Kc < 10-3 , reactants predominate over products, i.e., if Kc is very small, the reaction proceeds rarely. Consider the following examples:
(a)The decomposition of H2O into H2 and O2 at 500 K has a very small equilibrium constant, Kc = 4.1 × 10–48.
(b)N2(g) + O2(g)
2NO(g),at 298 K has Kc = 4.8 ×10–31.
3. If Kc is in the range of 10 –3 to 103, appreciable concentrations of both reactants and products are present.Consider the following examples:
(a) For reaction of H2 with I2 to give HI,Kc = 57.0 at 700K.
(b) Also, gas phase decomposition of N2O4 to NO2 is another reaction with a value of Kc = 4.64 × 10–3 at 25°C which is neither too small nor too large. Hence,equilibrium mixtures contain appreciable concentrations of both N2O4 and NO2.
These generarlisations are illustrated in Fig. 7.6
Fig.7.6 Dependence of extent of reaction on Kc
-
Predicting the Direction of the Reaction
Predicting the Direction of the Reaction
The equilibrium constant helps in predicting the direction in which a given reaction will proceed at any stage.For this purpose, we calculate the reaction quotient Q. The reaction quotient,Q (Qc with molar concentrations and QP with partial pressures)is defined in the same way as the equilibrium constant Kc except that the concentrations in Qc are not necessarily equilibrium values.For a general reaction:
a A + b B
c C + d D (7.19)
Qc = [C]c[D]d / [A]a[B]b (7.20)
Then,
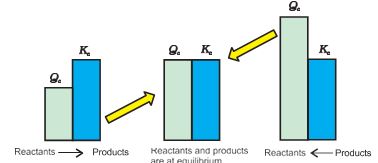
If Qc > Kc, the reaction will proceed in the direction of reactants (reverse reaction).
If Qc < Kc, the reaction will proceed in the direction of the products (forward reaction).
If Qc = Kc, the reaction mixture is already at equilibrium.
Consider the gaseous reaction of H2 with I2,
H2(g)+I2(g)
2HI(g);Kc = 57.0 at 700 K.
Suppose we have molar concentrations [H2]t=0.10M, [I2]t = 0.20 M and [HI]t = 0.40 M.(the subscript t on the concentration symbols means that the concentrations were measured at some arbitrary time t, not necessarily at equilibrium).
Thus, the reaction quotient,Qc at this stage of the reaction is given by,
Q = [HI]\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aadshaaeaacaaIYaaaaaaa!37D8! _t^2\) / [H2]t[I2]t= (0.40)2/ (0.10)×(0.20)=8.0
Now, in this case, Qc (8.0) does not equal Kc (57.0), so the mixture of H2(g), I2(g) and HI(g) is not at equilibrium; that is, more H2(g) and I2(g) will react to form more HI(g) and their concentrations will decrease till Qc = Kc.
The reaction quotient, Qc is useful in predicting the direction of reaction by comparing the values of Qc and Kc.
Thus, we can make the following
generalisations concerning the direction of the reaction (Fig. 7.7) :
Fig. 7.7 Predicting the direction of the reaction
1. If Qc< Kc , net reaction goes from left to right
2. If Qc > Kc , net reaction goes from right to left.
3. If Qc > Kc , no net reaction occurs.
PROBLEM 7
The value of Kc for the reaction 2A B
C is 2 × 10–3. At a given time, the composition of reaction mixture is [A] = [B] = [C] = 3 × 10–4 M. In which
direction the reaction will proceed?
SOLUTION
For the reaction the reaction quotient Qc is given by,
Qc = [B][C]/ [A]2
as [A] = [B] = [C] = 3 × 10–4M
Qc = (3 ×10 )(3 × 10 ) / (3 ×10 ) = 1
as Qc > Kc so the reaction will proceed in the reverse direction.
-
Calculating Equilibrium Concentrations
Calculating Equilibrium Concentrations
In case of a problem in which we know the initial concentrations but do not know any of the equilibrium concentrations, the following three steps shall be followed:
Step 1. Write the balanced equation for the reaction.
Step 2. Under the balanced equation, make a table that lists for each substance involved in the reaction:
a. the initial concentration,
b. the change in concentration on going to equilibrium, and
c. the equilibrium concentration.
In constructing the table, define x as the concentration (mol/L) of one of the substances that reacts on going to equilibrium, then use the stoichiometry of the reaction to determine the concentrations of the other substances in terms of x.
Step 3. Substitute the equilibrium concentrations into the equilibrium equation for the reaction and solve for x. If you are to solve a quadratic equation choose the mathematical solution that makes chemical sense.
Step 4. Calculate the equilibrium concentrations from the calculated value of x.
Step 5. Check your results by substituting them into the equilibrium equation.
PROBLEM 8
13.8g of N2O4 was placed in a 1L reaction vessel at 400K and allowed to attain equilibrium N2O4 (g)
2NO2 (g).The total pressure at equilbrium was found to be 9.15 bar. Calculate Kc, Kp and partial pressure at equilibrium.
SOLUTION
We know pV = nRT
Total volume (V ) = 1 L Molecular mass of N2O4 = 92 g
Number of moles = 13.8g/92 g = 0.15
of the gas (n)
Gas constant (R) = 0.083 bar L mol–1K–1 Temperature (T ) = 400 K
pV = nRT
p × 1L = 0.15 mol × 0.083 bar L mol–1K–1× 400 K
p = 4.98 bar
N2O4
2NO2
Initial pressure: 4.98 bar
At equilibrium: (4.98 – x) bar 2x bar Hence,
ptotal at equilibrium =\( {p_{{N_2}{O_4}}} + {p_{N{O_2}}}\)
9.15 = (4.98 – x) + 2x
9.15 = 4.98 + x
x = 9.15 – 4.98 = 4.17 bar
Partial pressures at equilibrium are,
\({p_{{N_2}{O_4}}}\)= 4.98 – 4.17 = 0.81bar
\({p_{N{O_2}}}\) = 2x = 2 × 4.17 = 8.34 bar
\( {K_p} = {\left( {{p_{N{O_2}}}} \right)^2}/{p_{{N_2}{O_4}}}\)
= (8.34)2/0.81 = 85.87
\({K_p} = {K_c}{\left( {RT} \right)^{\Delta n}}\)
85.87 = Kc (0.083 × 400)1
Kc = 2.586 = 2.6
PROBLEM 9
3.00 mol of PCl5 kept in 1L closed reaction vessel was allowed to attain equilibrium at 380K. Calculate composition of the mixture at equilibrium. Kc= 1.80.
SOLUTION
Initial PCl5
PCl3+Cl2
concentration: 3.0 0 0
Let x mol per litre of PCl5 be dissociated,
At equilibrium:(3x-x) x x
Kc = [PCl3][Cl2]/[PCl5]
1.8 = x2/ (3 – x)
x2 + 1.8x – 5.4 = 0
x = [–1.8 ±
(1.8)2 – 4(–5.4)]/2
x = [–1.8 ±
3.24 + 21.6]/2 x = [–1.8 ± 4.98]/2
x = [–1.8 + 4.98]/2 = 1.59
[PCl5] = 3.0 – x = 3 –1.59 = 1.41 M [PCl3] = [Cl2] = x = 1.59 M