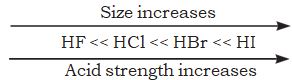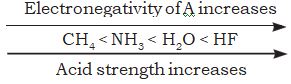-
Ionization of Acids and Bases
IONIZATIONOF ACIDS AND BASES
Arrhenius concept of acids and bases becomes useful in case of ionization of acids and bases as mostly ionizations in chemical and biological systems occur in aqueous medium.Strong acids like perchloric acid (HClO4),hydrochloric acid (HCl), hydrobromic acid (HBr), hyrdoiodic acid (HI), nitric acid (HNO3) and sulphuric acid (H2SO4) are termed strong because they are almost completely dissociated into their constituent ions in an aqueous medium, thereby acting as proton (H+) donors. Similarly, strong bases like lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), caesium hydroxide (CsOH) and barium hydroxide Ba(OH)2 are almost completely dissociated into ions in an aqueous medium giving hydroxyl ions, OH–. According to Arrhenius concept they are strong acids and bases as they are able to completely dissociate and produce \({H_3}{O^ + }\) and OH- ions respectively in the medium.
Alternatively, the strength of an acid or base may also be gauged in terms of Brönsted- Lowry concept of acids and bases, wherein a strong acid means a good proton donor and a strong base implies a good proton acceptor. Consider, the acid-base dissociation equilibrium of a weak acid HA,
HA(aq) + H2O(l)
H3O+(aq) + A–(aq)
conjugate conjugate
acid base acid base
In section 7.10.2 we saw that acid (or base) dissociation equilibrium is dynamic involving a transfer of proton in forward and reverse directions. Now, the question arises that if the equilibrium is dynamic then with passage of time which direction is favoured? What is the driving force behind it? In order to answer these questions we shall deal into the issue of comparing the strengths of the two acids (or bases) involved in the dissociation equilibrium. Consider the two acids HA and H3O+ present in the above mentioned acid-dissociation equilibrium. We have to see which amongst them is a stronger proton donor. Whichever exceeds in its tendency of donating a proton over the other shall be termed as the stronger acid and the equilibrium will shift in the direction of weaker acid. Say, if HA is a stronger acid than H3O+, then HA will donate protons and not H3O+, and the solution will mainly contain A and H3O+ ions.The equilibrium moves in the direction of formation of weaker acid and weaker base because the stronger acid donates a proton to the stronger base.
It follows that as a strong acid dissociates completely in water, the resulting base formed would be very weak i.e., strong acids have very weak conjugate bases. Strong acids like perchloric acid (HClO4), hydrochloric acid (HCl), hydrobromic acid (HBr), hydroiodic acid (HI), nitric acid (HNO3) and sulphuric acid (H2SO4) will give conjugate base ions ClO\(_4^ - \), Cl,Br–, I–, NO\( _3^ - \) and HSO\( _4^ - \), which are much weaker bases than H2O. Similarly a very strong base would give a very weak conjugate acid. On the other hand, a weak acid say HA is only partially dissociated in aqueous medium and thus, the solution mainly contains undissociated HA molecules. Typical weak acids are nitrous acid (HNO2), hydrofluoric acid (HF) and acetic acid (CH3COOH).It should be noted that the weak acids have very strong conjugate bases.For example, NH\(_2^ - \),O2– and H– are very good proton acceptors and thus, much stronger bases than H2O.
Certain water soluble organic compounds like phenolphthalein and bromothymol blue behave as weak acids and exhibit different
colours in their acid (HIn) and conjugate base (In ) forms.
HIn(aq) + H2O(1)
H3O+(aq)+In-(aq)
acid conjugate conjugate
indicator acid base
colour A colourB
Such compounds are useful as indicators in acid-base titrations, and finding out H+ ion concentration.
-
The Ionization Constant of Water and its Ionic Product
The Ionization Constant of Water and its Ionic Product
Some substances like water are unique in their ability of acting both as an acid and a base.We have seen this in case of water in section 7.10.2. In presence of an acid, HA it accepts a proton and acts as the base while in the presence of a base, B- it acts as an acid by donating a proton. In pure water, one H2O molecule donates proton and acts as an acid and another water molecules accepts a proton and acts as a base at the same time. The following equilibrium exists:
H2O(1)+H2O(1)
H3O+(aq)+OH-(aq)
acid base conjugate conjugate
acid base
The dissociation constant is represented by,
K=[H3O+][OH-]/[H2O] (7.26)
The concentration of water is omitted from the denominator as water is a pure liquid and its concentration remains constant. [H2O] is incorporated within the equilibrium constant to give a new constant,Kw,which is called the ionic product of water.
Kw=[H+][OH-] (7.27)
The concentration of H+ has been found out experimentally as 1.0 × 10–7 M at 298 K. And, as dissociation of water produces equal number of H+ and OH– ions, the concentration of hydroxyl ions, [OH–] = [H+] = 1.0 × 10–7 M. Thus, the value of Kw at 298K,
Kw=[H O+][OH ] =(1 × 10–7)2 = 1 × 10–14 M2 (7.28)
The value of Kw is temperature dependent as it is an equilibrium constant.The density of pure water is 1000 g / L and its molar mass is 18.0 g /mol. From this the molarity of pure water can be given as,
[H2O]=(1000g/L)(1mol/18.0 g)=55.55 M.
Therefore, the ratio of dissociated water to that of undissociated water can be given as:
10–7 / (55.55) = 1.8 × 10–9 or ~ 2 in 10–9 (thus,equilibrium lies mainly towards undissociated water)
We can distinguish acidic, neutral and basic aqueous solutions by the relative values of the H3O+ and OH– concentrations:
Acidic: [H3O ] > [OH-]
Neutral: [H3O ] = [OH-]
Basic : [H3O ] < [OH-
-
The pH Scale
The pH Scale
Hydronium ion concentration in molarity is more conveniently expressed on a logarithmic scale known as the pH scale. The pH of a solution is defined as the negative logarithm to base 10 of the activity (aH+) of hydrogen ion. In dilute solutions (< 0.01 M), activity of hydrogen ion (H+) is equal in magnitude to molarity represented by [H+]. It should be noted that activity has no units and is defined as:
= [H+] / mol L–1
From the definition of pH, the following can be written,
pH=-log aH+=-log{[H+]/mol L-1}
Thus, an acidic solution of HCl (10–2 M) will have a pH = 2. Similarly, a basic solution of NaOH having [OH ] =10-4 M and [H3O+]=10-10 M will have a pH = 10. At 25 °C, pure water has a concentration of hydrogen ions, [H+] = 10–7 M. Hence, the pH of pure water is given as:
pH = –log(10–7) = 7
Acidic solutions possess a concentration of hydrogen ions, [H+] > 10–7 M, while basic solutions possess a concentration of hydrogen ions, [H+] < 10–7 M. thus, we can summarise that
Acidic solution has pH < 7
Basic solution has pH > 7
Neutral solution has pH = 7
Now again, consider the equation (7.28) at 298 K
\({K_w} = \left[ {{H_3}{O^ + }} \right]\left[ {O{H^ - }} \right] = {10^{ - 14}}\)
Taking negative logarithm on both sides of equation, we obtain
\(\begin{array}{l} - \log {K_w} = - \log \left\{ {\left[ {{H_3}{O^ + }} \right]\left[ {O{H^ - }} \right]} \right\}\\ = - \log \left[ {{H_3}{O^ + }} \right] - \log \left[ {O{H^ - }} \right]\\ = - 1og{10^{ - 14}}\\ p{K_w} = pH + pOH = 14\,\,\,\,\,\,\,\,\left( {7.29} \right) \end{array}\)
Note that although Kw may change with temperature the variations in pH with temperature are so small that we often ignore it.pKw is a very important quantity for aqueous solutions and controls the relative concentrations of hydrogen and hydroxyl ions as their product is a constant. It should be noted that as the pH scale is logarithmic, a change in pH by just one unit also means change in [H+] by a factor of 10. Similarly, when the hydrogen ion concentration, [H+] changes by a factor of 100, the value of pH changes by 2 units.Now you can realise why the change in pH with temperature is often ignored.Measurement of pH of a solution is very essential as its value should be known when dealing with biological and cosmetic applications. The pH of a solution can be found roughly with the help of pH paper that has different colour in solutions of different pH.
Now-a-days pH paper is available with four strips on it.The different strips have different colours (Fig. 7.11) at the same pH.The pH in the range of 1-14 can be determined with an accuracy of ~0.5 using pH paper.
Fig.7.11 pH-paper with four strips that may have different colours at the same pH
For greater accuracy pH meters are used. pH meter is a device that measures the pH-dependent electrical potential of the test solution within 0.001 precision. pH meters of the size of a writing pen are now available in the market. The pH of some very common substances are given in Table 7.5 (page 212).
PROBLEM 16
The concentration of hydrogen ion in a sample of soft drink is 3.8 × 10–3M. what is its pH ?
SOLUTION
pH = – log[3.8 × 10–3]
= – {log[3.8] + log[10–3]}
= – {(0.58) + (– 3.0)} = – { – 2.42} = 2.42
Therefore, the pH of the soft drink is 2.42 and it can be inferred that it is acidic.
PROBLEM 17
Calculate pH of a 1.0 × 10-8 M solution of HCl.
SOLUTION: H3O + (aq) + OH– (aq)
2H2O(1)
H3O+(aq)+OH-(aq)
\(\begin{array}{l} {K_w} = \left[ {O{H^ - }} \right]\left[ {{H_3}{O^ + }} \right]\\ = {10^{ - 14}} \end{array}\)
Let,x=[OH-]=[H3O+] from H2O.The H3O+ concentration is generated (i) from the ionization of HCl dissolved i.e.,
HCl(aq) + H2O(l)
H3O+(aq)+Cl-(aq) and (ii) from ionization of H2O. In these very dilute solutions, both sources of H3O+ must be considered:
[H3O+] = 10–8 + x
K = (10–8 + x)(x) = 10–14
or x2 + 10–8 x – 10–14 = 0
[OH– ] = x = 9.5 × 10–8
So, pOH = 7.02 and pH = 6.98
-
Ionization Constants of Weak Acids
Ionization Constants of Weak Acids
Consider a weak acid HX that is partially ionized in the aqueous solution. The equilibrium can be expressed by:
HX(aq) + H2O(l) H3O+(aq) + X–(aq)
Initial
concentration (M)
c 0 0
Let \(\alpha\) be the extent of ionization Change (M)
-c\(\alpha\) +c\(\alpha\) +c\(\alpha\)
Equilibrium concentration (M)
c-c\(\alpha\) c\(\alpha\) c\(\alpha\)
Here, c = initial concentration of the undissociated acid, HX at time, t = 0. \(\alpha\) = extent up to which HX is ionized into ions. Using these notations, we can derive the equilibrium constant for the above discussed acid dissociation equilibrium:
\( {K_a} = {c^2}{\alpha ^2}/c\left( {1 - \alpha } \right) = c{\alpha ^2}/1 - \alpha \)
Ka is called the dissociation or ionization constant of acid HX. It can be represented alternatively in terms of molar concentration as follows,
Ka=[H+][X-]/[HX] (7.30)
At a given temperature T, Ka is a measure of the strength of the acid HX i.e., larger the value of Ka, the stronger is the acid. Ka is a dimensionless quantity with the understanding that the standard state concentration of all species is 1M.
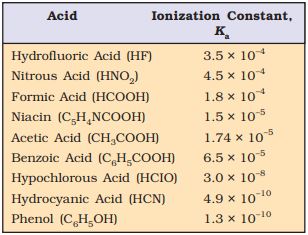
The values of the ionization constants of some selected weak acids are given in Table 7.6.
Table 7.6 The Ionization Constants of Some Selected Weak Acids (at 298K)
Knowing the ionization constant, Ka of an acid and its initial concentration, c, it is possible to calculate the equilibrium
concentration of all species and also the degree of ionization of the acid and the pH of the solution.
The pH scale for the hydrogen ion concentration has been so useful that besides pKw, it has been extended to other species and quantities.Thus, we have:
pKa=-log(Ka) (7.31)
Knowing the ionization constant, Ka of an acid and its initial concentration, c, it is possible to calculate the equilibrium
concentration of all species and also the degree of ionization of the acid and the pH of the solution.
A general step-wise approach can be adopted to evaluate the pH of the weak electrolyte as follows:
Step 1. The species present before dissociation are identified as Brönsted-Lowry acids / bases.
Step 2. Balanced equations for all possible reactions i.e., with a species acting both as acid as well as base are written.
Step 3. The reaction with the higher Ka is
identified as the primary reaction whilst the other is a subsidiary reaction.
Step 4. Enlist in a tabular form the following values for each of the species in the primary reaction.
- Initial concentration, c.
- Change in concentration on proceeding to equilibrium in terms of \(\alpha\), degree of ionization.
- Equilibrium concentration.
Step 5. Substitute equilibrium concentrations into equilibrium constant equation for principal reaction and solve for \(\alpha\).
Step 6. Calculate the concentration of species in principal reaction.
Step 7. Calculate pH = – log[H3O+]
The above mentioned methodology has been elucidated in the following examples.
PROBLEM 18
The ionization constant of HF is 3.2 × 10–4. Calculate the degree of dissociation of HF in its 0.02 M solution. Calculate the concentration of all species present (H3O+, F– and HF) in the solution and its pH.
SOLUTION
The following proton transfer reactions are possible:
1) HF + H2O
H3O+ + F-
Ka = 3.2 × 10–4
2)H2O+H2O
H3O++OH-
Kw=1.0\(\times\)10-14
As Ka >> Kw, [1] is the principle reaction.
HF+H2O
H3O++F-
Initial
concentration (M)
0.02 0 0
Change (M)
-0.02\(\alpha\) +0.02\(\alpha\) +0.02\(\alpha\)
Equilibrium concentration (M)
0.02 – 0.02 \(\alpha\) 0.02 \(\alpha\) 0.02\(\alpha\)
Substituting equilibrium concentrations in the equilibrium reaction for principal reaction gives:
Ka= (0.02\(\alpha\))2 / (0.02 – 0.02\(\alpha\))
= 0.02 \(\alpha\)2 / (1 –\(\alpha\)) = 3.2 × 10–4
We obtain the following quadratic equation:
\(\alpha\)2 + 1.6 × 10–2\(\alpha\) – 1.6 × 10–2 = 0
The quadratic equation in \(\alpha\) can be solved and the two values of the roots are:
\(\alpha\)= + 0.12 and – 0.12
The negative root is not acceptable and hence,
\(\alpha\) = 0.12
This means that the degree of ionization,
\(\alpha\)= 0.12, then equilibrium concentrations of other species viz., HF,F – and H3O+ are given by:
[H O+] = [F –] = c\(\alpha\) = 0.02 × 0.12
= 2.4 × 10–3 M
[HF] = c(1 – \(\alpha\)) = 0.02 (1 – 0.12)
= 17.6 × 10-3 M
pH = – log[H+] = –log(2.4 × 10–3) = 2.62
PROBLEM 19
The pH of 0.1M monobasic acid is 4.50. Calculate the concentration of species H+,A– and HA at equilibrium. Also, determine the value of Ka and pKa of the monobasic acid.
SOLUTION
pH = – log [H+]
Therefore, [H+] = 10 –pH = 10–4.50
= 3.16 × 10–5
[H+] = [A–] = 3.16 × 10–5
Thus,Ka = [H+][A-] / [HA]
[HA]eqlbm= 0.1 – (3.16 × 10-5) ∫ 0.1
Ka = (3.16 × 10–5)2 / 0.1 = 1.0 × 10–8
pKa = – log(10–8) = 8
Alternatively, “Percent dissociation” is another useful method for measure of strength of a weak acid and is given as:
Percent dissociation
= [HA]dissociated/[HA]initial × 100% (7.32)
PROBLEM 20
Calculate the pH of 0.08M solution of hypochlorous acid, HOCl.The ionization constant of the acid is 2.5×10–5.Determine the percent dissociation of HOCl.
SOLUTION
HOCl(aq) + H2O (1)
H3O+(aq) + ClO–(aq)
Initial concentration (M)
0.08 0 0
Change to reach equilibrium concentration (M)
– x + x +x
equilibrium concentartion (M)
0.08 – x x x
Ka = {[H3O+][ClO–] / [HOCl]}
= x2 / (0.08 –x)
As x << 0.08, therefore 0.08 – x ∫ 0.08 x2 / 0.08 = 2.5 × 10–5
x2 = 2.0 × 10–6, thus, x = 1.41 × 10–3
[H+] = 1.41 × 10–3 M.
Therefore,
Percent dissociation
= {[HOCl]dissociated / [HOCl]initial }× 100
= 1.41 × 10–3 × 102/ 0.08 = 1.76 %.
pH = –log(1.41 × 10–3) = 2.85.
-
Ionization of Weak Bases
Ionization of Weak Bases
The ionization of base MOH can be represented by equation:
MOH(aq)
M+(aq) + OH–(aq)
In a weak base there is partial ionization of MOH into M+ and OH–, the case is similar to that of acid-dissociation equilibrium. The equilibrium constant for base ionization is called base ionization constant and is represented by Kb. It can be expressed in terms of concentration in molarity of various species in equilibrium by the following equation:
Kb=[M+][OH–] / [MOH] (7.33)
Alternatively, if c = initial concentration of base and \(\alpha\) = degree of ionization of base i.e. the extent to which the base ionizes. When equilibrium is reached, the equilibrium constant can be written as:
Kb=(c\(\alpha\))2 / c (1-\(\alpha\)) = c\(\alpha\)2 / (1-\(\alpha\))
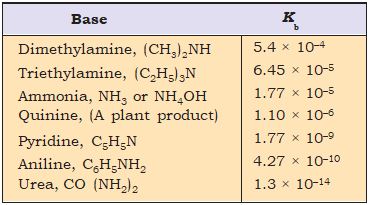
The values of the ionization constants of some selected weak bases, Kb are given in Table 7.7.
Table 7.7 The Values of the Ionization Constant of Some Weak Bases at 298 K
Many organic compounds like amines are weak bases.Amines are derivatives of ammonia in which one or more hydrogen atoms are replaced by another group. For example, methylamine, codeine, quinine and nicotine all behave as very weak bases due to their very small Kb. Ammonia produces OH– in aqueous solution:
NH3(aq) + H2O(1)
NH\(_4^ + \)(aq) + OH–(aq)
The pH scale for the hydrogen ion concentration has been extended to get:
pKb = –log (Kb) (7.34)
PROBLEM 21
The pH of 0.004M hydrazine solution is 9.7. Calculate its ionization constant Kb and pKb.
SOLUTION
NH2NH2 + H2O
NH2NH\(_3^ + \) + OH–
From the pH we can calculate the hydrogen ion concentration. Knowing hydrogen ion concentration and the ionic product of water we can calculate the concentration of hydroxyl ions. Thus we have:
[H+] = antilog (–pH)
= antilog (–9.7) = 1.67 ×10–10= 5.98 × 10–5
The concentration of the corresponding hydrazinium ion is also the same as that of hydroxyl ion.The concentration of both these ions is very small so the concentration of the undissociated base can be taken equal to 0.004M.
Thus,
Kb = [NH2NH\(_3^ + \)][OH–] / [NH2NH2]
= (5.98 × 10–5)2 / 0.004 = 8.96 × 10–7
pKb= –logKb = –log(8.96 × 10–7) = 6.04.
PROBLEM 22
Calculate the pH of the solution in which 0.2M NH4Cl and 0.1M NH3 are present. The pKb of ammonia solution is 4.75.
SOLUTION
NH3+H2O
NH\(_4^ + \)+OH-
The ionization constant of NH3,
Kb = antilog (–pKb ) i.e.
Kb= 10–4.75 = 1.77 × 10–5 M
NH3+H2O
NH\(\_4^ + \)+OH-
Initial concentration (M)
0.10 0.20 0
Change to reach equilibrium (M)
-x +x +x
At equilibrium (M)
0.10 – x 0.20+x x
Kb = [NH\(_4^ + \)][OH–] / [NH3]
= (0.20 + x)(x) / (0.1 – x) = 1.77 × 10–5
As Kb is small, we can neglect x in comparison to 0.1M and 0.2M. Thus,
[OH–] = x = 0.88 × 10–5
Therefore, [H+] = 1.12 × 10–9
pH = – log[H+] = 8.95.
-
Relation between Ka and Kb
Relation between Ka and Kb
As seen earlier in this chapter, Ka and Kb represent the strength of an acid and a base,respectively. In case of a conjugate acid-base pair, they are related in a simple manner so that if one is known, the other can be deduced.Considering the example of NH\(_4^ + \) and NH3 we see,
NH\( _4^ + \)(aq) + H2O(1)
H2O+(aq) + NH3 (aq)
Ka=[H3O+][NH3]/[NH\(_4^ + \)]=5.610-10
NH3(aq) + H2O(1)
NH\(_4^ + \)(aq) + OH–(aq)
Kb =[ NH\( _4^ + \)][ OH–] / NH3 = 1.8 × 10–5
Net 2H2O(1)
H3O+(aq)+OH-(aq)
Kw=[H2O+][ OH– ] = 1.0 × 10–14 M
Where, Ka represents the strength of NH\(_4^ + \) as an acid and Kb represents the strength of NH3 as a base.
It can be seen from the net reaction that the equilibrium constant is equal to the product of equilibrium constants Ka and Kb for the reactions added. Thus,
\(\begin{array}{l} {K_a} \times {K_b} = \left\{ {\left[ {{H_3}{O^ + }} \right]\left[ {N{H_3}} \right]/\left[ {NH_4^ + } \right]} \right\} \times \left\{ {\left[ {NH_4^ + } \right]\left[ {O{H^ - }} \right]/\left[ {N{H_3}} \right]} \right\}\\ = \left[ {{H_3}{O^ + }} \right]\left[ {O{H^ - }} \right] = {K_w}\\ = \left( {5.6 \times {{10}^{ - 10}}} \right) \times \left( {1.8 \times {{10}^{ - 5}}} \right) = 1.0 \times {10^{ - 14}}M \end{array}\)
This can be extended to make a generalisation. The equilibrium constant for a net reaction obtained after adding two (or more) reactions equals the product of the equilibrium constants for individual reactions:
KNET = K1 × K2 × …… (3.35)
Similarly, in case of a conjugate acid-base pair,
Ka × Kb = Kw (7.36)
Knowing one, the other can be obtained.It should be noted that a strong acid will have a weak conjugate base and vice-versa.
Alternatively, the above expression Kw = Ka× K b, can also be obtained by considering the base-dissociation equilibrium reaction:
B(aq) + H2O(1)
BH+(aq) + OH–(aq)
Kb= [BH+][OH–] / [B]
As the concentration of water remains constant it has been omitted from the denominator and incorporated within the dissociation constant. Then multiplying and dividing the above expression by [H+], we get:
Kb= [BH+][OH –][H+] / [B][H+]
={[ OH–][H+]}{[BH+] / [B][H+]}
= Kw / Ka
or Ka × Kb = Kw
It may be noted that if we take negative logarithm of both sides of the equation, then pK values of the conjugate acid and base are related to each other by the equation:
pKa+pKb=pKw=14(at 298K)
PROBLEM 23
Determine the degree of ionization and pH of a 0.05M of ammonia solution.The ionization constant of ammonia can be taken from Table 7.7. Also, calculate the ionization constant of the conjugate acid of ammonia.
SOLUTION
The ionization of NH3 in water is represented by equation:
NH3+H2O
NH\(_4^ + \)+OH–
We use equation (7.33) to calculatehydroxyl ion concentration,
[OH–] = c\(\alpha\) = 0.05 \(\alpha\)
Kb = 0.05 \(\alpha\)2 / (1 – \(\alpha\))
The value of \(\alpha\) is small, therefore the quadratic equation can be simplified by neglecting \(\alpha\) in comparison to 1 in the denominator on right hand side of the equation,
Thus,
Kb = c \(\alpha\)2 or \(\alpha\) =
(1.77 × 10–5 / 0.05)
= 0.018.
[OH–] = c\(\alpha\) = 0.05 × 0.018 = 9.4 × 10–4M.
[H+] = Kw / [OH–] = 10–14 / (9.4 × 10–4)
= 1.06 × 10–11
pH = –log(1.06 × 10–11) = 10.97.
Now, using the relation for conjugate acid-base pair,
Ka\(\times\)Kb=Kw
using the value of Kb of NH3 from Table 7.7.
We can determine the concentration of conjugate acid NH\( _4^ + \)
Ka = Kw / Kb = 10 / 1.77 × 10= 5.64 × 10–10.
-
Di-and Polybasic Acids and Di-and Polyacidic Bases
Di-and Polybasic Acids and Di-and Polyacidic Bases
Some of the acids like oxalic acid, sulphuric acid and phosphoric acids have more than one ionizable proton per molecule of the acid. Such acids are known as polybasic or polyprotic acids.
The ionization reactions for example for a dibasic acid H2X are represented by the equations:
H2X(aq)
H+(aq)+HX-(aq)
HX-(aq)
H+(aq)+X2-(aq)
And the corresponding equilibrium constants are given below:
\( {K_{{a_1}}} = \left\{ {\left[ {{H^ + }} \right]\left[ {H{X^ - }} \right]} \right\}/\left[ {{H_2}X} \right]\) and \({K_{{a_2}}} = \left\{ {\left[ {{H^ + }} \right]\left[ {{X^{2 - }}} \right]} \right\}/\left[ {H{X^ - }} \right]\)
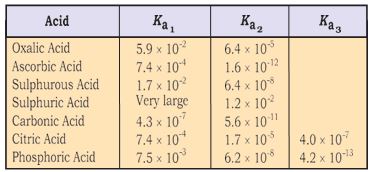
Here, \({K_{{a_1}}}\) and \( {K_{{a_2}}}\) are called the first and second ionization constants respectively of the acid H2 X. Similarly, for tribasic acids like H3PO4 we have three ionization constants.The values of the ionization constants for some common polyprotic acids are given in Table 7.8.
Table 7.8 The Ionization Constants of Some Common Polyprotic
It can be seen that higher order ionization constants \(\left( {{K_{{a_2}}},{K_{{a_3}}}} \right)\) are smaller than the lower order ionization constant (Ka ) of a polyprotic acid. The reason for this is that it is more difficult to remove a positively charged proton from a negative ion due to electrostatic forces. This can be seen in the case of removing a proton from the uncharged H2CO3 as compared from a negatively charged HCO\(_3^ - \). Similarly, it is more difficult to remove a proton from a doubly charged HPO\(_4^{2 - }\) anion as compared to \( {H_2}PO_4^ - \).
Polyprotic acid solutions contain a mixture of acids like H2A, HA– and A2– in case of a diprotic acid. H2A being a strong acid, the primary reaction involves the dissociation of H2A, and H3O+ in the solution comes mainly from the first dissociation step.
-
Factors Affecting Acid Strength
Factors Affecting Acid Strength
Having discussed quantitatively the strengths of acids and bases, we come to a stage where we can calculate the pH of a given acid solution. But, the curiosity rises about why should some acids be stronger than others? What factors are responsible for making them stronger? The answer lies in its being a complex phenomenon. But, broadly speaking we can say that the extent of dissociation of an acid depends on the strength and polarity of the H-A bond.
In general, when strength of H-A bond decreases, that is, the energy required to break the bond decreases, HA becomes a stronger acid. Also, when the H-A bond becomes more polar i.e., the electronegativity difference between the atoms H and A increases and there is marked charge separation, cleavage of the bond becomes easier thereby increasing the acidity.
But it should be noted that while comparing elements in the same group of the periodic table, H-A bond strength is a more important factor in determining acidity than its polar nature. As the size of A increases down the group, H-A bond strength decreases and so the acid strength increases. For example,
Similarly, H2S is stronger acid than H2O.
But, when we discuss elements in the same row of the periodic table, H-A bond polarity becomes the deciding factor for determining the acid strength. As the electronegativity of A increases, the strength of the acid also increases. For example,
-
Common Ion Effect in the Ionization of Acids and Bases
Common Ion Effect in the Ionization of Acids and Bases
Consider an example of acetic acid dissociation equilibrium represented as:
CH3COOH(aq)
H+(aq) + CH3COO– (aq)
or HAc(aq)
H+ (aq) + Ac– (aq)
Ka= [H+][Ac– ] / [HAc]
Addition of acetate ions to an acetic acid solution results in decreasing the concentration of hydrogen ions, [H+]. Also, if H+ ions are added from an external source then the equilibrium moves in the direction of undissociated acetic acid i.e., in a direction of reducing the concentration of hydrogen ions, [H+]. This phenomenon is an example of common ion effect.It can be defined as a shift in equilibrium on adding a substance that provides more of an ionic species already present in the dissociation equilibrium. Thus, we can say that common ion effect is a phenomenon based on the Le Chatelier’s principle discussed in section 7.8.
In order to evaluate the pH of the solution resulting on addition of 0.05M acetate ion to 0.05M acetic acid solution, we shall consider the acetic acid dissociation equilibrium once again,
HAc(aq)
H+(aq) + Ac–(aq)
Initial concentration (M)
0.05 0 0.05
Let x be the extent of ionization of acetic acid.
Change in concentration (M)
–x +x +x
Equilibrium concentration (M)
0.05-x x 0.05+x
Therefore,
Ka=[H+][Ac– ]/[H Ac] = {(0.05+x)(x)}/(0.05-x)
As Ka is small for a very weak acid,x<<0.05.
Hence, (0.05 + x) \(\approx \) (0.05 – x) \( \approx \) 0.05
Thus,
1.8 × 10–5 = (x) (0.05 + x) / (0.05 – x)
= x(0.05) / (0.05) = x = [H+] = 1.8 ×10-5M
pH = – log(1.8 × 10–5) = 4.74
PROBLEM 24
Calculate the pH of a 0.10M ammonia solution. Calculate the pH after 50.0 mL of this solution is treated with 25.0 mL of 0.10M HCl. The dissociation constant of ammonia, Kb= 1.77 × 10–5
SOLUTION
NH3+H2O\(\to\)NH4++OH-
Kb = [NH\(_4^ + \)][OH–] / [NH3] = 1.77 × 10–5
Before neutralization,
[NH\(_4^ + \)] = [OH–] = x
[NH3] = 0.10 – x ∫ 0.10
x2 / 0.10 = 1.77 × 10–5
Thus, x = 1.33 × 10–3 = [OH–]
Therefore,[H+] = K / [OH–] = 10–14 /(1.33 × 10–3) = 7.51 × 10–12
pH = –log(7.5 × 10–12) = 11.12
On addition of 25 mL of 0.1M HCl solution (i.e., 2.5 mmol of HCl) to 50 mL of 0.1M ammonia solution (i.e., 5 mmol of NH3), 2.5 mmol of ammonia molecules are neutralized.The resulting 75 mL solution contains the remaining unneutralized 2.5 mmol of NH3 molecules and 2.5 mmol of NH\(_4^ + \).
NH3+H2O\(\to\)NH4++OH-
2.5 2.5 0 0
At equilibrium
0 0 2.5 2.5
The resulting 75 mL of solution contains 2.5 mmol of NH\(\to\) ions (i.e., 0.033 M) and 2.5 mmol (i.e., 0.033 M ) of uneutralised NH3 molecules. This NH3 exists in the following equilibrium:
NH4OH
NH\(\to\) +OH-
0.033M – y y y
where, y = [OH–] = [NH\(\to\)]
The final 75mL solution after neutralisation already contains 2.5 m mol NH+ ions (i.e. 0.033M), thus total concentration of NH\(\to\) ions is given as:
[NH\(\to\)] = 0.033 + y
As y is small, [NH4OH] ∫ 0.033 M and [NH\(\to\)] ∫ 0.033M.
We know
Kb= [NH\(\to\)][OH–] / [NH4OH]
= y(0.033)/(0.033) = 1.77 × 10–5 M
Thus, y = 1.77 × 10–5 = [OH–]
[H+] = 10–14 / 1.77 × 10–5 = 0.56 × 10–9
Hence, pH = 9.24