-
Solubility Equilibria of Sparingly Soluble Salts
SOLUBILITY EQUILIBRIA OF SPARINGLY SOLUBLE SALTS
We have already known that the solubility of ionic solids in water varies a great deal. Some of these (like calcium chloride) are so soluble
that they are hygroscopic in nature and even absorb water vapour from atmosphere. Others (such as lithium fluoride) have so little solubility
that they are commonly termed as insoluble.
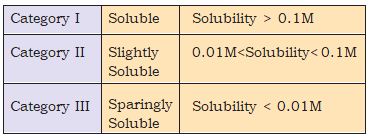
The solubility depends on a number of factors important amongst which are the lattice enthalpy of the salt and the solvation enthalpy of the ions in a solution. For a salt to dissolve in a solvent the strong forces of attraction between its ions (lattice enthalpy) must be overcome by the ion-solvent interactions. The solvation enthalpy of ions is referred to in terms of solvation which is always negative i.e. energy released in the process of solvation. The amount of solvation enthalpy depends on the nature of the solvent. In case of a non-polar (covalent) solvent, solvation enthalpy is small and hence, not sufficient to overcome lattice enthalpy of the salt. Consequently, the salt does not dissolve in non-polar solvent. As a general rule , for a salt to be able to dissolve in a particular solvent its solvation enthalpy must be greater than its lattice enthalpy so that the latter may be overcome by former. Each salt has its characteristic solubility which depends on temperature. We classify salts on the basis of their solubility in the following three categories.
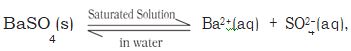
We shall now consider the equilibrium between the sparingly soluble ionic salt and its saturated aqueous solution
-
Solubility Product Constant
Solubility Product Constant
Let us now have a solid like barium sulphate in contact with its saturated aqueous solution. The equilibrium between the undisolved solid and the ions in a saturated solution can be represented by the equation:
The equilibrium constant is given by the equation:
K={[Ba2+][SO\( _4^{2 - }\)]}/[BaSO4]
For a pure solid substance the concentration remains constant and we can write
\({K_{sp}} = K\left[ {BaS{O_4}} \right] = \left[ {B{a^{2 + }}} \right]\left[ {SO_4^{2 - }} \right]\,\,\,\,\,\,\left( {7.43} \right)\)
We call Ksp the solubility product constant or simply solubility product. The experimental value of Ksp in above equation at 298K is 1.1 × 10–10. This means that for solid barium sulphate in equilibrium with its saturated solution, the product of the concentrations of barium and sulphate ions is equal to its solubility product constant.The concentrations of the two ions will be equal to the molar solubility of the barium sulphate. If molar solubility is S, then
1.1 × 10–10 = (S)(S) = S2
or S = 1.05 × 10–5.
Thus, molar solubility of barium sulphate will be equal to 1.05 × 10–5 mol L–1.
A salt may give on dissociation two or more than two anions and cations carrying different charges. For example, consider a salt like zirconium phosphate of molecular formula \({\left( {Z{r^{4 + }}} \right)_3}{\left( {PO_4^{3 - }} \right)_4}\).It dissociates into 3 zirconium cations of charge +4 and 4 phosphate anions of charge –3. If the molar solubility of zirconium phosphate is S, then it can be seen from the stoichiometry of the compound that
[Zr4+] = 3S and [PO\({_4^{3 - }}\)] = 4S
and Ksp = (3S)3 (4S)4 = 6912 (S)7
or S = {Ksp / (33 × 44)}1/7 = (Ksp / 6912)1/7
A solid salt of the general formula \(M_x^{p + }X_y^{q - }\) with molar solubility S in equilibrium with its saturated solution may be represented by the equation:
MxXy (s)x
Mp+(aq)+yX-q(aq)
(where x × p+ = y × q–)
And its solubility product constant is given as:
Ksp== [Mp+]x[Xq– ]y = (xS)x(yS)y (7.44)
= xx . yy . S(x + y)
S(x + y) =Ksp/xx . yy
S=(Ksp/xx . yy)1/x+y (7.45)
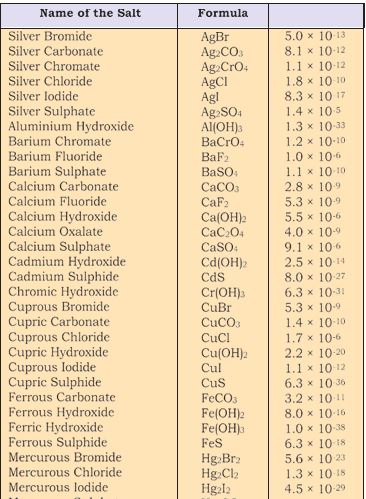
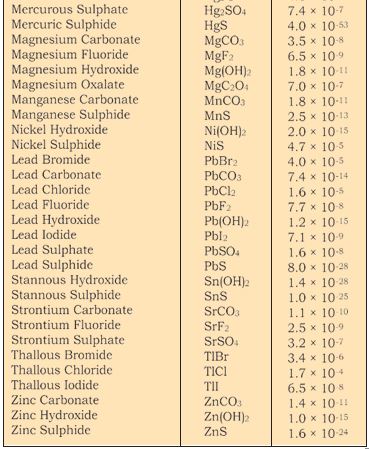
The term Ksp in equation is given by Qsp (section 7.6.2) when the concentration of one or more species is not the concentration under equilibrium.Obviously under equilibrium conditions Ksp = Qsp but otherwise it gives the direction of the processes of precipitation or dissolution.The solubility product constants of a number of common salts at 298K are given in Table 7.9.
PROBLEM 29
Calculate the solubility of A2X3 in pure water, assuming that neither kind of ion reacts with water.The solubility product of A2X3, Ksp = 1.1 × 10-23.
SOLUTION
A2X3 \(\to\) 2A3++ 3X2-
Ksp = [A3+]2 [X2–]3 = 1.1 × 10–23
If S = solubility of A2X3, then [A3+] = 2S; [X2–] = 3S
therefore, Ksp = (2S) (3S) = 108S5= 1.1 × 10–23
thus, S5 = 1 × 10–25
S = 1.0 × 10–5 mol/L.
PROBLEM 27
The values of Ksp of two sparingly soluble salts Ni(OH)2 and AgCN are 2.0 × 10–15 and 6 × 0–17 respectively. Which salt is more soluble? Explain.
SOLUTION
AgCN
Ag++CN-
Ksp = [Ag+][CN–] = 6 × 10–17
Ni(OH)2
Ni2++2OH-
Ksp= [Ni2+][OH–]2 = 2 × 10–15
Let [Ag+] = S1, then [CN-] = S1
Let [Ni2+] = S2, then [OH–] = 2S2
S\( _1^2\)= 6 × 10–17 , S1 = 7.8 × 10–9
(S2)(2S2)2 = 2 × 10-15,S = 0.58 × 10-4
Ni(OH)2 is more soluble than AgCN.
Table 7.9 The Solubility Product Constants,Ksp of Some Common Ionic Salts at 298K.
-
Common Ion Effect on Solubility of Ionic Salts
Common Ion Effect on Solubility of Ionic Salts
It is expected from Le Chatelier’s principle that if we increase the concentration of any one of the ions, it should combine with the ion of its
opposite charge and some of the salt will be precipitated till once again Ksp= Qsp. Similarly,if the concentration of one of the ions is decreased, more salt will dissolve to increase the concentration of both the ions till once again Ksp= Qsp.This is applicable even to soluble salts like sodium chloride except that due to higher concentrations of the ions, we use their activities instead of their molarities in the expression for Qsp.Thus if we take a saturated solution of sodium chloride and pass HCl gas through it, then sodium chloride is precipitated due to increased concentration (activity) of chloride ion available from the dissociation of HCl. Sodium chloride thus obtained is of very high purity and we can get rid of impurities like sodium and magnesium sulphates. The common ion effect is also used for almost complete precipitation of a particular ion as its sparingly soluble salt, with very low value of solubility product for gravimetric estimation. Thus we can precipitate silver ion as silver chloride, ferric ion as its hydroxide (or hydrated ferric oxide) and barium ion as its sulphate for quantitative estimations.
PROBLEM 28
Calculate the molar solubility of Ni(OH)2 in 0.10 M NaOH. The ionic product of Ni(OH)2 is 2.0 × 10-15.
SOLUTION
Let the solubility of Ni(OH)2 be equal to S.Dissolution of S mol/L of Ni(OH)2 provides S mol/L of Ni2+ and 2S mol/L of OH–, but
the total concentration of OH– = (0.10 + 2S) mol/L because the solution already contains 0.10 mol/L of OH– from NaOH.
Ksp= 2.0 × 10–15 = [Ni2+] [OH–]2
= (S) (0.10 + 2S)2
As Ksp is small, 2S << 0.10, thus, (0.10 + 2S) \( \approx \) 0.10
Hence,
2.0 × 10–15 = S (0.10)2
S = 2.0 × 10–13 M = [Ni2+]
The solubility of salts of weak acids like phosphates increases at lower pH. This is because at lower pH the concentration of the anion decreases due to its protonation.This in turn increase the solubility of the salt so that Ksp = Qsp. We have to satisfy two equilibria simultaneously i.e.,
Ksp= [M+] [X–],
HX(aq)
H+(aq)+X-(aq);
\({K_a} = \frac{{\left[ {{H^ + }(aq)} \right]\left[ {{X^ - }(aq)} \right]}}{{\left[ {HX(aq)} \right]}}\)
[X –] / [HX] = Ka/ [H+]
Taking inverse of both side and adding 1 we get
\(\begin{array}{l} \frac{{\left[ {HX} \right]}}{{\left[ X \right]}} + 1 = \frac{{\left[ {{H^ + }} \right]}}{{{K_a}}} + 1\\ \frac{{\left[ {HX} \right] + \left[ {{H^ - }} \right]}}{{\left[ {{X^ - }} \right]}} = \frac{{\left[ {{H^ + }} \right] + {K_a}}}{{{K_a}}} \end{array}\)
Now, again taking inverse, we get [X–] / {[X–] + [HX]} = f = Ka/ (Ka + [H+]) and it can be seen that ‘f’ decreases as pH decreases. If S is the solubility of the salt at a given pH then \({K_{sp}} = \left[ S \right]\left[ {fS} \right] = {S^2}\left\{ {{K_a}/\left( {{K_a} + \left[ {{H^ + }} \right]} \right)} \right\}\) and \(S = {\left\{ {{K_{sp}}\left( {\left[ {{H^ + }} \right] + {K_a}} \right)/{K_a}} \right\}^{1/2}}\) (7.46)
Thus solubility S increases with increase in [H+] or decrease in pH.