-
Properties of metals
Properties of Materials
Have you ever wondered why a tumbler is not made with a piece of cloth? Recall our experiments with pieces of cloth in Chapter 3 and also keep in mind that we generally use a tumbler to keep a liquid. Therefore, would it not be silly, if we were to make a tumbler out of cloth (Fig 4.2)! What we need for a tumbler is glass, plastics, metal, or other such material that will hold water. Similarly, it would not be wise to use paper-like materials for cooking vessels.
Fig 4.2: Using a cloth tumbler
We see then, that we choose a material to make an object depending on its properties, and the purpose for which the object is to be used.
So, what are all the properties of materials that would be important for their usage? Some properties are discussed here.
Appearance
Materials usually look different from each other. Wood looks very different from iron. Iron appears different from copper or aluminium. At the same time, there may be some similarities between iron, copper and aluminium that are not there in the wood.
Activity-3:
Collect small pieces of different materials — paper, cardboard, wood, copper wire, aluminium sheet, chalk. Do any of these appear shiny? Separate the shiny materials into a group.
Now, observe as the teacher cuts each material into two pieces and look at the freshly cut surface (Fig. 4.3). What do you notice?
Fig. 4.3 Cutting pieces of materials to see other hue lustre
Does the freshly cut surface of some of these materials appear shiny? Include these objects also in the group of shiny materials.
Do you notice such a shine or lustre in the other materials, cut them anyway as you can? Repeat this in the class with as many materials as possible and make a list of those with and without lustre. Instead of cutting, you can rub the surface of the material with sandpaper to see if it has lustre.
Materials that have such lustre are usually metals. Iron, copper, aluminium and gold are examples of metals. Some metals often lose their shine arid appear dull, because of the action of air and moisture on them. We, therefore, notice the lustre, only on their freshly cut surface. When you visit an ironsmith or a workshop, look out for freshly cut surfaces of metal rods to see if they have lustre.
-
Hardness
Hardness
When you press different materials with your hands, some of them may be hard to compress while others can be easily compressed. Take a metal key and try to scratch with it, the surface of a piece of wood, aluminium, a piece of stone, a nail, candle, chalk, any other material or object. You can easily scratch some materials, while some cannot be scratched so easily. Materials that can be compressed or scratched easily are called soft while some other materials which are difficult to compress are called hard. For example, cotton or sponge is soft while the iron is hard.
In appearance, materials can have different properties, like lustre, hardness, be rough or smooth. Can you think of other properties that describe the appearance of a material?
-
Hardness
Soluble or Insoluble?
Activity 4
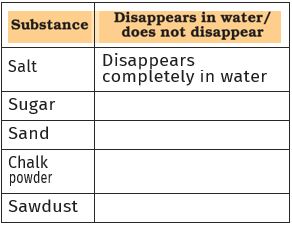
Collect samples of some solid substances such as sugar, salt, chalk powder, sand, and sawdust. Take five glasses or beakers. Fill each one of them about two-thirds with water. Add a small amount (spoonful) of sugar to the first glass, salt to the second, and similarly, add small amounts of the other substances into the other glasses. Stir the contents of each of them with a spoon. Wait for a few minutes. Observe what happens to the substances added to water (Fig. 4.4). Note your observations as shown in Table 4.3.

Fig. 4.4 What disappears, what doesn’t?
Table 4.3 Mixing different solid materials in water
You will notice that some substances have completely disappeared or dissolved in water. We say that these substances are soluble in water. Other substances do not mix with water and do not disappear even after we stir for a long time. These substances are insoluble in water.
Water plays an important role in the functioning of our body because it can dissolve a large number of substances. Do liquids also dissolve in water?
Activity 5
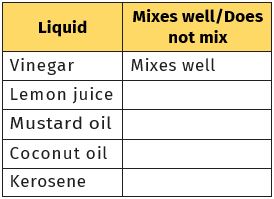
Collect samples of vinegar, lemon juice, mustard oil or coconut oil, kerosene, or any other liquid. Take a glass tumbler. Fill it up to half with water. Add a few spoonfuls of one liquid to this and stir it well. Let it stand for five minutes. Observe whether the liquid mixes with water (Fig. 4.5).

Repeat the same with other liquids, as many different liquids as are available to you. Write your observations in Table 4.4.
Table 4.4 Solubility of some common liquids in water
We notice that some liquids get completely mixed with water. Some others do not mix with water and form a separate layer when kept aside for some time.
Some gases are soluble in water whereas others are not. Water, usually, has small quantities of some gases dissolved in it. For example, oxygen gas dissolved in water is very important for the survival of animals and plants that live in water.
-
Hardness
Soluble or Insoluble?
Activity 4
Collect samples of some solid substances such as sugar, salt, chalk powder, sand, and sawdust. Take five glasses or beakers. Fill each one of them about two-thirds with water. Add a small amount (spoonful) of sugar to the first glass, salt to the second, and similarly, add small amounts of the other substances into the other glasses. Stir the contents of each of them with a spoon. Wait for a few minutes. Observe what happens to the substances added to water (Fig. 4.4). Note your observations as shown in Table 4.3.

Fig. 4.4 What disappears, what doesn’t?
Table 4.3 Mixing different solid materials in water
You will notice that some substances have completely disappeared or dissolved in water. We say that these substances are soluble in water. Other substances do not mix with water and do not disappear even after we stir for a long time. These substances are insoluble in water.
Water plays an important role in the functioning of our body because it can dissolve a large number of substances. Do liquids also dissolve in water?
Activity 5
Collect samples of vinegar, lemon juice, mustard oil or coconut oil, kerosene, or any other liquid. Take a glass tumbler. Fill it up to half with water. Add a few spoonfuls of one liquid to this and stir it well. Let it stand for five minutes. Observe whether the liquid mixes with water (Fig. 4.5).

Repeat the same with other liquids, as many different liquids as are available to you. Write your observations in Table 4.4.
Table 4.4 Solubility of some common liquids in water
We notice that some liquids get completely mixed with water. Some others do not mix with water and form a separate layer when kept aside for some time.
Some gases are soluble in water whereas others are not. Water, usually, has small quantities of some gases dissolved in it. For example, oxygen gas dissolved in water is very important for the survival of animals and plants that live in water.
-
Soluble or Insoluble?
Soluble or Insoluble?
Activity 4
Collect samples of some solid substances such as sugar, salt, chalk powder, sand, and sawdust. Take five glasses or beakers. Fill each one of them about two-thirds with water. Add a small amount (spoonful) of sugar to the first glass, salt to the second, and similarly, add small amounts of the other substances into the other glasses. Stir the contents of each of them with a spoon. Wait for a few minutes. Observe what happens to the substances added to water (Fig. 4.4). Note your observations as shown in Table 4.3.

Fig. 4.4 What disappears, what doesn’t?
Table 4.3 Mixing different solid materials in water
You will notice that some substances have completely disappeared or dissolved in water. We say that these substances are soluble in water. Other substances do not mix with water and do not disappear even after we stir for a long time. These substances are insoluble in water.
Water plays an important role in the functioning of our body because it can dissolve a large number of substances. Do liquids also dissolve in water?
Activity 5
Collect samples of vinegar, lemon juice, mustard oil or coconut oil, kerosene, or any other liquid. Take a glass tumbler. Fill it up to half with water. Add a few spoonfuls of one liquid to this and stir it well. Let it stand for five minutes. Observe whether the liquid mixes with water (Fig. 4.5).

Repeat the same with other liquids, as many different liquids as are available to you. Write your observations in Table 4.4.
Table 4.4 Solubility of some common liquids in water
We notice that some liquids get completely mixed with water. Some others do not mix with water and form a separate layer when kept aside for some time.
Some gases are soluble in water whereas others are not. Water, usually, has small quantities of some gases dissolved in it. For example, oxygen gas dissolved in water is very important for the survival of animals and plants that live in water.
-
Objects may float or sink in water
Objects may float or sink in water
While doing Activity 4, you might have noticed that the insoluble solids separated out from water. You may have also noticed this with some liquids in Activity 5. Some of these materials that did not mix with water, floated to the surface of water. Others may have sunk to the bottom of the tumbler, right? We notice many examples of objects that float in water or sink (Fig. 4.6). Dried leaves fallen on the surface of a pond, a stone that you throw into this pond, few drops of honey that you let fall into a glass of water. What happens to all of these?

Figure 4.6 Some objects float in water while others sink in it
Boojho would like you to give him five examples each, of objects that float arid those that sink in water. What about testing these same materials to see if they float or sink in other liquids like oil?
-
Transparency
Transparency
You might have played the game of hide and seek. Think of some places where you would like to hide so that you are not seen by others. Why did you choose those places? Would you have tried to hide behind a glass window? Obviously not, as your friends can see through that and spot you. Can you see through all the materials? Those substances or materials, through which things can be seen, are called transparent (Fig. 4.7).

Fig. 4.7 Looking through opaque, transparent, or /translucent material
Glass, water, air and some plastics are examples of transparent materials. Shopkeepers usually prefer to keep biscuits, sweets and other eatables in transparent containers of glass or plastic, so that buyers can easily see these items (Fig. 4.8).

Fig. 4.8 Transparent bottles in a shop
On the other hand, there are some materials through which you are not able to see. These materials are called opaque. You cannot tell what is kept in a closed wooden box, a cardboard carton, or a metal container. Wood, cardboard, and metals are examples of opaque materials.
Do we find that we can group all materials and objects, without any confusion, as either opaque or transparent?
Take a sheet of paper and look through it towards a lighted bulb. Make a note of your observation. Now, put 2-3 drops of some oil and spread it on the sheet of paper. Look again towards the lighted bulb through that portion of the paper on which the oil has been spread. Do you find that the bulb is more clearly visible than before? But, can you see clearly through the oiled paper? Is everything on the other side of it visible? Perhaps not. The materials through which objects can be seen, but not clearly, are known as translucent. Remember the oily patch on paper when we tested food items for the presence of fats? That was translucent too. Can you think of some more examples of translucent materials?
We can therefore group materials as opaque, transparent, and translucent. Paheli suggests covering the glass of a torch with your palm in a dark place. Switch on the torch and observe the other side of the palm. She wants to know whether the palm of your hand is opaque, transparent, or translucent?
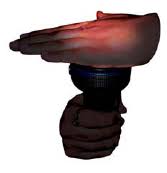
Fig. 4.9 Does torch light pass through your palm?
We learned that materials differ in their appearance and the way they mix in water or other liquids. They may float or sink in water or maybe transparent, opaque, or translucent. Materials can be grouped on the basis of similarities or differences in their properties.
Why do we need to group materials? In everyday life, we often group materials for our convenience. At home, we usually store things in such a manner that similar objects are placed together. Such an arrangement helps us to locate them easily. Similarly, a grocer usually keeps all types of biscuits at one corner of his shop, all soaps at another while grains and pulses are stored at some other place. There is another reason why we find such grouping useful. Dividing materials into groups makes it convenient to study their properties and also observe any patterns in these properties. We will study more about this in higher classes.