-
Methods Of Separation
METHODS OF SEPARATION
We will discuss some simple methods of separating substances that are mixed together. You may come across some of these methods being used in day-to-day activities.
Handpicking
Activity-2
Bring a packet of food grain purchased from a shop to the classroom. Now, spread the grains on a sheet of paper. Do you find only one kind of grain on the sheet of paper? Are there pieces of stone, husks, broken grain, and particles of any other grain in it? Now, remove with your hand the pieces of stone, husks, and other grains from it.
This method of handpicking can be used for separating slightly larger-sized impurities like pieces of dirt, stone, and husk from wheat, rice, or pulses (Fig. 5.3).

Fig. 5.3 Handpicking stones from grain
The quantity of such impurities is usually not very large. In such situations, we find that handpicking is a convenient method of separating substances.
-
Threshing
Threshing
You must have seen bundles of wheat or paddy stalks lying in fields after harvesting the crop. Stalks are dried in the sun before the grain is separated from them. Each stalk has many grain seeds attached to it. Imagine the number of grain seeds in hundreds of bundles of stalk lying in the field! How does the farmer separate grain seeds from those bundles of stalks?
One may pluck mangoes or guavas from the trees. But, grain seeds are much smaller than mangoes or guavas. So, plucking them from their stalks would be impossible. How does one separate grain seeds from their stalks? The process that is used to separate grain from stalks etc. is threshing. In this process, the stalks are beaten to free the grain seeds (Fig. 5.4). Sometimes threshing is done with the help of bullocks. Machines are also used to thresh large quantities of grain.

-
Winnowing
Winnowing
Activity-3
Make a mixture of dry sand with sawdust or powdered dry leaves. Keep this mixture on a plate or a newspaper. Look at this mixture carefully. Can the two different components be made out easily? Are the sizes of particles of the two components similar? Would it be possible to separate the components by handpicking?
Now, take your mixture to an open ground and stand on a raised platform. Put the mixture in a plate or sheet of paper. Hold the plate or the sheet of paper containing the mixture, at your shoulder height. Tilt it slightly, so that the mixture slides out slowly.
What happens? Do both the components — sand and sawdust (or powdered leaves) fall at the same place? Is there a component that blows away? Did the wind manage to separate the two components?
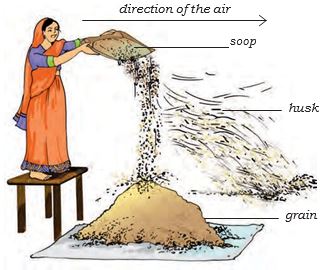
This method of separating components of a mixture is called winnowing. Winnowing is used to separate heavier and lighter components of a mixture by wind or by blowing air.
This method is commonly used by farmers to separate lighter husk particles from heavier seeds of grain (Fig. 5.5).
Fig. 5.5 Winnowing
The husk particles are carried away by the wind. The seeds of grain get separated and form a heap near the platform for winnowing. The separated husk is used for many purposes such as fodder for cattle.
-
Sieving
Sieving
Sometimes, we may wish to prepare a dish with flour. We need to remove impurities and bran that may be present in it. What do we do? We use a sieve and pour the flour into it (Fig. 5.6).
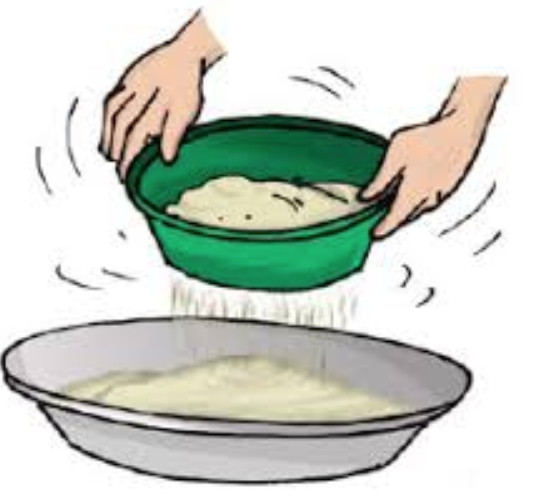
Fig. 5.6 Sieving
Sieving allows the fine flour particles to pass through the holes of the sieve while the bigger impurities remain on the sieve.
In a flour mill, impurities like husk and stones are removed from wheat before grinding it. Usually, a bagful of wheat is poured on a slanting sieve. The sieving removes pieces of stones, stalk and husk that may still remain with wheat after threshing and winnowing. You may have also noticed similar sieves being used at construction sites to separate pebbles and stones from sand (Fig. 5.7).

Fig. 5.7 Pebbles and stones are removed from sand by sieving
Activity-4
Bring a sieve and a small quantity of flour from home, to the class. Sieve the flour to separate any impurities in it. Now, make a fine powder of chalk pieces and mix it with the flour. Can we separate the flour and the powdered chalk by sieving?
Sieving is used when components of a mixture have different sizes.
-
Sedimentation, Decantation and Filteration
Sedimentation, Decantation, and Filtration
Sedimentation and Decantation
Sometimes, it may not be possible to separate components of a mixture by winnowing and handpicking. For example, there may be lighter impurities like dust or soil particles in rice or pulses. How are such impurities separated from rice or pulses before cooking?
Rice or pulses are usually washed before cooking. When you add water to these, the impurities like dust particles get separated. These impurities go into water. Now, what will sink to the bottom of the vessel — rice or dust? Why? Have you seen that the vessel is tilted to pour out the dirty water?
When the heavier component in a mixture settles after water is added to it, the process is called sedimentation. When the water (along with the dust) is removed, the process is called decantation (Fig. 5.8). Let us find a few other mixtures that can be separated through sedimentation and decantation.
Fig. 5.8 Separating two components of a mixture by Sedimentation and Decantation
The same principle is used for separating a mixture of two liquids that do not mix with each other. For example, oil and water from their mixture can be separated by this process. If a mixture of such liquids is allowed to stand for some time, they form two separate layers. The component that forms the top layer can then be separated by decantation.
Let us again consider a mixture of a solid and liquid. After preparing tea, what do you do to remove the tea leaves? Usually, we use strainer to remove tea leaves. Try decantation. It helps a little. But, do you still get a few leaves in your tea? Now, pour the tea through a strainer. Did all the tea leaves remain in the strainer? This process is called filtration (Fig. 5.1). Which method of separating tea leaves from prepared tea is better, decantation or filtration?

Let us now consider the example of water that we use. Do all of us, at all times, get safe water to drink? Sometimes, water supplied through taps may be muddy. The water collected from ponds or rivers may also be muddy, especially after rains. Let us see if we can use some method of separation to remove insoluble impurities like soil from the water.
Activity 5
Collect some muddy water from a pond or a river. If it is not available, mix some soil to water in a glass. Let it stand for half an hour. Observe the water carefully and note your observations.
Does some soil settle at the bottom of water? Why? What will you call this process?
Now, slightly tilt the glass without disturbing the water. Let the water from the top flow into another glass (Fig. 5.8). What will you call this process?
Fig. 5.8 Separating two components of a mixture by Sedimentation and Decantation
Is the water in the second glass still muddy or brown in colour? Now filter it. Did the tea strainer work? Let us try filtering the water through a piece of cloth. In a piece of cloth, small holes or pores remain in between the woven threads. These pores in a cloth can be used as a filter.
If the water is still muddy, impurities can be separated by a filter that has even smaller pores. A filter paper is one such filter that has very fine pores in it. Fig. 5.9 shows the steps involved in using a filter paper.
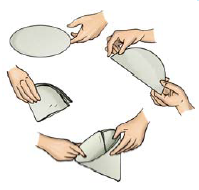
Fig. 5.9 Folding a filter paper to make a cone

Fig. 5.10 Filtration using a filter paper
A filter paper folded in the form of a cone is fixed onto a funnel (Fig. 5.10). The mixture is then poured on the filter paper. Solid particles in the mixture do not pass through it and remain on the filter.
Fruit and vegetable juices are usually filtered before drinking to separate the seeds and solid particles of pulp. The method of filtration is also used in the process of preparing cottage cheese (paneer) in our homes. You might have seen that for making paneer, a few drops of lemon juice are added to milk as it boils. This gives a mixture of particles of solid paneer and a liquid. The paneer is then separated by filtering the mixture through a fine cloth or a strainer.
-
Evaporation
Evaporation
Activity 6
Add two spoons of salt to water in another beaker and stir it well. Do you see any change in the colour of water? Can you see any salt in the beaker, after stirring? Heat the beaker containing the salt water (Fig. 5.11). Let the water boil away. What is left in the beaker? In this activity, we used the process of evaporation, to separate a mixture of water and salt.
The process of conversion of water into its vapour is called evaporation. The process of evaporation takes place continuously wherever water is present. Where do you think, salt comes from? Sea water contains many salts mixed in it. One of these salts is the common salt. When sea water is allowed to stand in shallow pits, water gets heated by sunlight and slowly turns into water vapour, through evaporation. In a few days, the water evaporates completely leaving behind the solid salts (Fig. 5.12).

Fig. 5.12 Obtaining salt from sea water
Common salt is then obtained from this mixture of salts by further purification.
-
Evaporation
Use of more than one method of separation
Evaporation and Condensation
We have studied some methods for separation of substances from their mixtures. Often, one method is not sufficient to separate the different substances present in a mixture. In such a situation, we need to use more than one of these methods.
Activity 7
Take a mixture of sand and salt. How will we separate these? We already saw that handpicking would not be a practical method for separating these.
Keep this mixture in a beaker and add some water to it. Leave the beaker aside for some time. Do you see the sand settling down at the bottom? The sand can be separated by decantation or filtration. What does the decanted liquid contain? Do you think this water contains the salt which was there in the mixture at the beginning?
Now, we need to separate salt and water from the decanted liquid. Transfer this liquid to a kettle and close its lid. Heat the kettle for some time. Do you notice steam coming out from the spout of the kettle?
Take a metal plate with some ice on it. Hold the plate just above the spout of the kettle as shown in Fig. 5.13. What do you observe? Let all the water in the kettle boil off.
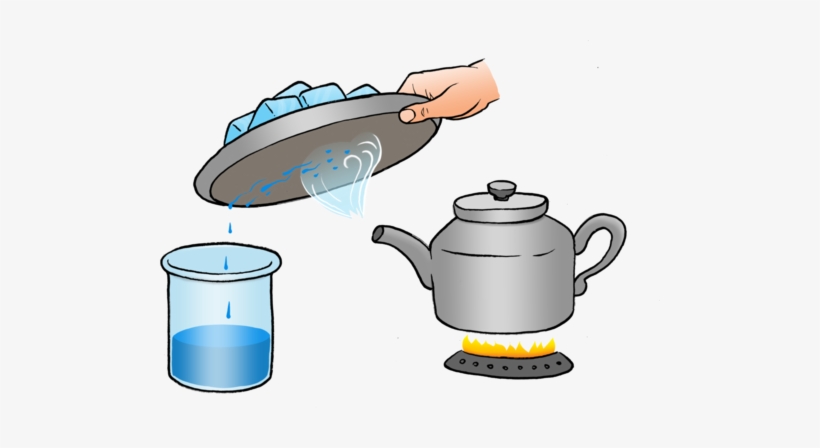
Fig. 5.13 Evaporation and condensation
When the steam comes in contact with the metal plate cooled with ice, it condenses and forms liquid water. The water drops that you observed falling from the plate, were due to condensation of steam. The process of conversion of water vapour into its liquid form is called condensation.
Did you ever see water drops condensed under a plate that has been used to cover a vessel containing milk that has just been boiled?
After all the water has evaporated, what is left behind in the kettle?
We have thus, separated salt, sand and water using processes of decantation, filtration, evaporation and condensation.
Paheli faced a problem while recovering salt mixed with sand. She has mixed a packet of salt in a small amount of sand. She then tried the method suggested in Activity 7, to recover the salt. She found, however, that she could recover only a small part of the salt that she had taken. What could have gone wrong?