-
Electron Transfer Redox Reactions
REDOX REACTIONS IN TERMS OF ELECTRON TRANSFER REACTIONS
We have already learnt that the reactions (g) 2NaCl (s) \(\to\)2Na(s) + Cl2 (8.12)
4Na(s) + O2(g) \(\to\) 2Na2O(s) (8.13)
2Na(s) + S(s) \(\to\) Na2S(s) (8.14)
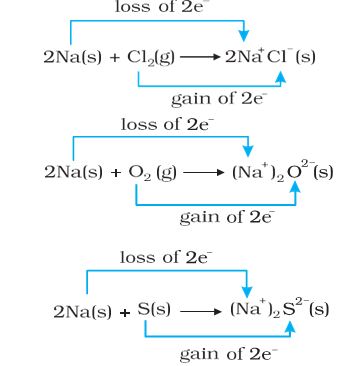
are redox reactions because in each of these reactions sodium is oxidized due to the addition of either oxygen or more electronegative element to sodium. Simultaneously, chlorine, oxygen, and sulphur are reduced because to each of these, the electropositive element sodium has been added. From our knowledge of chemical bonding we also know that sodium chloride, sodium oxide, and sodium sulphide are ionic compounds and perhaps better written as Na+Cl– (s), (Na+)2 O2–(s), and (Na+) S2–(s). Development of charges on the species produced suggests we rewrite the reactions (8.12 to 8.14) in the following manner :
For convenience, each of the above processes can be considered as two separate steps, one involving the loss of electrons and the other the gain of electrons. As an illustration, we may further elaborate on one of these, say, the formation of sodium chloride.
2 Na(s) \(\to\) 2 Na+(g) + 2e–
Cl (g) + 2e– \(\to\) 2 Cl–(g)
Each of the above steps is called a half-reaction, which explicitly shows the involvement of electrons. The Sum of the half-reactions gives the overall reaction :
2 Na(s) + Cl2 (g) \(\to\) 2 Na+ Cl– (s) or 2 NaCl (s)
Reactions 8.12 to 8.14 suggest that half-reactions that involve loss of electrons are called oxidation reactions. Similarly, the half-reactions that involve the gain of electrons are called reduction reactions. It may not be out of context to mention here that the new way of defining oxidation and reduction has been achieved only by establishing a correlation between the behavior of species as per the classical idea and their interplay in electron-transfer change. In reactions (8.12 to 8.14) sodium, which is oxidized, acts as a reducing agent because it donates electrons to each of the elements interacting with it and thus helps in reducing them. Chlorine, oxygen, and sulphur are reduced and act as oxidizing agents because these accept electrons from sodium. To summarise, we may mention that
Oxidation: Loss of electron(s) by any species.
Reduction: Gain of electron(s) by any species. Oxidizing agent: Acceptor of electron(s).
Reducing agent: Donor of electron(s).
-
Problems on Electron Transfer Redox Reactions
PROBLEM 2
Justify that the reaction : 2 Na(s) + H2(g) \(\to\) 2 NaH (s) is a redox change.
SOLUTION
Since in the above reaction the compound formed is an ionic compound, which may also be represented as Na+H– (s), this suggests that one half-reaction in this process is :
2 Na (s) \(\to\) 2 Na+(g) + 2e– and the other half-reaction is:
H2 (g) + 2e– \(\to\) 2 H–(g)
This splitting of the reaction under examination into two half-reactions automatically reveals that here sodium is oxidized and hydrogen is reduced, therefore, the complete reaction is a redox change.