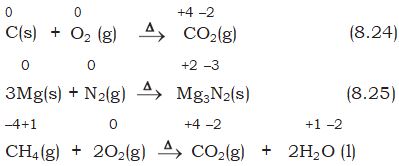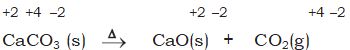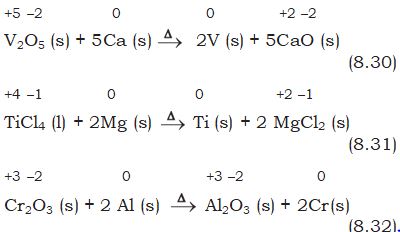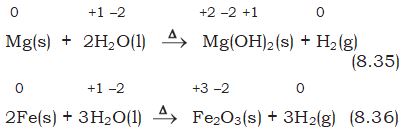-
Combination and Decomposition Reaction
1. Combination reactions
A combination reaction may be denoted in the manner:
A + B \(\to\) C
Either A and B or both A and B must be in the elemental form for such a reaction to be a redox reaction. All combustion reactions, which make use of elemental dioxygen, as well as other reactions involving elements other than dioxygen, are redox reactions. Some important examples of this category are:
2. Decomposition reactions
Decomposition reactions are the opposite of combination reactions. Precisely, a decomposition reaction leads to the breakdown of a compound into two or more components at least one of which must be in the elemental state. Examples of this class of reactions are:
It may carefully be noted that there is no change in the oxidation number of hydrogen in methane under combination reactions and that of potassium in potassium chlorate in reaction (8.28). This may also be noted here that all decomposition reactions are not redox reactions. For example, decomposition of calcium carbonate is not a redox reaction.
-
Displacement Reaction
3. Displacement reactions
In a displacement reaction, an ion (or an atom) in a compound is replaced by an ion (or an atom) of another element. It may be denoted as:
X + YZ \(\to\) XZ + Y
Displacement reactions fit into two categories: metal displacement and non-metal displacement.
(a) Metal displacement: A metal in a compound can be displaced by another metal in the uncombined state. We have already discussed about this class of the reactions under section 8.2.1. Metal displacement reactions find many applications in metallurgical processes in which pure metals are obtained from their compounds in ores. A few such examples are:
+2 +6 –2 0 0 +2 +6 –2
CuSO4(aq) + Zn (s) \(\to\) Cu(s) + ZnSO4 (aq) (8.29)
In each case, the reducing metal is a better reducing agent than the one that is being reduced which evidently shows more capability to lose electrons as compared to the one that is reduced.
(b) Non-metal displacement: The non-metal displacement redox reactions include hydrogen displacement and a rarely occurring reaction involving oxygen displacement.
All alkali metals and some alkaline earth metals (Ca, Sr, and Ba) which are very good reductants, will displace hydrogen from cold water.
0 +1 –2 +1 –2 +1 0
2Na(s) + 2H2O(l) \(\to\) 2NaOH(aq) + H2(g) (8.33)
0 +1 –2 +2 –2 +1 0
Ca(s) + 2H2O(1) \(\to\) Ca(OH)2 (aq) + H2(g) (8.34)
Less active metals such as magnesium and iron react with steam to produce dihydrogen gas:
Many metals, including those which do not react with cold water, are capable of displacing hydrogen from acids. Dihydrogen from acids may even be produced by such metals which do not react with steam. Cadmium and tin are the examples of such metals. A few examples for the displacement of hydrogen from acids are:
0 +1 –1 +2 –1 0
Zn(s) + 2HCl(aq) \(\to\) ZnCl2 (aq) + H2 (g) (8.37)
0 +1 –1 +2 –1 0
Mg (s) + 2HCl (aq) \(\to\) MgCl2 (aq) + H2 (g) (8.38)
0 +1 –1 +2 –1 0
Fe(s) + 2HCl(aq) \(\to\) FeCl2(aq) + H2(g) (8.39)
Reactions (8.37 to 8.39) are used to prepare dihydrogen gas in the laboratory. Here, the reactivity of metals is reflected in the rate of hydrogen gas evolution, which is the slowest for the least active metal Fe, and the fastest for the most reactive metal, Mg. Very less active metals, which may occur in the native state such as silver (Ag), and gold (Au) do not react even with hydrochloric acid.In section (8.2.1) we have already discussed that the metals – zinc (Zn), copper (Cu) and silver (Ag) through tendency to lose electrons show their reducing activity in the order Zn> Cu>Ag. Like metals, activity series also exists for the halogens. The power of these elements as oxidising agents decreases as we move down from fluorine to iodine in group 17 of the periodic table. This implies that fluorine is so reactive that it can replace chloride, bromide and iodide ions in solution. In fact, fluorine is so reactive that it attacks water and displaces the oxygen of water :
+1 –2 0 +1 –1 0
2H2O (l) + 2F2 (g) \(\to\) 4HF(aq) + O2(g) (8.40)
It is for this reason that the displacement reactions of chlorine, bromine and iodine using fluorine are not generally carried out in aqueous solution. On the other hand, chlorine can displace bromide and iodide ions in an aqueous solution as shown below:
0 +1 –1 +1 –1 0
Cl2 (g) + 2KBr (aq) \(\to\) 2 KCl (aq) + Br2 (1) (8.41)
0 +1–1 +1 –1 0
Cl2 (g) + 2KI (aq) \(\to\) 2 KCl (aq) + I2 (s) (8.42)
As Br2 and I2 are coloured and dissolve in CCl4, can easily be identified from the colour of the solution. The above reactions can be written in ionic form as:
0 –1 –1 0
Cl2 (g) + 2Br (aq) \(\to\) 2Cl (aq) + Br (1) (8.41a)
0 –1 –1 0
Cl2 (g) + 2I (aq) \(\to\) 2Cl (aq) + I (s) (8.42b)
Reactions (8.41) and (8.42) form the basis of identifying Br– and I– in the laboratory through the test popularly known as ‘Layer Test’. It may not be out of place to mention here that bromine likewise can displace iodide ion in solution:
0 –1 –1 0
Br2 (l) + 2I (aq) \(\to\) 2Br (aq) + I (s) (8.43)
The halogen displacement reactions have a direct industrial application. The recovery of halogens from their halides requires an oxidation process, which is represented by:
2X– \(\to\) X2 + 2e– (8.44)
here X denotes a halogen element. Whereas chemical means are available to oxidise Cl–, Br– and I–, as fluorine is the strongest oxidising agent; there is no way to convert F- ions to F2 by chemical means. The only way to achieve F from F– is to oxidise electrolytically, the details of which you will study at a later stage.
-
Disproportionation Reactions
4. Disproportionation reactions
Disproportionation reactions are a special type of redox reactions. In a disproportionation reaction an element in one oxidation state is simultaneously oxidised and reduced. One of the reacting substances in a disproportionation reaction always contains an element that can exist in at least three oxidation states. The element in the form of reacting substance is in the intermediate oxidation state; and both higher and lower oxidation states of that element are formed in the reaction. The decomposition of hydrogen peroxide is a familiar example of the reaction, where oxygen experiences disproportionation.
+1 –1 +1 –2 0
2H2O2 (aq) \(\to\) 2H2O(l) + O2(g) (8.45)
Here the oxygen of peroxide, which is present in –1 state, is converted to zero oxidation state in O2 and decreases to –2 oxidation state in H2O.
Phosphorous, sulphur and chlorine undergo disproportionation in the alkaline medium as shown below :
The reaction (8.48) describes the formation of household bleaching agents. The hypochlorite ion (ClO–) formed in the reaction oxidises the colour-bearing stains of the substances to colourless compounds.
It is of interest to mention here that whereas bromine and iodine follow the same trend as exhibited by chlorine in reaction (8.48), fluorine shows deviation from this behaviour when it reacts with alkali. The reaction that takes place in the case of fluorine is as follows:
2 F2 (g) + 2OH–(aq) \(\to\) 2 F–(aq) + OF2 (g) + H2O(1) (8.49)
(It is to be noted with care that fluorine in reaction (8.49) will undoubtedly attack water to produce some oxygen also). This departure shown by fluorine is not surprising for us as we know the limitation of fluorine that, being the most electronegative element, it cannot exhibit any positive oxidation state. This means that among halogens, fluorine does not show a disproportionation tendency.
-
Problems on Redox Reactions
PROBLEM 5
Which of the following species, do not show disproportionation reaction and why? ClO , ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _2^ - \) , ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _3^ - \) and ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _4^ - \) Also write reaction for each of the species that disproportionates.
SOLUTION
Among the oxoanions of chlorine listed above, ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _4^ - \) does not disproportionate because in this oxoanion chlorine is present in its highest oxidation state that is, +7. The disproportionation reactions for the other three oxoanions of chlorine are as follows:
+1 –1 +5
3ClO \(\to\)\(\) 2Cl-+ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _3^ - \)
+3 +5 -1
6ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _2^ - \)
4ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _3^ - \)+2Cl-
+5 -1 +7
4ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _3^ - \) \(\to\) Cl- +3ClO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _4^ - \)
PROBLEM 6
Suggest a scheme of classification of the following redox reactions
(a) N2 (g) + O2 (g) \(\to\) 2 NO (g)
(b) 2Pb(NO3)2(s) \(\to\) 2PbO(s) + 4 NO2 (g) +O2 (g)
(c) NaH(s) + H2O(1) \(\to\) NaOH(aq) + H2 (g)
(d) 2NO2 (g) + 2OH- (aq) \(\to\)NO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _2^ - \) (aq) +NO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaikdaaeaacqGHsislaaaaaa!37CC! _3^ - \) (aq)+H2O(1)
SOLUTION
In reaction (a), the compound nitric oxide is formed by the combination of the elemental substances, nitrogen and oxygen; therefore, this is an example of combination redox reactions. The reaction
(b) involves the breaking down of lead nitrate into three components; therefore, this is categorised under decomposition redox reaction. In reaction
(c) hydrogen of water has been displaced by hydride ion into dihydrogen gas. Therefore, this may be called as displacement redox reaction. The reaction
(d) involves disproportionation of NO2 (+4 state)into NO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaiodaaeaacqGHsislaaaaaa!37CD! _2^ - \) (+3 state) and NO\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaWaa0baaSqaai % aaiodaaeaacqGHsislaaaaaa!37CD! _3^ - \) (+5 state). Therefore reaction (d) is an example of disproportionation redox reaction.
PROBLEM 7
Why do the following reactions proceed differently ?
Pb3O4 + 8HCl \(\to\) 3PbCl2 + Cl2 + 4H2O
and
Pb3O4 + 4HNO3 \(\to\) 2Pb(NO3)2 + PbO2 +2H2O
SOLUTION
Pb3O4 is actually a stoichiometric mixture of 2 mol of PbO and 1 mol of PbO2. In PbO2, lead is present in +4 oxidation state, whereas the stable oxidation state of lead in PbO is +2. PbO2 thus can act as an oxidant (oxidising agent) and, therefore, can oxidise Cl– ion of HCl into chlorine. We may also keep in mind that PbO is a basic oxide. Therefore, the reaction.
Pb3O4 + 8HCl \(\to\) 3PbCl2 + Cl2 + 4H2O
can be splitted into two reactions namely: 2PbO + 4HCl \(\to\) 2PbCl2 + 2H2O
(acid-base reaction)
+4 –1 +2 0
PbO2 + 4HCl \(\to\) PbCl2 + Cl2 +2H2O
(redox reaction)
Since HNO3 itself is an oxidising agent therefore, it is unlikely that the reaction may occur between PbO2 and HNO3. However, the acid-base reaction occurs between PbO and HNO3 as:
2PbO + 4HNO3 \(\to\) 2Pb(NO3)2 + 2H2O
It is the passive nature of PbO2 against HNO3 that makes the reaction different from the one that follows with HCl.