-
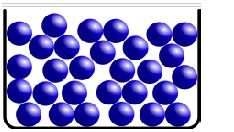
Solids
SOLIDS:
Solids are one of the three main states of matter, along with liquids and gases. In a solid, particles are closely packed together in a fixed and orderly arrangement. This arrangement gives solids their definite shape and volume. Here are some key characteristics and properties of solids:
Particle Arrangement:
In solids, particles (atoms, molecules, or ions) are closely packed and arranged in a regular and repeating pattern. This arrangement gives solids a fixed shape.
The particles in a solid vibrate around fixed positions, but they do not move from their places.Definite Shape and Volume:
Solids have a definite shape and volume. The fixed arrangement of particles prevents them from flowing or taking the shape of their container.
The shape of a solid is determined by the arrangement of its particles, and it remains constant unless an external force is applied.Density:
Solids are typically denser than liquids and gases because of the close packing of particles.
Density is defined as mass per unit volume, and solids generally have a higher density compared to the same substance in its liquid or gaseous state.Incompressibility:
Solids are generally incompressible, meaning that their volume is difficult to reduce. When pressure is applied, the particles are already tightly packed, leaving little room for further compression.Melting Point:
Solids have a characteristic melting point, which is the temperature at which they transition from a solid to a liquid state.
The melting point is influenced by factors such as intermolecular forces and the strength of bonds between particles.Thermal Expansion:
Like liquids and gases, solids also exhibit thermal expansion when heated. However, the expansion is typically less pronounced compared to liquids and gases.Electrical Conductivity:
Electrical conductivity in solids varies. Some solids conduct electricity well (metals), while others are insulators (wood, rubber) or semiconductors (silicon).
The ability to conduct electricity is related to the mobility of electrons or charged particles within the solid.Crystal Structure:
Many solids exhibit a repeating and orderly arrangement of particles known as a crystal lattice. Crystalline solids have a well-defined geometric pattern, while amorphous solids lack a regular structure.Examples
Examples of solids include metals, minerals, wood, plastics, and ice. Understanding the properties of solids is crucial in various scientific and engineering fields, as it provides insights into their behavior under different conditions.
-
Solids
SOLIDS:
Solids are one of the three main states of matter, along with liquids and gases. In a solid, particles are closely packed together in a fixed and orderly arrangement. This arrangement gives solids their definite shape and volume. Here are some key characteristics and properties of solids:
Particle Arrangement:
In solids, particles (atoms, molecules, or ions) are closely packed and arranged in a regular and repeating pattern. This arrangement gives solids a fixed shape.
The particles in a solid vibrate around fixed positions, but they do not move from their places.Definite Shape and Volume:
Solids have a definite shape and volume. The fixed arrangement of particles prevents them from flowing or taking the shape of their container.
The shape of a solid is determined by the arrangement of its particles, and it remains constant unless an external force is applied.Density:
Solids are typically denser than liquids and gases because of the close packing of particles.
Density is defined as mass per unit volume, and solids generally have a higher density compared to the same substance in its liquid or gaseous state.Incompressibility:
Solids are generally incompressible, meaning that their volume is difficult to reduce. When pressure is applied, the particles are already tightly packed, leaving little room for further compression.Melting Point:
Solids have a characteristic melting point, which is the temperature at which they transition from a solid to a liquid state.
The melting point is influenced by factors such as intermolecular forces and the strength of bonds between particles.Thermal Expansion:
Like liquids and gases, solids also exhibit thermal expansion when heated. However, the expansion is typically less pronounced compared to liquids and gases.Electrical Conductivity:
Electrical conductivity in solids varies. Some solids conduct electricity well (metals), while others are insulators (wood, rubber) or semiconductors (silicon).
The ability to conduct electricity is related to the mobility of electrons or charged particles within the solid.Crystal Structure:
Many solids exhibit a repeating and orderly arrangement of particles known as a crystal lattice. Crystalline solids have a well-defined geometric pattern, while amorphous solids lack a regular structure.Examples
Examples of solids include metals, minerals, wood, plastics, and ice. Understanding the properties of solids is crucial in various scientific and engineering fields, as it provides insights into their behavior under different conditions.
-
Solids
SOLIDS:
Solids are one of the three main states of matter, along with liquids and gases. In a solid, particles are closely packed together in a fixed and orderly arrangement. This arrangement gives solids their definite shape and volume. Here are some key characteristics and properties of solids:
Particle Arrangement:
In solids, particles (atoms, molecules, or ions) are closely packed and arranged in a regular and repeating pattern. This arrangement gives solids a fixed shape.
The particles in a solid vibrate around fixed positions, but they do not move from their places.Definite Shape and Volume:
Solids have a definite shape and volume. The fixed arrangement of particles prevents them from flowing or taking the shape of their container.
The shape of a solid is determined by the arrangement of its particles, and it remains constant unless an external force is applied.Density:
Solids are typically denser than liquids and gases because of the close packing of particles.
Density is defined as mass per unit volume, and solids generally have a higher density compared to the same substance in its liquid or gaseous state.Incompressibility:
Solids are generally incompressible, meaning that their volume is difficult to reduce. When pressure is applied, the particles are already tightly packed, leaving little room for further compression.Melting Point:
Solids have a characteristic melting point, which is the temperature at which they transition from a solid to a liquid state.
The melting point is influenced by factors such as intermolecular forces and the strength of bonds between particles.Thermal Expansion:
Like liquids and gases, solids also exhibit thermal expansion when heated. However, the expansion is typically less pronounced compared to liquids and gases.Electrical Conductivity:
Electrical conductivity in solids varies. Some solids conduct electricity well (metals), while others are insulators (wood, rubber) or semiconductors (silicon).
The ability to conduct electricity is related to the mobility of electrons or charged particles within the solid.Crystal Structure:
Many solids exhibit a repeating and orderly arrangement of particles known as a crystal lattice. Crystalline solids have a well-defined geometric pattern, while amorphous solids lack a regular structure.Examples
Examples of solids include metals, minerals, wood, plastics, and ice. Understanding the properties of solids is crucial in various scientific and engineering fields, as it provides insights into their behavior under different conditions.
-
Liquids
Liquids
Liquids are one of the three main states of matter, alongside solids and gases. Unlike solids, which have a fixed shape, and gases, which fill the entire volume of their container, liquids have distinct characteristics that set them apart. Here are some key features and properties of liquids:
Particle Arrangement:
In liquids, particles (atoms, molecules, or ions) are still closely packed, but they are not in a fixed position. The arrangement is more random compared to solids.
The particles in a liquid have more freedom of movement than those in a solid. They can slide past each other, allowing liquids to flow and take the shape of their container.Definite Volume but Indefinite Shape:
Liquids have a definite volume, meaning they have a fixed amount of space they occupy.
Unlike solids, liquids do not have a fixed shape. They take the shape of the container they are in while maintaining their volume.Density:
The density of liquids is generally higher than that of gases but lower than that of solids. It is influenced by factors such as temperature and pressure.
The density of a liquid is typically less than that of the same substance in its solid state.Incompressibility:
Liquids are generally considered to be incompressible, meaning their volume is not easily reduced under pressure.
While liquids are more compressible than solids, they are still much less compressible than gases.Surface Tension:
Liquids exhibit surface tension, a property that causes the surface of a liquid to behave like a stretched elastic membrane. This is due to the cohesive forces between the liquid particles. Due to this liquids occupy definite volume
Surface tension is responsible for phenomena such as the formation of droplets and the ability of certain insects to "walk on water."Vaporization:
Liquids can undergo vaporization, a process in which they turn into a gas. This can occur through evaporation (at the liquid's surface) or boiling (throughout the liquid).
The temperature at which a liquid turns into a gas is known as its boiling point.Thermal Expansion:
Similar to solids, liquids also exhibit thermal expansion when heated. However, their expansion is generally more pronounced than that of solids.Viscosity:
Viscosity is a measure of a liquid's resistance to flow. Liquids with high viscosity flow more slowly, while those with low viscosity flow more easily.
Viscosity is influenced by factors such as temperature and the size and shape of the liquid particles.
Examples
Examples of liquids include water, oil, milk, and alcohol. Understanding the properties of liquids is essential in fields such as chemistry, physics, and engineering, as it helps explain their behavior under different conditions and contributes to the design and optimization of various processes. -
Gases
Gases:
Gases are one of the three main states of matter, alongside solids and liquids. Gases exhibit distinct characteristics that distinguish them from the other states. Here are some key features and properties of gases:
Particle Arrangement:
In gases, particles (atoms or molecules) are widely spaced and move freely with high kinetic energy.
The particles are not held in a fixed position, and they travel in straight lines until they collide with each other or the walls of the container.Volume and Shape:
Gases do not have a fixed shape or volume. They expand to fill the entire container they occupy.
The shape of a gas is determined by the shape of its container, and it takes on the volume of the container.Density:
Gases generally have a much lower density compared to liquids and solids. The low density is a result of the large spaces between gas particles.
The density of a gas is influenced by factors such as temperature and pressure.Compressibility:
Gases are highly compressible, meaning their volume can be reduced significantly under pressure.
The compressibility of gases is attributed to the large distances between particles, allowing them to be squeezed closer together.Diffusion and Effusion:
Gases exhibit rapid diffusion, which is the process of particles spreading from areas of higher concentration to areas of lower concentration.
Effusion is the escape of gas particles through a tiny opening. Gases with lower molar masses (lower masses) effuse more quickly than those with higher molar masses (higher masses) (Note: you will learn about molar masses in higher classes).Boyle's Law, Charles's Law, and Avogadro's Law:
Boyle's Law states that, at constant temperature, the pressure of a gas is inversely proportional to its volume.
Charles's Law states that, at constant pressure, the volume of a gas is directly proportional to its temperature.
Avogadro's Law states that, at constant temperature and pressure, the volume of a gas is directly proportional to the number of moles of gas.Temperature and Pressure:
The behavior of gases is highly influenced by temperature and pressure. The ideal gas law, PV = nRT (where P is pressure, V is volume, n is the number of moles, R is the gas constant, and T is temperature), describes the relationship between these variables for an ideal gas. (NOTE: You will learn more about Ideal gas equation in higher classes)
Phase Changes:
Gases can undergo phase changes, such as condensation (from gas to liquid) and deposition (from gas to solid).
The temperature at which a gas condenses into a liquid is called its dew point.Examples
Examples of gases include oxygen, nitrogen, hydrogen, and carbon dioxide. Understanding the properties of gases is essential in various scientific and industrial applications, such as in the design of gas storage systems, combustion processes, and the study of atmospheric phenomena.