-
VALENCY & ELECTROPOSITIVE IONS
When atoms of two or more elements combine, a molecule of a compound is formed. The capacities of these elements to combine with each other determine the formula of the compound formed.
The capacity of an element to combine with other elements is known as its valency.
Explanation of Valency
General Defnition of Valency :
Number of hydorgen atoms or chlorine atoms or double the number of oxygen atoms with which one atom of an element combines is called its Valency.
It will be evident from the following that the combining capacities of all elements are not the same.
1. One atom of Cl combines with one atom of H to form a molecule of hydrogen chloride (HCl).
2. One atom of O combines with two atoms of H to form a molecule of water (H2O).
3. One atom of N combines with three atoms of H to form a molecule of ammonia (NH3).
Thus, the combining capacity of O is twice that of Cl, and that of N is thrice that of Cl.
H is assigned a valency of 1. So the valencies of Cl, O and N are 1, 2 and 3 respectively. However, many atoms do not combine with H. Their valencies are calculated by the number of Cl atoms they combine with, since Cl and H have the same valency, i.e., 1.- The valency of an element is given by the number of H or Cl atoms mainly that an atom of the element combines with.
Modern Definition of Valency :
According to the new concepts, Valency can be defind as the number of electrons that are lost or gained or shared with one atom of an element, to acquire the stable configuration of the nearest noble gas element.
In order to understand modern definition of Valency, we should know few concepts of atom
What is atom made up of ?
An atom consists of two parts :
(a) Nucleus :
Nucleus is situated in the centre of an atom. All the protons & neutrons are situated in the nucleus, therefore, the entire mass of an atom is almost concentrated in the nucleus. The overall charge of nucleus is positive due to the presence of positively charged protons (neutrons have no charge). The protons & neutrons are collectively called nucleons. The radius of the nucleus of an atom is of the order of 10-15 m.
(b) Extra nuclear region :
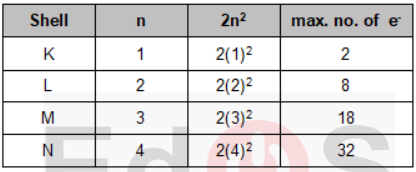
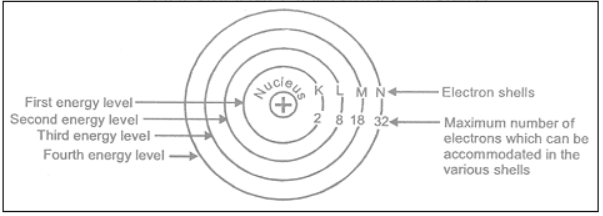
In extra nuclear part or in the region outside the nucleus, electrons are present which revolve around the nucleus in orbits of fixed energies. These orbits are called energy levels. These energy levels are designated as K, L, M, N and so on.
i) The maximum number of electrons that can be accommodated in a shell is given by the formula 2n2 (where n = number of shell i.e. 1, 2, 3—)
Diagramatical representation of Atom
(ii) In the outermost shell of any atom, the maximum possible number of electrons is 8, except in the first shell which can have at the most 2 electrons.
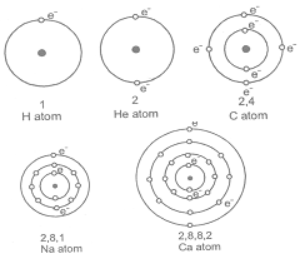
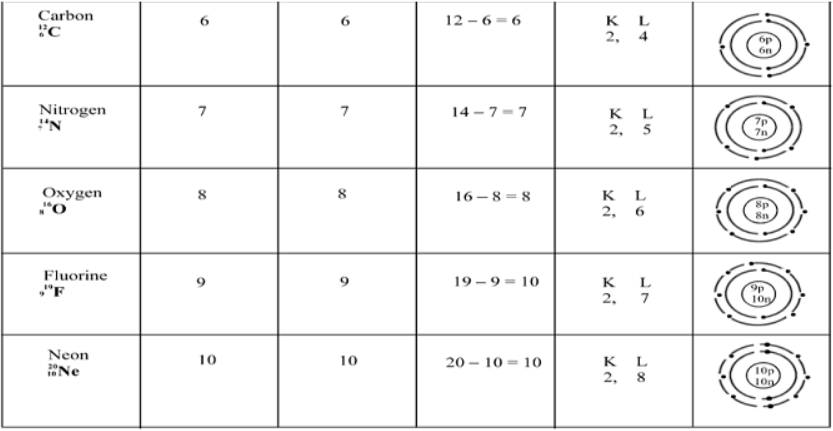
(iii) The arrangement of the electrons in different shells is known as the electronic configuration of the element. The pictorial representations of Bohr’s model of hydrogen, helium, carbon, sodium and calcium atoms having 1, 2, 6, 11 and 20 electrons respectively are shown in the figure where the centre of the circle represents the nucleus.
Atomic number or Nuclear charge
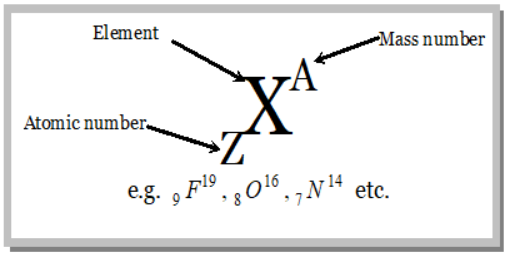
(i) The number of protons present in the nucleus of the atom is called atomic number (Z)
(ii) Atomic number = Number of positive charge on nucleus = Number of protons in nucleus = Number of electrons in neutral atom.
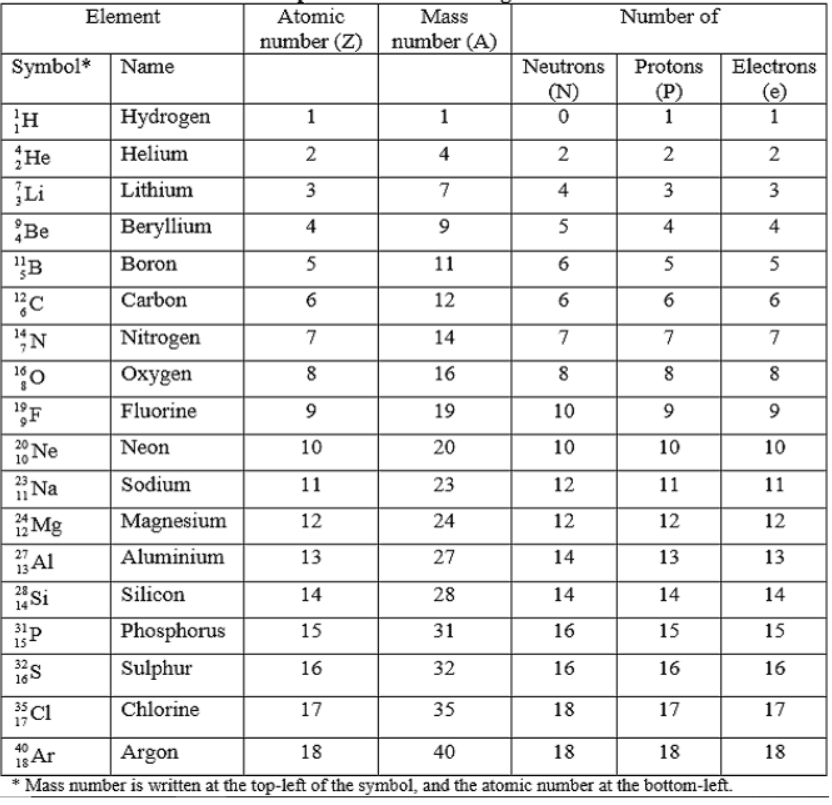
(iii) Two different elements can never have identical atomic number.Mass Number
(i) The sum of proton and neutrons present in the nucleus is called mass number.
Mass number (A) = Number of protons + Number of neutrons or Atomic number (Z) or Number of neutrons = A – Z .
(ii) Since mass of a proton or a neutron is not a whole number (on atomic weight scale), weight is not necessarily a whole number.
(iii) The atom of an element X having mass number (A) and atomic number (Z) may be represented by a symbol,
Nuclear Composition of Elements
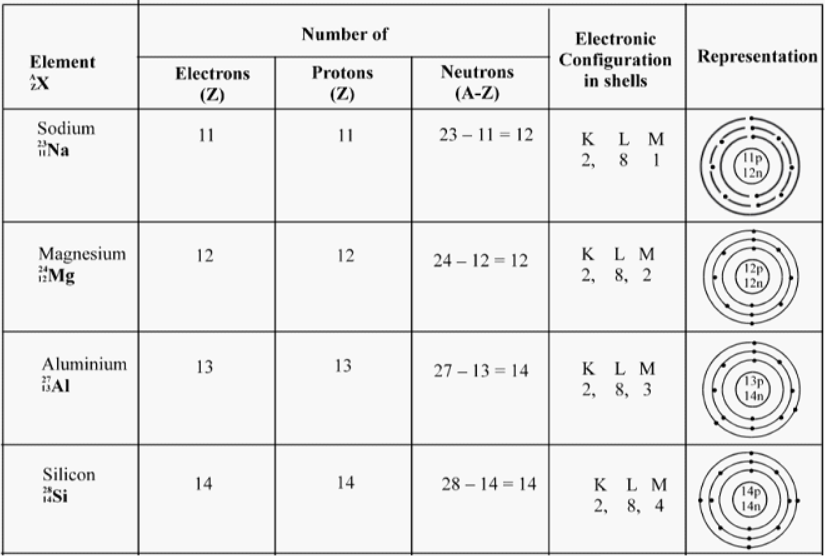
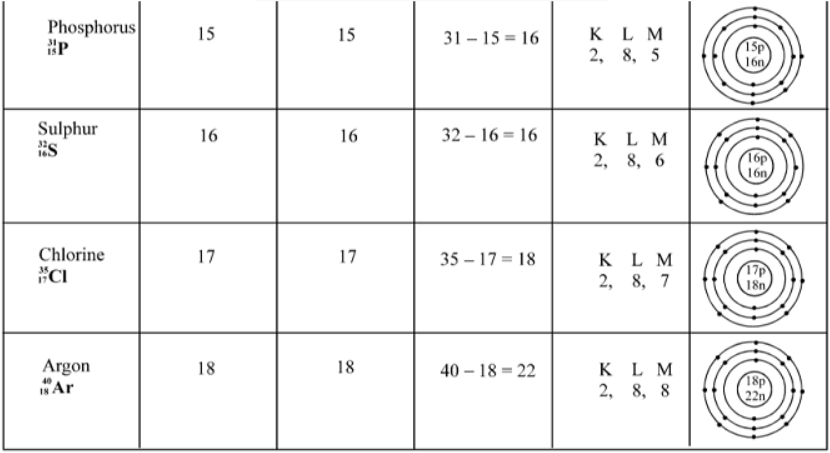
Electronic Configuration of Elements
The distribution of electrons in different orbitals is known as electronic Configuration
Valence electrons:
The electrons in the outermost shell of an atom are called its valence electrons.
Example : The number of valence electron in sodium is one. The number of valence electrons in chlorine is seven.
Explanation of Valency with respect to Configuration
As per the modern defnition, The number of electrons gained, lost or contributed for sharing by an atom of the element to acquire the stable configuration of the nearest noble gas element gives us directly the combining capacity or valency of the element.
Valency of an element is determined by the number of valence electrons in an atom of the element.
Valency, a whole number : Valency of an element varies from 1 to 8. It may be zero for an element which does not combine with any other element. (inert gases like He, Ne, Ar, Xe).The valency of an element = number of valence electrons
(when number of valence electrons are from 1 to 4)
The valency of an element = 8 - number of valence electrons.
(when number of valence electrons are more than 4)RADICAL or ION
When an atom looses or gains electrons it converts into Radical or Ion. (or) An atom or group of atoms with charge on them is called Radical or ion
(ii) Classification of ions: Ions can be classified as follows into two types of based on the type of atoms involved in ion formation.
(a) Simple ion : It is an ion which contains one or more atoms of the same element.
Ex : , etc,.
(b) Compound ion : It consists of two or more atoms of different elements forming a single unit.
Ex: \( SO_4 ^{2 - } ,NO_3 ^ - \), etc,.
Positive valency (Cation) :
If the atom donates or looses its electrons from its outermost orbit to get stability, the valency is called as positive valency and the that ion is called as cation or Electropositive ion. (or) Radical or ion having positive charge on them is called Cation and shows positive valency. Cations are also called Basic Radicals.
Example: Na+, Ca2+, Al3+,
Negative valency (Anion) :
When an atom gain one or more electrons to get stability, The number of electrons accepted by an atom of an element is its negative valency and the ion is called Anion or Electronegative ion. (or)Radical or ion having negative charge on them is called anion. Anions are also called Acidic Radicals.
Example: \( Cl^ - ,O^{2 - } ,N^{3 - } ,SO_4^{2 - } ,O_2^{2 - } \)
Discriptive Examples
i) Consider Sodium atom (Na)
The atomic number of sodium (Na) is 11.
Its electronic configuration is 2, 8, 1.
The number of electrons present in outermost shell (Valence electrons) is 1.
\( \therefore \)Sodium looses 1 electron and attains the stable configuration of 2,8 i.e., Neon which is a noble gas element
So, valency of Sodium is taken as 1
Ion Formation
After loosing one electron Sodium converts into Sodium ion. As it looses electron it converts to positive ion (generally called Cation).
\(\begin{gathered} \,\,\,\,\,Na\,\,\,\,\,\,\,\,\,\,\, \to \,\,\,\,\,\,\,Na^ + \,\, + \,\,1e^ - \hfill \\ 2,8,1\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,2,8 \hfill \\ \end{gathered} \)
ii) Consider Calcium atom (Ca)
The atomic number of Calcium (Ca) is 20.
Its electronic configuration is 2, 8, 8, 2.
The number of electrons present in outermost shell (Valence electrons) is 2.
Calcium looses 2 electron and attains the stable configuration of 2,8,8 i.e., Argon which is a noble gas element
So, valency of Calcium is taken as 2
Ion Formation
After loosing two electron Calcium converts into Calcium ion.
\( \begin{gathered} \,\,\,\,\,\,\,\,Ca\,\,\,\,\,\,\,\,\,\,\, \to \,\,\,\,\,\,\,\,\,\,\,\,\,\,Ca^{2 + } \,\, + \,2e^ - \hfill \\ 2,8,8,2\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,2,8,8 \hfill \\ \end{gathered} \)iii) Consider Chlorine atom (Cl)
The atomic number of Chlorine (Cl) is 17.
Its electronic configuration is 2, 8, 7.
The number of electrons present in outermost shell (Valence electrons) is 7.
\( \therefore \)Chlorine gaines 1 electron and attains the stable configuration of 2,8,8 i.e., Argon which is a noble gas element
So, valency of Chlorine is taken as 1
Ion Formation
After gaining one electron Chorine converts into Chloride ion. As it gains one electron it converts to negative ion (generally called Anion).
\( \begin{gathered} \,\,\,\,\,\,\,\,Cl\,\,\,\,\,\, + \,\,\,\,\,1e^ - \,\,\, \to \,\,\,\,\,\,\,\,\,\,Cl^ - \,\, \hfill \\ 2,8,7\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,2,8,8 \hfill \\ \end{gathered} \)Relation between valence electron and metallic and nonmetallic nature of Elements
(1) Elements having 1, 2 or 3 valency electrons are metals (except hydrogen).
3Li (2, 1), 11Na (2, 8, 1), 19K (2, 8, 8, 1) Valency electron 1 = Alkali metals
12Mg (2, 8, 2), 20Ca (2, 8, 8, 2) Valency electrons are 2 = Alkaline earth metal
13Al (2, 8, 3) Valency electrons are 3 = metal
(2) Elements having 5, 6, 7 valency electrons are nonmetals
7N (2, 5), 15P (2, 8, 5), 33Sb (2, 8, 18, 5) Valency electrons are 5 = nonmetals
8O (2, 6), 16S (2, 8, 6), 34Se (2, 8, 18, 6) Valency electrons are 6 = nonmetal
Halogens
9F (2, 7), 17Cl (2, 8, 7), 35Br (2, 8, 18, 7), 53lodine (2, 8, 18,18, 7)
Valency electrons are 7 = nonmetal
Inert gases
2He (2), 10Ne (2, 8), 18Ar (2, 8, 8) 36Kr (2, 8, 18, 8), 54Xe (2, 8, 18,18, 8)
Valency electrons are 8 = non-metal
Electropositive ions
When an atom looses one or more electrons to get stability, The number of electrons lost by an atom of an element is its Positive valency and the ion is called Cation or Electropositive ion. (or) Radical or ion having positive charge on them is called Electropositive ion (or) Cation.
Properties of Electropositive ions
1. They are called Basic Radicals.
2. Metals which have 1 to 3 electrons in outermost orbit (Valence shell) looses electrons and converts to Electropositive ions
3. The number of electrons in electropositive ions are less in number than protons due to loss of electons
4. The Size of the Electropositive ion is considerably less than a neutral atom due to increase of attractions of Protons on electrons left after froming ion.
5. Size of the Electropositive ion is inversly proportional to number of electrons lost for a particular element.
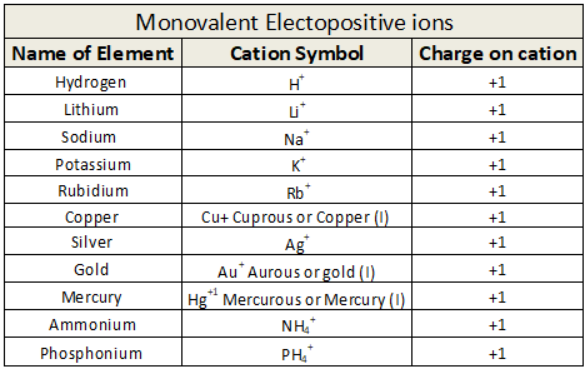
Monovalent electropositive ions
When an atom looses one electron or if any Radical or ion has uni positive charge on it then it is known as Monovalent electropositive ion or Univalent Electropositive ion.
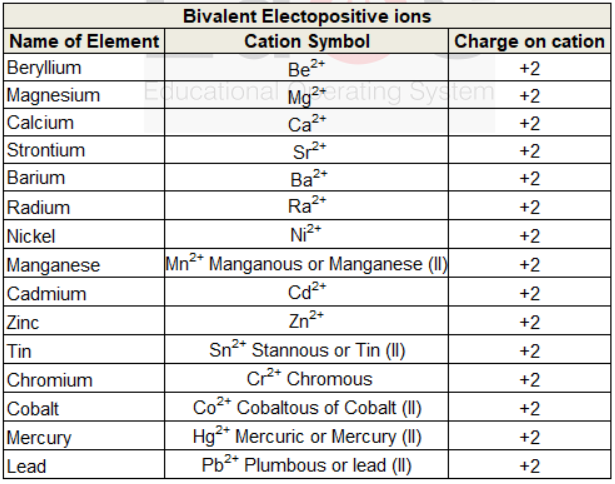
Bivalent electropositive ions
When an atom looses two electrons or if any Radical or ion has dipositive charge on it then it is known as Bivalent electropositive ion.
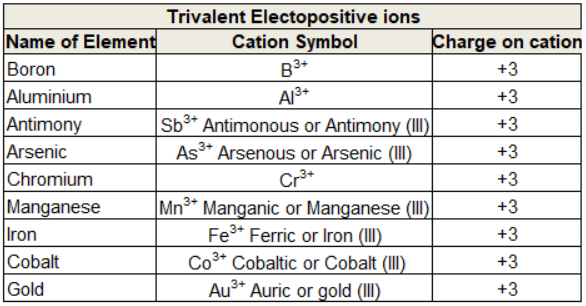
Trivalent electropositive ions
When an atom looses three electrons or if any Radical or ion has tripositive charge on it then it is known as Trivalent electropositive ion
Tetravalent electropositive ions
When an atom looses four electrons or if any Radical or ion has tetrapositive charge on it then it is known as Tetravalent electropositive ion
Pentavalent electropositive ions
Radical or ion has pentapositive charge on it then it is known as Pentavalent electropositive ion
Concept of Variable Valency
Most elements such as Na, K, Mg, Al, Ca, Ba etc..exhibit a fixed valency in all their compounds, Sometimes the same element may form two or more different compounds with another element by involving different number of electrons. Due to this the same element exhibits different valency which is called as Variable Valency and it is usually exhibited by Metals
Reason for showing Variable Valency
The reason for variable valency in certain metals is due to involvement of electrons in outermost orbit (Valence shell ) as well as the electrons in shell of preceeding orbit (called as penultimate orbit).
For Example : Under normal conditions Iron looses 2 electrons from outermost orbit to form Fe2+ ion and shows Valency of 2, But under definite experimental conditions Iron looses 3 electrons ( 2 from outermost orbit and one more electron from penultimate orbit and forms Fe3+ and shows valency of 3
Naming of compounds showing variable valency
The compound in which the element shows the lower valencies is indicated by the suffix -ous
The compound in which the element shows the higher valency is indicated by the suffix - ic
Examples :-
Iron exhibit two valencies 2 and 3
Iron in its lower valency 2 is named as ferrous
(Ex: Ferrous Chloride or iron (II) chloride FeCl2)
Iron in its higher valency 3, is named as ferric
(Ex: Ferric Chloride or iron (III) chloride FeCl3)
Copper shows valency 1 (monovalent) in (Cuprous Chloride, CuCl ) and shows Valency 2 (Bivalent) in (Cupric Chloride, CuCl2 )
Tin is divalent in stannous chloride (SnCl2) and tetravalent in stannic chloride (SnCl4).
The valency of sulphur is 4 in SO2 and 6 in SO3.