-
COMBUSTION, FUELS AND FLAMES
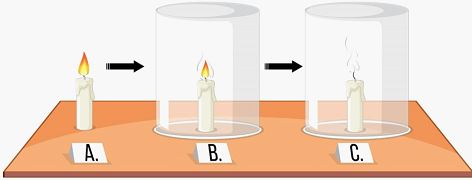
Does air is necessary for combustion process?
Lets do an activity:
To test the necessity of air (specifically, oxygen) for burning, you can perform a simple experiment. Keep in mind that safety precautions should always be taken when conducting experiments involving fire. Here's a basic demonstration you can try:
Materials:
Candle
Glass jar or small glass container
Plate or tray
Matches or a lighter
Procedure:
Light the candle and let it burn for a moment to establish a flame.
Quickly place the glass jar or container over the burning candle, completely covering it. Make sure the jar is large enough to fully enclose the flame.
Observations:
Initially, you'll observe the flame inside the jar.
Over time, the flame will begin to diminish and eventually extinguish.
Explanation:
As the candle burns, it consumes oxygen from the air to sustain the combustion reaction. When you place the jar over the candle, it creates a sealed environment. As the candle burns and uses up the available oxygen inside the jar, the flame goes out. This demonstrates the necessity of oxygen from the air for combustion.
This experiment illustrates the principle that combustion requires oxygen, and when the oxygen supply is limited or cut off, the flame is extinguished. It's important to note that safety precautions should be followed, and the experiment should be conducted in a well-ventilated area. Additionally, never leave a burning candle unattended, even in a controlled experiment.
Lab Experiment:
Aim: To prove that oxygen helps in burning
Materials:
Test tube, Test tube holder, Spirit lamp or alcohol burner, Matchbox, Incense stick (agarbatti), Potassium permanganate crystals
Procedure:
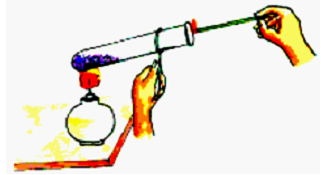
Set up the Test Tube:
Place the test tube in the test tube holder.
Add a small amount of potassium permanganate crystals to the test tube.
Ignite the Spirit Lamp:
Use the matchbox to light the spirit lamp.
Combustion of Potassium Permanganate:
Hold the test tube with potassium permanganate crystals over the flame of the spirit lamp. Ensure the crystals are above the flame, not in direct contact with it.
Observe the reaction.
Observations:
You may notice the potassium permanganate crystals start to undergo a reaction with the heat from the flame.
The crystals may change color and produce a purple or violet flame.
There may be the release of gas or other visible changes.
Explanation:
The combustion of potassium permanganate in this experiment is not a typical burning of a fuel. Instead, it's a reaction that involves the decomposition of potassium permanganate when exposed to heat. The purple or violet flame is characteristic of manganese ions formed during the reaction.
Please note that experiments involving chemicals should be conducted with caution. Potassium permanganate can cause stains and should be handled carefully. Ensure proper ventilation, and perform the experiment in a controlled environment. If you are unsure about any aspect of the experiment or the materials used, it's advisable to seek guidance from a knowledgeable individual, such as a chemistry teacher or laboratory supervisor.