-
Solvent Extraction
SEPERATION OF MIXTURES:
Mixture is said to be pure when its components are known and they are put together in a desired quantity.We need to seperate to remove unwanted substances from required substances.
Principles involved in the seperation of components of mixture are:
1.Physical state of constituents of the mixture.
2.Difference in one or more physical properties of the constituents of the mixture.
Separation of solid-solid mixture:
Different methods are employed for the separation of solid-solid mixture.
They are:
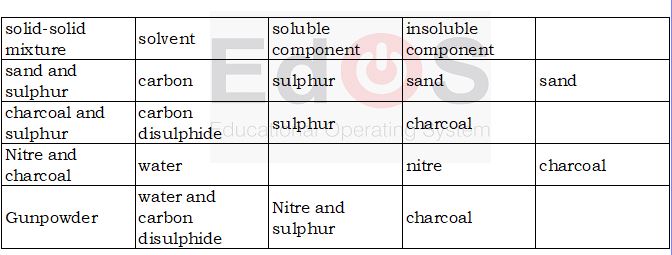
i) Solvent extraction:
This process is based on the property that one of the component is soluble in the solvent.
Ex: A mixture of sulphur and sand. Sulphur is soluble in CS2 and sand is insoluble. Based on solvents we can seperate mixture by using solubility. -
Solvent Extraction
i) Solvent extraction:
This process is based on the property that one of the component is soluble in the solvent.
Ex: A mixture of sulphur and sand. Sulphur is soluble in CS2 and sand is insoluble. Based on solvents we can seperate mixture by using solubility.
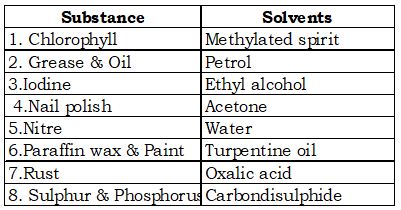
List of solvents:
In the above figure salt is soluble in water it is nothing but solubility.
-
Magnetic Separation and Gravity Separation
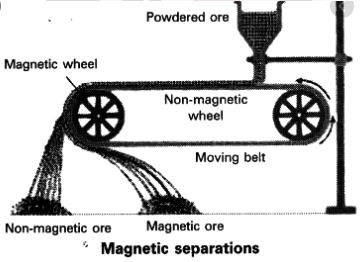
ii) Magnetic separation: This process is based on the magnetic property of one of the component.
Ex: Mixture of iron ore and sulphur. Iron ore is attracted by magnet and sulphur is left behind.Magnetic separation is the process of separating components of mixtures by using magnets to attract magnetic materials.[1] The process that is used for magnetic separation detaches non-magnetic material from magnetic.
Magnetic substances are Iron, Cobalt ,Nickel,Steel...
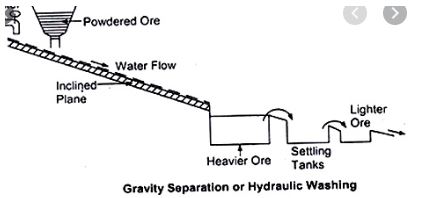
iii)Gravity separation: This process is based on the difference in the densities of components.
Ex: Mixture of sand and chalk powder. Sand being heavier than chalk sinks in water whereas chalk powder floats on water.
Among Sand and Saw dust,Sand is heavier and saw dust is lighter component. Among salt and sand ,Sand is heavier salt is lighter.
Among Charcoal and Limestone, Limestone is heavier and Charcoal is lighter. -
Sublimation and Fractional Crystallization
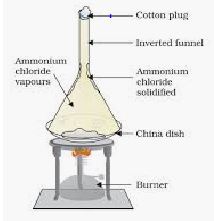
iv) Sublimation: This process is based on the ability of one component to sublime.
Ex: Mixture of iodine and sand. On heating iodine sublimes leaving behind sand. The vapours on cooling gives solid iodine.
*Sublimable solids are Ammonium chloride, Camphor, Iodine,Benzoic acid, Naphthalene.
Mixture is placed in a china dish and heated bylow flame.An inverted dry funnel is placed over china dish and its stem is closed with cotton wool.Sublimable solid in the mixture gets vapourised and condensed to the sides of funnel to form powder.
The residue left in china dish is non-sublimable solid.
v) Fractional crystallization: This process is based on the difference in the solubility of the components in the same solvent.
Ex: Mixture of potassium chlorate and potassium chloride.
Potassium chlorate is more soluble than potassium chloride when aqueous solution of this mixture is subjected to heating, more soluble potassium chlorate escapes along with water vapour and potassium chloride is left behind and crystallises out.
The process of seperation of two different soluble substances from their solution by crystallization at controlled temperature, such that one of the solid crystallises is called Fractional Crystallisation.
Ex: 1. Among Potassium nitrate and sodium chloride ,Potassium nitrate is more soluble and sodium chloride is less soluble component.
2. Among Sodium nitrate and Sodium chloride ,Sodium nitrate is more soluble and sodium chloride is less soluble.