-
Physical Classification Of Matter
Examples
Question:What is the general name of :
(a) rigid form of matter ?
(b) fluid forms of matter ?Solution: (a) Solid
(b) Liquid and Gas
Question: Which diffuses faster : a liquid or a gas ?
Solution: GasQuestion: We can get the smell of perfume sitting several metres away, comment.
Solution:Since gases diffuse rapidly, the particles of vapours of perfume (gas) diffuse or intermix with the particles of air (gas) rapidly and thus reach us sitting at some distance. Consequently we can get the smell of perfume sitting several metres away.Question:Arrange the following substances in increasing order of forces of attraction between particles – water, sugar, oxygen.
Solution:Water is a liquid, sugar is a solid & oxygen is a gas. The increasing order of forces of attraction between particles is : gas < liquid < solid.
Hence, increasing order of forces of attraction of particles is : oxygen < water < sugar
Question:Give two reasons to justify :
(a) Water at room temperature is a liquid
(b) An iron almirah is a solid at room temperature.
Solution: (a) Water at room temperature is a liquid because :
(i) It has a fixed volume but does not have a definite shape.
(ii) It can flow easily from one vessel to another, so it has fluidity.
(b) An iron almirah is a solid at room temperature because :
(i) It has a fixed volume and definite shape.
(ii) It can not be compressed and it has distinct boundaries.Question:The mass per unit volume of a substance is called density. Arrange the following in order of increasing density: air, exhaust from chimneys, honey, water, chalk, cotton and iron. Explain
Solution: The order of increasing density is :
air < exhaust from chimneys < water < honey < cotton < chalk < iron.
Explanation:
air, exhaust from chimney \( \to \) gases
water, honey \( \to \) liquids
cotton, chalk & Iron \( \to \) solids
Among air and exhaust from chimneys, the mass of exhaust from chimneys
(which contains some heavier gases like CO2, SO2, NO2 etc and minute solid particles) is much higher than air (which contains only O2 & N2 as main constituents). Therefore density of exhaust from chimneys is expected to be higher than that of air. Thus order of densities among different gases like air & exhaust from chimneys is - air<exhaust from chimneys.
·
Among honey and water, the mass of particles of honey is much higher and inter particle distances are much smaller than those in water. Therefore, the density of honey is higher than that of water. Thus order of densities among different liquids like water and honey is - water < honey
Among chalk and iron, the mass of the particles of iron is much higher and inter particle distances are much smaller than those in chalk (since particles of iron are very closely packed than of chalk) Therefore, density of iron is much higher than that of chalk. Cotton is also a solid, but its density is lower than that of other solids like chalk & iron due to spaces in which air is trapped. Thus order of densities among different solids like cotton, chalk & iron, is - cotton < chalk < iron.Order of densities among solids, liquids & gases :
Now since gases have larger inter particle distances than liquids, they have lower densities than liquids.
i.e. air < exhaust from chimneys < water < honey. But since liquids have larger inter particle distances than solids, they have lower densities than solids.
i,e water < honey < cotton < chalk < iron
Thus the overall order of increasing density is: air < exhaust from chimneys < water < honey < cotton < chalk < iron.
Question 11: Comment upon the following – Rigidity, compressibility, fluidity, kinetic energy and density
Solution: Rigidity: It means tendency to maintain shape when some outside force is applied. Solids possess high rigidity due to smaller inter particle distances and stronger inter particle forces of attraction.
Compressibility: It means tendency to decrease volume, when some outside force is applied. The gases possess high compressibility due to larger inter particle distances i.e. their volume decreases when some pressure is applied on them.
Fluidity: It means tendency to flow. Since gases have larger inter particle distances and weaker forces of attraction, they possess highest fluidity. Liquids also have tendency to flow but their fluidity is lower than that of gases due to comparatively smaller inter particle distances and relatively stronger forces of attraction
Kinetic Energy (K.E.): It means energy possessed by particles due to motion. The gases possess highest kinetic energy due to random motion of their particles (i.e the particles of a gas move in different directions with different velocities) because of larger inter particle distances & weaker forces of attraction.
Liquids have lower K.E. while solids have least K.E. at any particular temperature i.e. room temperature.Density: It means mass per unit volume. Since the particles of a solid are closely packed, those of liquids are less closely packed while those of gases are loosely packed, thus solids possess highest density, liquids possess lower density, while gases have the lowest density.
Question: Give Reasons :
(a) A gas completely fills the vessel in which it is kept?
(b) A gas exerts pressure on the walls of the container
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood, we need a karate expert.Solution:(a) A gas has neither a fixed shape nor a fixed volume. Particles of gas move freely in a random manner and assume the shape and the volume of the vessel in which it is kept.
(b) Due to random motion, the particles of a gas collide with each other and with the walls of the container in which gas is kept. The collisions of gas particles on the walls of the container exert a steady force and hence result in the pressure of the gas (because force per unit area is called the pressure of gas).
Thus, gases exert pressure due to the collisions of the particles of the gas on the walls of the containing vessel.(c) A wooden table has a definite shape, hence it is a solid.
(d) There are larger inter particle distances (or lot of empty spaces) and hence weaker forces of attraction between the particles of gas (i.e. air) as compared to solids. As a result, we can easily move our hand in air but can not do so in case of a solid block of wood. In other words we need, a karate expert with almost incredible power to separate the solid particles apart (due to stronger inter particle forces of attraction in solids)
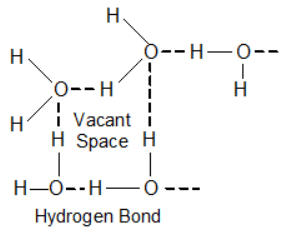
Question:Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why?Solution:The water in form of ice (solid) forms hydrogen bonds, due to which, some empty spaces are created between the molecules of water in ice. As a result, volume increases for the same mass of water in ice. In other words mass per unit volume or density of ice is less than that of water and hence ice floats on water.