-
Mole Concept
Mole Concept
Mole Concept
We shall first recap of concepts learnt in previous classes about Atoms, Molecules, Elements, Compounds and Mixtures.
- Atom: Atom is the smallest particle of an element which may or may not have free existence but participates in chemical changes on behalf of the element. It is represented by the symbol of the element.
Ex: Sodium atom (Na), Oxygen atom (O)
Sulphur atom (S), Ozone atom (O)
- Molecule: Molecule is the smallest particle of an element of a compound which has free existence. In chemical equations generally molecular formulae of the substances involved are written.
Note: For ionic compounds exact molecules are not possible due to non-directional electrostatic forces of attraction. Hence, for those compounds we take the smallest ratio of the number of oppoistively charged ions and frame the formula unit which sometimes is called the molecular formula, though it is technically wrong to state like that
- Element: The substance that contains same type of atoms and it cannot be split up into new substances by ordinary physical or chemical changes.
The term element is qualitative. An element may have atoms with different mass numbers known as isotopes, but due to nearly same properties and same atomic number they are given the same place in the periodic table (iso=same; topas = place). Knowing the % abundance of each isotope, the atomic mass of the element is calculated as the average of the masses of these isotopes.
If an element has three isotopes with mass numbers a,b,c and % abundance of the three isotopes are p,q and r respectively,
the atomic mass of the element = \( \frac{{{p \times a + q \times b + r \times c}}} {{100}} \)
- Compound: Compound is a substance formed by the chemical combination of different elements combined in a fixed proportion by masses.
Compounds in chemical equations are written through their molecular formulae.
For a compound if its moleculer formula is Ax By Cz then molecular mass (X × atomic mass of A+y × at mass of B+Z × atomic mass of C).
- Mixture: Mixture is obtained by the physical combination of two or more elements, compounds or elements and compound.
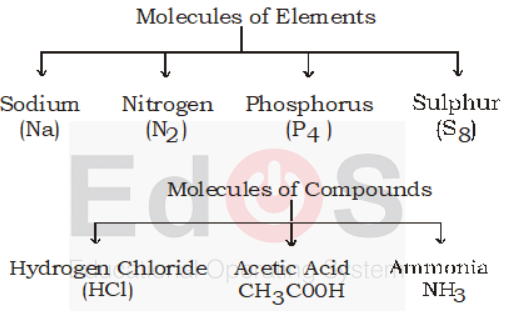
- Atomicity : Number of atoms present in one molecule of a substance is called atomicity.
i. Atomicity of inert gases is 1
ii. Elementary gases like oxygen, nitrogen, hydrogen etc are diatomic.
iii. Ozone is a triatomic gas.
iv. Phosphorous, Arsenic and antimony are tetraatomic.
v. Sulphur is octaatomic