-
PHYSICAL CLASSIFICATION OF MATTER
Matter may be defined as anything that occupies space, possesses mass and presence of which can be felt by any one or more of our five senses (i.e. sight, smell, taste, touch and hearing)
PHYSICAL NATURE OF MATTER
Particle Nature of Matter – Matter is made up of particles
To show the particle nature of matter, we perform the following experiment
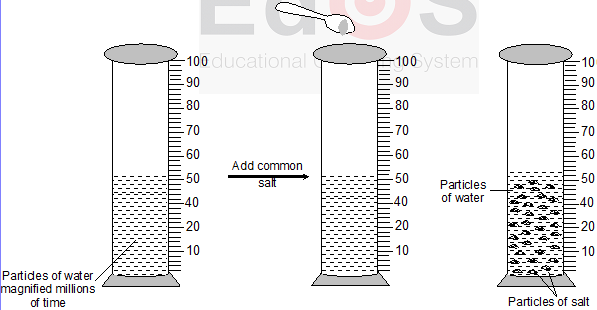
Experiment : Take about 50ml water in a graduated cylinder and dissolve small amount of common salt (NaCl) or sugar in it with the help of a glass rod
Observation and explanation: The salt or sugar dissolves in water and there is no noticeable change in the level of water. This is because, there are some spaces in between the particles of water, which are occupied by salt or sugar particles (when salt or sugar dissolves in water) and thus the level of water does not rise.
When salt dissolves in water, the particles of salt get into the spaces between the particles of water and the level of solution does not rise
Conclusion : From above experiment, we led to conclude that, there are some spaces between the particles of matter, or in other words matter is made up of particles.
Ø How small are these particles of matter?
To know how small are the particles of matter from which it is made up of, let us perform the experiment
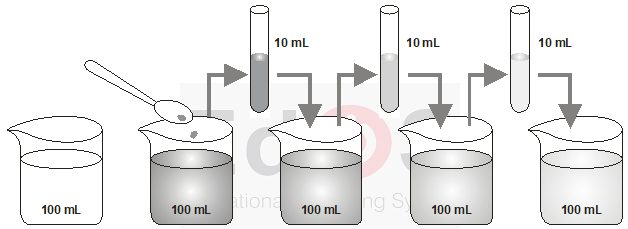
Experiment : Take about one crystal of potassium permanganate (KMnO4) and dissolve it in 100ml of water. The colour of solution will be dark pink. Take out approximate 10ml of this solution (dark pink) and put it into 90 ml of clear water. Now take 10ml of this solution and put it into another 90ml of clear water. Keep diluting the solution like this 5 – 8 times
Observation and Explanation : The pink colour will not disappear altogether, though it becomes lighter and lighter with each dilution. This is because, there must be millions of tiny particles present in one crystal of KMnO4 which keep on dividing into smaller and smaller number with each dilution, thereby making colour lighter and lighter.
Estimating how small are the particles of matter. With every dilution, though the colour becomes light, it is still visible
Conclusion : From above experiment we conclude that “matter is made up of extremely small particles which can not be seen even with a powerful microscope.
or The particles of matter are very small….. they are small beyond our imagination !!!!!.
The size of a particle of matter is of the order of nanometer, 1nm = 10m
CHARACTERISTICS OF PARTICLES OF MATTER
* Particles of matter have spaces between them
When potassium permanganate (KMnO4), dettol, sugar or salt are dissolved in water, then their particles get evenly distributed between the spaces present among the particles of water as discussed in above experiments. Similarly when we make tea, coffee or lemonade (nimbu pani), the particles of one type of matter get into the spaces between the particles of other type of matter.
This shows that there are spaces between particles of matter
*Particles of matter are continuously moving
The continuous motion of particles of matter can be explained more clearly by performing the following experiment
Experiment-1 : Put one unlit incense stick (Agarbati) in one room & one lit incense stick (Agarbati) in another room
Observation and Explanation : We will get smell while sitting at a distance from the lit stick, but to smell the unlit stick, we will have to go near it. This is because, when stick is lit, the temperature rises and hence the kinetic energy of the incense particles also increases. As a result, the particles of incense move rapidly and thus intermix with the particles of air rapidly so, we get smell of incense even when we are sitting at a distance.
On the other hand, when incense stick is not lit, temperature is low, and hence kinetic energy of incense particles is less. As a result particles of incense stick do not intermix with air rapidly, so that we have to go near the incense stick to get its smell (when it is not lit).
Conclusion: From above discussion we led to conclude that, particles of matter are never at rest, but are moving continuously. And their average speed increases with increase in temperature due to increase in kinetic energy of moving particles. As a result, rate of intermixing or rate of diffusion increases
Gas particles always keep on moving in a zig-zag manner. This movement is called Brownian movement.
Experiment-2: Drop a crystal of copper sulphate (CuSO4) or potassium permanganate (KMnO4) into a glass of hot water and another containing cold water. Allow the crystals to settle at the bottom without stirring the solution
Observation: The crystals of CuSO4 or KMnO4 dissolves more quickly in hot water than in cold water
Explanation: The particles of CuSO4 or KMnO4 crystals do not move and thus remain fixed in their respective positions due to strong forces of attraction. On the other hand, the particles of cold water are continuously moving and thus possess some kinetic energy. Because of their K.E., the particles of cold water overcome the forces of attraction between particles of CuSO4 or KMnO4 crystals. As a result, the particles of CuSO4 or KMnO4 crystals move in between the spaces of particles of cold water and the crystals of CuSO4 or KMnO4 dissolves in cold water.
As the temperature rises the K.E. of both. i.e. particles of CuSO4 or KMnO4 crystals and water increases. Due to greater K.E., the forces of attraction between particles of CuSO4 or KMnO4 crystals decrease. Further due to greater K.E., the particles of water (hot water) move faster and more easily overcome the weaker forces of attraction between particles of CuSO4 or KMnO4 crystals than cold water
As a result, the rate of intermixing (or rate of diffusion) increases and CuSO4 or KMnO4 crystals dissolves more quickly in hot water
Conclusion : From above discussion, we led to conclude that particles of matter are continuously moving and their average speed increases with increase in temperature due to increase in K.E. of moving particles. As a result, rate of diffusion becomes faster and hence solid dissolves more quickly in hot water than in cold water
Intermixing of particles of different types of matter on their own is called diffusion
Experiment-3 : Take two beakers filled with water and put a drop of blue or red ink slowly along the sides of the first beaker and honey in the same way in the second beaker. Keep them undisturbed for some time
Observation : The particles of ink quickly get distributed in water. As a result, colour of ink spreads throughout the water. On the other hand, particles of honey take a long time to. get distributed throughout the water
Explanation : The particles of ink move rapidly due to weak forces of attraction between them. As a result, the particles of ink rapidly get into the spaces between the particles of water and hence quickly get evenly distributed in water. In contrast, the particles of honey move slowly due to strong forces of attraction between them. As a result, it takes a long time for the particles of honey to get into the spaces between the particles of water and to get evenly distributed throughout water
Conclusion : From above discussion we led to conclude that particles are continuously moving but their average speed at any particular temperature depends upon the forces of attraction
Stronger the forces of attraction, lower is the average speed, and thus lower will be the rate of diffusion.
Particles of matter attract each other
The particles of matter have a force acting between them, which keeps these particles together. To illustrate this force of attraction, we perform the following experiments
Experiment-1: Take an iron nail, a piece of chalk and a rubber band. Try to break each one of them by hammering, cutting or stretching
Observation & Explanation : It is most difficult to break the iron nail, followed by piece of chalk & then rubber band.
This is because, the particles of iron nail are held together by the strongest forces followed by the piece of chalk, while particles of rubber band are held together by weakest forces of attraction
Conclusion : From above experiment, we conclude that “Particles of matter attract each other”. The strength of this force, however differs from one kind of matter to other
Experiment-2 : Try to break the stream of tap water with your fingers
Observation & Explanation : The stream can not be cut because particles of water attract each other strongly and hence tend to remain together
Conclusion : Particles of matter attract each other.
ILLUSTRATIONS
Question 1: Kitchen salt (NaCl) when added to water, gets evenly distributed in it. Which characteristic of particles of matter is exhibited by this
Solution: It shows that there is enough space between the particles of matter
Question 2: Define diffusion, is it faster in winter or summer season
Solution: The phenomenon of intermixing of particles of different types of matter, on their own, is called, diffusion. The rate of diffusion becomes faster with an increase in temperature due to increase in K.E. of moving particles. Hence diffusion is faster in summer than in winter
Question 3: When we light an incense stick (agarbatti) in a corner of our room, why does its fragrance spread in the whole room very quickly
Solution: When we light an incense stick, the temperature rises and hence kinetic energy of the incense particles also increases. As a result, the incense particles move rapidly and thus the rate of diffusion of incense particles with air particles becomes faster. That is why, fragrance of agarbatti (when we light it) spreads in the whole room very quickly.
Now since, the particles of hot vapours coming out of hot sizzling food move faster, therefore, they easily reach you even when you are several metres away. On the other hand, the particles of vapours coming out of cold food travel only slowly and hence do not reach you. Therefore, to get the smell from cold food, you have to go close to the food.
In short, due to diffusion, which becomes faster at higher temperature, vapours from hot sizzling food move faster and reach you several metres away. But you have to go close to get smell from cold food
The matter around us exists in three physical states on the basis of physical properties
Question 4: Give reasons for the following observations. The smell of hot sizzling food reaches when you are several meters away, but to get smell from the cold food, you have to go close to it
Solution: The particles of matter are continuously moving, but their average speed increases with increase in temperature due to increase in K.E. of moving particles.
(a) Water exists as ice (solid state), as liquid (liquid state) and as steam (gaseous state).
(b) Bones and teeth are solids, the blood that flows in our veins is a liquid and the air that we breathe in is a gas
The three physical states of matter (i.e solid, liquid or gaseous) arise due to variation in the characteristics of the particles of matter.SOLID STATE
Matter in solid state has a definite shape and definite volume.
Examples: Silver, copper, sand, sugar, gold, ice, wood, stone, book, needle, pencil, piece of thread, etc.
Properties of Solids
(a) Solids have a definite shape and distinct boundaries: The solids have a fixed shape and distinct boundaries due to small inter particle distances and strong forces of attraction. e.g. when a pen is put in different containers, it does not change its shape.
However, when sugar and salt, are placed in different containers, they take up the shape of the containers, yet they (sugar & salt) are solids. This is because, the shape of individual sugar or salt crystal remains fixed whether we take it in our hand, or put in a jar or in plate.
The highly ordered arrangement of constituent particles of a solid is called a lattice. This gives rise to a regular geometrical shape to the crystals.
(b) Solids possess rigidity: The solids have the tendency to maintain shape, when some outside force is applied (known as rigidity). They may break when dropped or hammered
However some solids like rubber band, changes its shape when stretched under influence of force, but it regains its original position, when force is withdrawn. However, if excessive force is applied, rubber band breaks
(c) Solids have a definite volume : Solids have a definite volume as they can not be compressed due to small inter particle distances.
However some solids like sponge can be easily compressed. This is because sponge has minute holes in which air is trapped so that when we press it, air is expelled and the sponge is compressed
(d) Solids do not possess the property of diffusion :- The solids do not have the property of diffusion into other solids (i.e. the particles of two solids do not intermix). This is because the particles of solid do not move much from their positions due to small inter particle distances and strong forces of attraction.
However particles of some solids like chalk have diffused into other solids like blackboard. i.e. if we write something on blackboard with the chalk and leave it uncleaned for sometime, we will find that it becomes difficult to clean the board. This is because of diffusion of chalk particles in between the particles of blackboard and hence it becomes difficult to rub them off.
LIQUID STATE
The matter in liquid state have a definite volume and no definite shape.
Examples : Water, blood, benzene, alcohol, milk, petrol, cooking oil, juice, cold drink etc
Properties of Liquids
(a) Liquid do not have fixed shape but have a fixed volume : The liquids have a fixed volume due to strong inter particle forces of attraction in them which are strong to keep the particles together.
But these forces are not strong enough to keep the particles in fixed position, therefore, liquids do not have a fixed shape, they take up the shape of vessel in which they are placed
(b) Liquids are not rigid but have a property to flow : Liquids can flow and change shape due to larger inter particle distances and weaker forces of attraction in them, than solids. Thus liquids are not rigid but they possess fluidity (i.e. they have property to flow)
Relative fluidity of liquids differ from one liquid to other. e.g. water flows faster than honey
(c) Liquids possess the property of Diffusion : Due to larger inter particle distances in liquids than in solids, the particles of a liquid have more freedom of motion than solids. Thus solids, liquids and gases all can diffuse into liquids as discussed below:
(i) Diffusion of solids into liquids: When a crystal of copper sulphate or potassium permanganate (solid) is added to water (liquid), the particles of CuSO4 or KMnO4 quickly diffuse in between the particles of water to form a solution.
(ii) Diffusion of liquids into liquids: When water is added to alcohol or vice-versa, the two liquids quickly diffuse into each other to form a solution
(iii) Diffusion of gases into liquids: Some gases especially O2 and CO2 diffuse into water i.e. dissolve in water. So that aquatic animals can breathe under water due to presence of dissolved oxygen in water
Thus solids, liquids & gases – all can diffuse into liquids. However, the rate of diffusion of liquids is much higher than that of solids
§ Rate of diffusion of different liquids :- Different liquids have different rates of diffusion. For example a drop of blue or red ink diffuses faster than a drop of honey into water
§ Rate of diffusion increase with rise in temperature :- Rate of diffusion increases with rise in temperature, hence sugar dissolves much more quickly in hot water than in cold water.
GASEOUS STATE
The matter in gaseous state has neither definite volume nor shape.
Examples : Air, oxygen, nitrogen, hydrogen, ammonia, carbon dioxide, compressed natural gas (CNG) etc
Properties of Gases
(a) Gases neither have a definite shape nor a definite volume : Gases do not have a definite shape, but they acquire the shape of the vessel in which they are placed.
Similarly, gases do not have a definite volume, but attain the volume of container to which they are transferred
(b) Gases have maximum fluidity and least rigidity : The gases have high fluidity (property to flow) and least rigidity (tendency to maintain shape) due to large inter particle space and weak inter particle forces of attraction in them
(c) Gases are highly compressible : The gases are highly compressible due to large inter particle spaces in them. Due to high compressibility, large volume of a gas can be compressed into a small cylinder and transported easily. e.g. L.P.G. gas & O2 supplied to hospitals in cylinders is compressed gas. Similarly these days, compressed natural gas (CNG) is used as a fuel in vehicles
Gases are highly compressible while liquids are almost incompressible, while solids are completely incompressible.
This can be explained by the following experiment
Experiment to illustrate comparison between solids, liquid and gases in terms of compressibility. or
Experiment to study the compressibility of solids, liquids & gases :
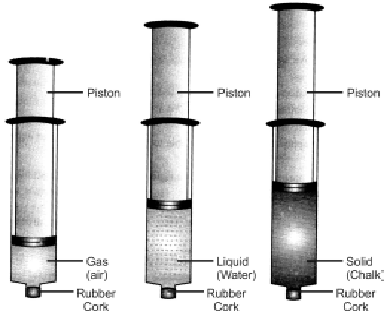
Take three syringes (about 100ml) and close their nozzles by rubber corks. Now remove the pistons from all syringes. Fill some water (liquid) in second syringe and chalk pieces (solid) in the third & leaving first syringe untouched. Now insert pistons back into syringes.
Observation and explanation: The piston of first syringe (left untouched) which contained air (gas) was easily pushed in. The piston of the second syringe which contained water (liquid) was pushed in only a little, while the piston of the third syringe which contained chalk pieces could not be pushed in at all. Thus, air is easily compressible, water is almost incompressible, while chalk pieces are completely incompressible
Conclusion: The spaces between particles of gases are maximum, intermediate in liquids and minimum in solids. Thus, gases are highly compressible, liquids are almost incompressible, while solids are completely incompressible.
(d) Gases have low density : Gases have low density as compared to solids and liquids due to large inter molecular spaces in them. i.e. mass per unit volume of a gas is small and hence gases have low density
(e) The Kinetic energy of particles in the gaseous state is quite high :- Due to large inter particle distances and weak forces of attraction, the particles of a gas can move freely & thus have large rotational, translational and vibrational motion and due to large translational motion, their kinetic energy is quite high which can be further increased by increasing the temperature of gas
(f) Gases exert pressure : Due to larger inter particle distances and weaker inter particle forces of attractions, particles of a gas are moving continuously in different directions with different velocities. Due to this random motion, the particles of gas collide with each other and also with the walls of the containing vessel. Due to these collisions, the particles of the gas exert a force on the walls of the container. This force per unit area exerted by the particles of the gas on the walls of containing vessel is called the pressure of the gas
Random motion means motion in different directions with different velocities. The random motion of particles of a gas is due to larger inter particle distances and weaker inter particle forces of attraction between them, unlike liquids & solid
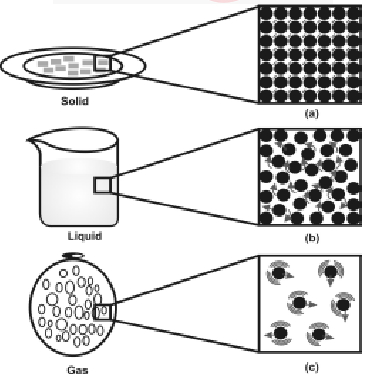
The motion and inter particle distances in solids, liquids & gases are as shown in fig.
a, b and c show the magnified schematic pictures of the three states of matter. The motion of the particles can be seen and compared in the three states of matter
(g) Gases diffuse very rapidly : Due to random motion, the particles of one gas readily move into spaces between the particles of the other gas. (called diffusion)
Thus, gases diffuse very rapidly, rate of diffusion increases with increase in temperature.
The most familiar example of diffusion of gases is found in our homes, e.g. we come to know what is cooked in the kitchen without even entering there, by the smell due to rapid diffusion. (i.e. rapid intermixing of particles of aroma with particles of air). Since rate of diffusion becomes faster at high temperature the smell of hot cooked food travels faster than that of the cold food
The rate of diffusion of a gas is inversely proportional to the square root of its density, this is called Graham’s law of diffusion
Differences in the characteristics of states of matter (solids, liquids & gases)
Plasma : Plasma (a mixture of free electrons and ions) is the fourth state of matter (e.g. fluorescent tube or neon sign
Plasma is an ionised gas, a gas to which sufficient energy is provided to get free electrons from atoms or molecules to allow both the species, ions and electrons to coexist. It exists in the presence of high temperature
Ex: The gases in the sun and stars exist in plasma state.
Bose – Einstein-condensate (BEC) : It is the fifth state of matter obtained by cooling extremely low density gas to super low temperature
Bose-Einstein condensate is a super fluid phase formed by atoms, cooled very near to absolute zero temperature. It is opposite to plasma state as it occurs at very low temperature.
Ex: First condensate was produced by Rubidium atoms (Rb) cooling to 170 nanokelvin
EXAMPLES
Question: What is the general name of
(a) rigid form of matter ?
(b) fluid forms of matter
Solution: (a) Solid (b) Liquid and Gas
Question: Which diffuses faster : a liquid or a gas ?
Solution: Gas
Question: We can get the smell of perfume sitting several metres away, comment.
Solution: Since gases diffuse rapidly, the particles of vapours of perfume (gas) diffuse or intermix with the particles of air (gas) rapidly and thus reach us sitting at some distance. Consequently we can get the smell of perfume sitting several metres away
Question: Arrange the following substances in increasing order of forces of attraction between particles – water, sugar, oxygen.
Solution: Water is a liquid, sugar is a solid & oxygen is a gas. The increasing order of forces of attraction between particles is : gas < liquid < solid.
Hence, increasing order of forces of attraction of particles is : oxygen < water < sugar
Question: Give two reasons to justify :
(a) Water at room temperature is a liquid
(b) An iron almirah is a solid at room temperature.
Solution: (a) Water at room temperature is a liquid because :
(i) It has a fixed volume but does not have a definite shape.
(ii) It can flow easily from one vessel to another, so it has fluidity.
(b) An iron almirah is a solid at room temperature because :
(i) It has a fixed volume and definite shape.
(ii) It can not be compressed and it has distinct boundaries
Question: The mass per unit volume of a substance is called density. Arrange the following in order of increasing density:
air, exhaust from chimneys, honey, water, chalk, cotton and iron. Explai
Solution: The order of increasing density is :
air < exhaust from chimneys < water < honey < cotton < chalk < iron.
Explanation:
air, exhaust from chimney \(\to\) gases
water, honey \(\to\) liquids
cotton, chalk & Iron \(\to\) solids
Among air and exhaust from chimneys, the mass of exhaust from chimneys
(which contains some heavier gases like CO2, SO2, NO2 etc and minute solid particles) is much higher than air (which contains only O2 & N2 as main constituents). Therefore density of exhaust from chimneys is expected to be higher than that of air. Thus order of densities among different gases like air & exhaust from chimneys is - air ·Among honey and water, the mass of particles of honey is much higher and inter particle distances are much smaller than those in water. Therefore, the density of honey is higher than that of water. Thus order of densities among different liquids like water and honey is - water < honey
Among chalk and iron, the mass of the particles of iron is much higher and inter particle distances are much smaller than those in chalk (since particles of iron are very closely packed than of chalk) Therefore, density of iron is much higher than that of chalk. Cotton is also a solid, but its density is lower than that of other solids like chalk & iron due to spaces in which air is trapped. Thus order of densities among different solids like cotton, chalk & iron, is - cotton < chalk < iron
Order of densities among solids, liquids & gases :
Now since gases have larger inter particle distances than liquids, they have lower densities than liquids.
i.e. air < exhaust from chimneys < water < honey. But since liquids have larger inter particle distances than solids, they have lower densities than solids.
i,e water < honey < cotton < chalk < iron
Thus the overall order of increasing density is: air < exhaust from chimneys < water < honey < cotton < chalk < iron
Question 11: Comment upon the following – Rigidity, compressibility, fluidity, kinetic energy and densit
Solution: Rigidity: It means tendency to maintain shape when some outside force is applied. Solids possess high rigidity due to smaller inter particle distances and stronger inter particle forces of attraction
Compressibility: It means tendency to decrease volume, when some outside force is applied. The gases possess high compressibility due to larger inter particle distances i.e. their volume decreases when some pressure is applied on them
Fluidity: It means tendency to flow. Since gases have larger inter particle distances and weaker forces of attraction, they possess highest fluidity. Liquids also have tendency to flow but their fluidity is lower than that of gases due to comparatively smaller inter particle distances and relatively stronger forces of attraction
Kinetic Energy (K.E.): It means energy possessed by particles due to motion. The gases possess highest kinetic energy due to random motion of their particles (i.e the particles of a gas move in different directions with different velocities) because of larger inter particle distances & weaker forces of attraction.
Liquids have lower K.E. while solids have least K.E. at any particular temperature i.e. room temperature
Density: It means mass per unit volume. Since the particles of a solid are closely packed, those of liquids are less closely packed while those of gases are loosely packed, thus solids possess highest density, liquids possess lower density, while gases have the lowest density.
Question: Give Reasons :
(a) A gas completely fills the vessel in which it is kept?
(b) A gas exerts pressure on the walls of the container?
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood, we need a karate expert
Solution: (a) A gas has neither a fixed shape nor a fixed volume. Particles of gas move freely in a random manner and assume the shape and the volume of the vessel in which it is kept
(b) Due to random motion, the particles of a gas collide with each other and with the walls of the container in which gas is kept. The collisions of gas particles on the walls of the container exert a steady force and hence result in the pressure of the gas (because force per unit area is called the pressure of gas).
Thus, gases exert pressure due to the collisions of the particles of the gas on the walls of the containing vessel
(c) A wooden table has a definite shape, hence it is a solid.
(d) There are larger inter particle distances (or lot of empty spaces) and hence weaker forces of attraction between the particles of gas (i.e. air) as compared to solids. As a result, we can easily move our hand in air but can not do so in case of a solid block of wood. In other words we need, a karate expert with almost incredible power to separate the solid particles apart (due to stronger inter particle forces of attraction in solids)
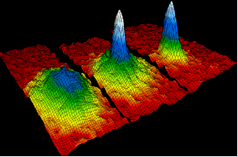
Question: Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why?
Solution: The water in form of ice (solid) forms hydrogen bonds, due to which, some empty spaces are created between the molecules of water in ice. As a result, volume increases for the same mass of water in ice. In other words mass per unit volume or density of ice is less than that of water and hence ice floats on water.
-
PHYSICAL CLASSIFICATION OF MATTER
Matter may be defined as anything that occupies space, possesses mass and presence of which can be felt by any one or more of our five senses (i.e. sight, smell, taste, touch and hearing)
PHYSICAL NATURE OF MATTER
Particle Nature of Matter – Matter is made up of particles
To show the particle nature of matter, we perform the following experiment
Experiment : Take about 50ml water in a graduated cylinder and dissolve small amount of common salt (NaCl) or sugar in it with the help of a glass rod
Observation and explanation: The salt or sugar dissolves in water and there is no noticeable change in the level of water. This is because, there are some spaces in between the particles of water, which are occupied by salt or sugar particles (when salt or sugar dissolves in water) and thus the level of water does not rise.
When salt dissolves in water, the particles of salt get into the spaces between the particles of water and the level of solution does not rise
Conclusion : From above experiment, we led to conclude that, there are some spaces between the particles of matter, or in other words matter is made up of particles.
Ø How small are these particles of matter?
To know how small are the particles of matter from which it is made up of, let us perform the experiment
Experiment : Take about one crystal of potassium permanganate (KMnO4) and dissolve it in 100ml of water. The colour of solution will be dark pink. Take out approximate 10ml of this solution (dark pink) and put it into 90 ml of clear water. Now take 10ml of this solution and put it into another 90ml of clear water. Keep diluting the solution like this 5 – 8 times
Observation and Explanation : The pink colour will not disappear altogether, though it becomes lighter and lighter with each dilution. This is because, there must be millions of tiny particles present in one crystal of KMnO4 which keep on dividing into smaller and smaller number with each dilution, thereby making colour lighter and lighter.
Estimating how small are the particles of matter. With every dilution, though the colour becomes light, it is still visible
Conclusion : From above experiment we conclude that “matter is made up of extremely small particles which can not be seen even with a powerful microscope.
or The particles of matter are very small….. they are small beyond our imagination !!!!!.
The size of a particle of matter is of the order of nanometer, 1nm = \(10^{-9}m\)
CHARACTERISTICS OF PARTICLES OF MATTER
* Particles of matter have spaces between them
When potassium permanganate (KMnO4), dettol, sugar or salt are dissolved in water, then their particles get evenly distributed between the spaces present among the particles of water as discussed in above experiments. Similarly when we make tea, coffee or lemonade (nimbu pani), the particles of one type of matter get into the spaces between the particles of other type of matter.
This shows that there are spaces between particles of matter
*Particles of matter are continuously moving
The continuous motion of particles of matter can be explained more clearly by performing the following experiment
Experiment-1 : Put one unlit incense stick (Agarbati) in one room & one lit incense stick (Agarbati) in another room
Observation and Explanation : We will get smell while sitting at a distance from the lit stick, but to smell the unlit stick, we will have to go near it. This is because, when stick is lit, the temperature rises and hence the kinetic energy of the incense particles also increases. As a result, the particles of incense move rapidly and thus intermix with the particles of air rapidly so, we get smell of incense even when we are sitting at a distance.
On the other hand, when incense stick is not lit, temperature is low, and hence kinetic energy of incense particles is less. As a result particles of incense stick do not intermix with air rapidly, so that we have to go near the incense stick to get its smell (when it is not lit).
Conclusion: From above discussion we led to conclude that, particles of matter are never at rest, but are moving continuously. And their average speed increases with increase in temperature due to increase in kinetic energy of moving particles. As a result, rate of intermixing or rate of diffusion increases
Gas particles always keep on moving in a zig-zag manner. This movement is called Brownian movement.
Experiment-2: Drop a crystal of copper sulphate (CuSO4) or potassium permanganate (KMnO4) into a glass of hot water and another containing cold water. Allow the crystals to settle at the bottom without stirring the solution
Observation: The crystals of CuSO4 or KMnO4 dissolves more quickly in hot water than in cold water
Explanation: The particles of CuSO4 or KMnO4 crystals do not move and thus remain fixed in their respective positions due to strong forces of attraction. On the other hand, the particles of cold water are continuously moving and thus possess some kinetic energy. Because of their K.E., the particles of cold water overcome the forces of attraction between particles of CuSO4 or KMnO4 crystals. As a result, the particles of CuSO4 or KMnO4 crystals move in between the spaces of particles of cold water and the crystals of CuSO4 or KMnO4 dissolves in cold water.
As the temperature rises the K.E. of both. i.e. particles of CuSO4 or KMnO4 crystals and water increases. Due to greater K.E., the forces of attraction between particles of CuSO4 or KMnO4 crystals decrease. Further due to greater K.E., the particles of water (hot water) move faster and more easily overcome the weaker forces of attraction between particles of CuSO4 or KMnO4 crystals than cold water
As a result, the rate of intermixing (or rate of diffusion) increases and CuSO4 or KMnO4 crystals dissolves more quickly in hot water
Conclusion : From above discussion, we led to conclude that particles of matter are continuously moving and their average speed increases with increase in temperature due to increase in K.E. of moving particles. As a result, rate of diffusion becomes faster and hence solid dissolves more quickly in hot water than in cold water
Intermixing of particles of different types of matter on their own is called diffusion
Experiment-3 : Take two beakers filled with water and put a drop of blue or red ink slowly along the sides of the first beaker and honey in the same way in the second beaker. Keep them undisturbed for some time
Observation : The particles of ink quickly get distributed in water. As a result, colour of ink spreads throughout the water. On the other hand, particles of honey take a long time to. get distributed throughout the water
Explanation : The particles of ink move rapidly due to weak forces of attraction between them. As a result, the particles of ink rapidly get into the spaces between the particles of water and hence quickly get evenly distributed in water. In contrast, the particles of honey move slowly due to strong forces of attraction between them. As a result, it takes a long time for the particles of honey to get into the spaces between the particles of water and to get evenly distributed throughout water
Conclusion : From above discussion we led to conclude that particles are continuously moving but their average speed at any particular temperature depends upon the forces of attraction
Stronger the forces of attraction, lower is the average speed, and thus lower will be the rate of diffusion.
Particles of matter attract each other
The particles of matter have a force acting between them, which keeps these particles together. To illustrate this force of attraction, we perform the following experiments
Experiment-1: Take an iron nail, a piece of chalk and a rubber band. Try to break each one of them by hammering, cutting or stretching
Observation & Explanation : It is most difficult to break the iron nail, followed by piece of chalk & then rubber band.
This is because, the particles of iron nail are held together by the strongest forces followed by the piece of chalk, while particles of rubber band are held together by weakest forces of attraction
Conclusion : From above experiment, we conclude that “Particles of matter attract each other”. The strength of this force, however differs from one kind of matter to other
Experiment-2 : Try to break the stream of tap water with your fingers
Observation & Explanation : The stream can not be cut because particles of water attract each other strongly and hence tend to remain together
Conclusion : Particles of matter attract each other.
ILLUSTRATIONS
Question 1: Kitchen salt (NaCl) when added to water, gets evenly distributed in it. Which characteristic of particles of matter is exhibited by this
Solution: It shows that there is enough space between the particles of matter
Question 2: Define diffusion, is it faster in winter or summer season
Solution: The phenomenon of intermixing of particles of different types of matter, on their own, is called, diffusion. The rate of diffusion becomes faster with an increase in temperature due to increase in K.E. of moving particles. Hence diffusion is faster in summer than in winter
Question 3: When we light an incense stick (agarbatti) in a corner of our room, why does its fragrance spread in the whole room very quickly
Solution: When we light an incense stick, the temperature rises and hence kinetic energy of the incense particles also increases. As a result, the incense particles move rapidly and thus the rate of diffusion of incense particles with air particles becomes faster. That is why, fragrance of agarbatti (when we light it) spreads in the whole room very quickly.
Now since, the particles of hot vapours coming out of hot sizzling food move faster, therefore, they easily reach you even when you are several metres away. On the other hand, the particles of vapours coming out of cold food travel only slowly and hence do not reach you. Therefore, to get the smell from cold food, you have to go close to the food.
In short, due to diffusion, which becomes faster at higher temperature, vapours from hot sizzling food move faster and reach you several metres away. But you have to go close to get smell from cold food
The matter around us exists in three physical states on the basis of physical properties
Question 4: Give reasons for the following observations. The smell of hot sizzling food reaches when you are several meters away, but to get smell from the cold food, you have to go close to it
Solution: The particles of matter are continuously moving, but their average speed increases with increase in temperature due to increase in K.E. of moving particles.
(a) Water exists as ice (solid state), as liquid (liquid state) and as steam (gaseous state).
(b) Bones and teeth are solids, the blood that flows in our veins is a liquid and the air that we breathe in is a gas
The three physical states of matter (i.e solid, liquid or gaseous) arise due to variation in the characteristics of the particles of matter.SOLID STATE
Matter in solid state has a definite shape and definite volume.
Examples: Silver, copper, sand, sugar, gold, ice, wood, stone, book, needle, pencil, piece of thread, etc.
Properties of Solids
(a) Solids have a definite shape and distinct boundaries: The solids have a fixed shape and distinct boundaries due to small inter particle distances and strong forces of attraction. e.g. when a pen is put in different containers, it does not change its shape.
However, when sugar and salt, are placed in different containers, they take up the shape of the containers, yet they (sugar & salt) are solids. This is because, the shape of individual sugar or salt crystal remains fixed whether we take it in our hand, or put in a jar or in plate.
The highly ordered arrangement of constituent particles of a solid is called a lattice. This gives rise to a regular geometrical shape to the crystals.
(b) Solids possess rigidity: The solids have the tendency to maintain shape, when some outside force is applied (known as rigidity). They may break when dropped or hammered
However some solids like rubber band, changes its shape when stretched under influence of force, but it regains its original position, when force is withdrawn. However, if excessive force is applied, rubber band breaks
(c) Solids have a definite volume : Solids have a definite volume as they can not be compressed due to small inter particle distances.
However some solids like sponge can be easily compressed. This is because sponge has minute holes in which air is trapped so that when we press it, air is expelled and the sponge is compressed
(d) Solids do not possess the property of diffusion :- The solids do not have the property of diffusion into other solids (i.e. the particles of two solids do not intermix). This is because the particles of solid do not move much from their positions due to small inter particle distances and strong forces of attraction.
However particles of some solids like chalk have diffused into other solids like blackboard. i.e. if we write something on blackboard with the chalk and leave it uncleaned for sometime, we will find that it becomes difficult to clean the board. This is because of diffusion of chalk particles in between the particles of blackboard and hence it becomes difficult to rub them off.
LIQUID STATE
The matter in liquid state have a definite volume and no definite shape.
Examples : Water, blood, benzene, alcohol, milk, petrol, cooking oil, juice, cold drink etc
Properties of Liquids
(a) Liquid do not have fixed shape but have a fixed volume : The liquids have a fixed volume due to strong inter particle forces of attraction in them which are strong to keep the particles together.
But these forces are not strong enough to keep the particles in fixed position, therefore, liquids do not have a fixed shape, they take up the shape of vessel in which they are placed
(b) Liquids are not rigid but have a property to flow : Liquids can flow and change shape due to larger inter particle distances and weaker forces of attraction in them, than solids. Thus liquids are not rigid but they possess fluidity (i.e. they have property to flow)
Relative fluidity of liquids differ from one liquid to other. e.g. water flows faster than honey
(c) Liquids possess the property of Diffusion : Due to larger inter particle distances in liquids than in solids, the particles of a liquid have more freedom of motion than solids. Thus solids, liquids and gases all can diffuse into liquids as discussed below:
(i) Diffusion of solids into liquids: When a crystal of copper sulphate or potassium permanganate (solid) is added to water (liquid), the particles of CuSO4 or KMnO4 quickly diffuse in between the particles of water to form a solution.
(ii) Diffusion of liquids into liquids: When water is added to alcohol or vice-versa, the two liquids quickly diffuse into each other to form a solution
(iii) Diffusion of gases into liquids: Some gases especially O2 and CO2 diffuse into water i.e. dissolve in water. So that aquatic animals can breathe under water due to presence of dissolved oxygen in water
Thus solids, liquids & gases – all can diffuse into liquids. However, the rate of diffusion of liquids is much higher than that of solids
§ Rate of diffusion of different liquids :- Different liquids have different rates of diffusion. For example a drop of blue or red ink diffuses faster than a drop of honey into water
§ Rate of diffusion increase with rise in temperature :- Rate of diffusion increases with rise in temperature, hence sugar dissolves much more quickly in hot water than in cold water.
GASEOUS STATE
The matter in gaseous state has neither definite volume nor shape.
Examples : Air, oxygen, nitrogen, hydrogen, ammonia, carbon dioxide, compressed natural gas (CNG) etc
Properties of Gases
(a) Gases neither have a definite shape nor a definite volume : Gases do not have a definite shape, but they acquire the shape of the vessel in which they are placed.
Similarly, gases do not have a definite volume, but attain the volume of container to which they are transferred
(b) Gases have maximum fluidity and least rigidity : The gases have high fluidity (property to flow) and least rigidity (tendency to maintain shape) due to large inter particle space and weak inter particle forces of attraction in them
(c) Gases are highly compressible : The gases are highly compressible due to large inter particle spaces in them. Due to high compressibility, large volume of a gas can be compressed into a small cylinder and transported easily. e.g. L.P.G. gas & O2 supplied to hospitals in cylinders is compressed gas. Similarly these days, compressed natural gas (CNG) is used as a fuel in vehicles
Gases are highly compressible while liquids are almost incompressible, while solids are completely incompressible.
This can be explained by the following experiment
Experiment to illustrate comparison between solids, liquid and gases in terms of compressibility. or
Experiment to study the compressibility of solids, liquids & gases :
Take three syringes (about 100ml) and close their nozzles by rubber corks. Now remove the pistons from all syringes. Fill some water (liquid) in second syringe and chalk pieces (solid) in the third & leaving first syringe untouched. Now insert pistons back into syringes.
Observation and explanation: The piston of first syringe (left untouched) which contained air (gas) was easily pushed in. The piston of the second syringe which contained water (liquid) was pushed in only a little, while the piston of the third syringe which contained chalk pieces could not be pushed in at all. Thus, air is easily compressible, water is almost incompressible, while chalk pieces are completely incompressible
Conclusion: The spaces between particles of gases are maximum, intermediate in liquids and minimum in solids. Thus, gases are highly compressible, liquids are almost incompressible, while solids are completely incompressible.
(d) Gases have low density : Gases have low density as compared to solids and liquids due to large inter molecular spaces in them. i.e. mass per unit volume of a gas is small and hence gases have low density
(e) The Kinetic energy of particles in the gaseous state is quite high :- Due to large inter particle distances and weak forces of attraction, the particles of a gas can move freely & thus have large rotational, translational and vibrational motion and due to large translational motion, their kinetic energy is quite high which can be further increased by increasing the temperature of gas
(f) Gases exert pressure : Due to larger inter particle distances and weaker inter particle forces of attractions, particles of a gas are moving continuously in different directions with different velocities. Due to this random motion, the particles of gas collide with each other and also with the walls of the containing vessel. Due to these collisions, the particles of the gas exert a force on the walls of the container. This force per unit area exerted by the particles of the gas on the walls of containing vessel is called the pressure of the gas
Random motion means motion in different directions with different velocities. The random motion of particles of a gas is due to larger inter particle distances and weaker inter particle forces of attraction between them, unlike liquids & solid
The motion and inter particle distances in solids, liquids & gases are as shown in fig.
a, b and c show the magnified schematic pictures of the three states of matter. The motion of the particles can be seen and compared in the three states of matter
(g) Gases diffuse very rapidly : Due to random motion, the particles of one gas readily move into spaces between the particles of the other gas. (called diffusion)
Thus, gases diffuse very rapidly, rate of diffusion increases with increase in temperature.
The most familiar example of diffusion of gases is found in our homes, e.g. we come to know what is cooked in the kitchen without even entering there, by the smell due to rapid diffusion. (i.e. rapid intermixing of particles of aroma with particles of air). Since rate of diffusion becomes faster at high temperature the smell of hot cooked food travels faster than that of the cold food
The rate of diffusion of a gas is inversely proportional to the square root of its density, this is called Graham’s law of diffusion
Differences in the characteristics of states of matter (solids, liquids & gases)
Plasma : Plasma (a mixture of free electrons and ions) is the fourth state of matter (e.g. fluorescent tube or neon sign
Plasma is an ionised gas, a gas to which sufficient energy is provided to get free electrons from atoms or molecules to allow both the species, ions and electrons to coexist. It exists in the presence of high temperature
Ex: The gases in the sun and stars exist in plasma state.
Bose – Einstein-condensate (BEC) : It is the fifth state of matter obtained by cooling extremely low density gas to super low temperature
Bose-Einstein condensate is a super fluid phase formed by atoms, cooled very near to absolute zero temperature. It is opposite to plasma state as it occurs at very low temperature.
Ex: First condensate was produced by Rubidium atoms (Rb) cooling to 170 nanokelvin
EXAMPLES
Question: What is the general name of
(a) rigid form of matter ?
(b) fluid forms of matter
Solution: (a) Solid (b) Liquid and Gas
Question: Which diffuses faster : a liquid or a gas ?
Solution: Gas
Question: We can get the smell of perfume sitting several metres away, comment.
Solution: Since gases diffuse rapidly, the particles of vapours of perfume (gas) diffuse or intermix with the particles of air (gas) rapidly and thus reach us sitting at some distance. Consequently we can get the smell of perfume sitting several metres away
Question: Arrange the following substances in increasing order of forces of attraction between particles – water, sugar, oxygen.
Solution: Water is a liquid, sugar is a solid & oxygen is a gas. The increasing order of forces of attraction between particles is : gas < liquid < solid.
Hence, increasing order of forces of attraction of particles is : oxygen < water < sugar
Question: Give two reasons to justify :
(a) Water at room temperature is a liquid
(b) An iron almirah is a solid at room temperature.
Solution: (a) Water at room temperature is a liquid because :
(i) It has a fixed volume but does not have a definite shape.
(ii) It can flow easily from one vessel to another, so it has fluidity.
(b) An iron almirah is a solid at room temperature because :
(i) It has a fixed volume and definite shape.
(ii) It can not be compressed and it has distinct boundaries
Question: The mass per unit volume of a substance is called density. Arrange the following in order of increasing density:
air, exhaust from chimneys, honey, water, chalk, cotton and iron. Explai
Solution: The order of increasing density is :
air < exhaust from chimneys < water < honey < cotton < chalk < iron.
Explanation:
air, exhaust from chimney \(\to\) gases
water, honey \(\to\) liquids
cotton, chalk & Iron \(\to\) solids
Among air and exhaust from chimneys, the mass of exhaust from chimneys
(which contains some heavier gases like CO2, SO2, NO2 etc and minute solid particles) is much higher than air (which contains only O2 & N2 as main constituents). Therefore density of exhaust from chimneys is expected to be higher than that of air. Thus order of densities among different gases like air & exhaust from chimneys is - air ·Among honey and water, the mass of particles of honey is much higher and inter particle distances are much smaller than those in water. Therefore, the density of honey is higher than that of water. Thus order of densities among different liquids like water and honey is - water < honey
Among chalk and iron, the mass of the particles of iron is much higher and inter particle distances are much smaller than those in chalk (since particles of iron are very closely packed than of chalk) Therefore, density of iron is much higher than that of chalk. Cotton is also a solid, but its density is lower than that of other solids like chalk & iron due to spaces in which air is trapped. Thus order of densities among different solids like cotton, chalk & iron, is - cotton < chalk < iron
Order of densities among solids, liquids & gases :
Now since gases have larger inter particle distances than liquids, they have lower densities than liquids.
i.e. air < exhaust from chimneys < water < honey. But since liquids have larger inter particle distances than solids, they have lower densities than solids.
i,e water < honey < cotton < chalk < iron
Thus the overall order of increasing density is: air < exhaust from chimneys < water < honey < cotton < chalk < iron
Question 11: Comment upon the following – Rigidity, compressibility, fluidity, kinetic energy and densit
Solution: Rigidity: It means tendency to maintain shape when some outside force is applied. Solids possess high rigidity due to smaller inter particle distances and stronger inter particle forces of attraction
Compressibility: It means tendency to decrease volume, when some outside force is applied. The gases possess high compressibility due to larger inter particle distances i.e. their volume decreases when some pressure is applied on them
Fluidity: It means tendency to flow. Since gases have larger inter particle distances and weaker forces of attraction, they possess highest fluidity. Liquids also have tendency to flow but their fluidity is lower than that of gases due to comparatively smaller inter particle distances and relatively stronger forces of attraction
Kinetic Energy (K.E.): It means energy possessed by particles due to motion. The gases possess highest kinetic energy due to random motion of their particles (i.e the particles of a gas move in different directions with different velocities) because of larger inter particle distances & weaker forces of attraction.
Liquids have lower K.E. while solids have least K.E. at any particular temperature i.e. room temperature
Density: It means mass per unit volume. Since the particles of a solid are closely packed, those of liquids are less closely packed while those of gases are loosely packed, thus solids possess highest density, liquids possess lower density, while gases have the lowest density.
Question: Give Reasons :
(a) A gas completely fills the vessel in which it is kept?
(b) A gas exerts pressure on the walls of the container?
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood, we need a karate expert
Solution: (a) A gas has neither a fixed shape nor a fixed volume. Particles of gas move freely in a random manner and assume the shape and the volume of the vessel in which it is kept
(b) Due to random motion, the particles of a gas collide with each other and with the walls of the container in which gas is kept. The collisions of gas particles on the walls of the container exert a steady force and hence result in the pressure of the gas (because force per unit area is called the pressure of gas).
Thus, gases exert pressure due to the collisions of the particles of the gas on the walls of the containing vessel
(c) A wooden table has a definite shape, hence it is a solid.
(d) There are larger inter particle distances (or lot of empty spaces) and hence weaker forces of attraction between the particles of gas (i.e. air) as compared to solids. As a result, we can easily move our hand in air but can not do so in case of a solid block of wood. In other words we need, a karate expert with almost incredible power to separate the solid particles apart (due to stronger inter particle forces of attraction in solids)
Question: Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why?
Solution: The water in form of ice (solid) forms hydrogen bonds, due to which, some empty spaces are created between the molecules of water in ice. As a result, volume increases for the same mass of water in ice. In other words mass per unit volume or density of ice is less than that of water and hence ice floats on water.