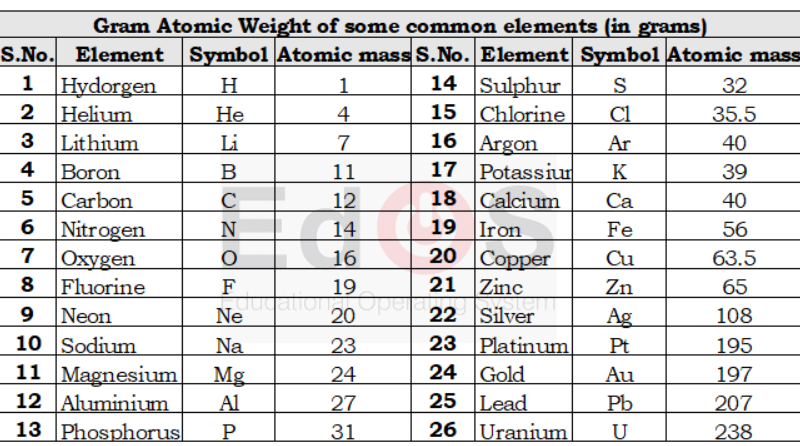
-
Atomic Weight And Molecular Weight
Atomic Weight And Molecular Weight
Relation between atomic weight in amu and Gram Atomic Weight
Lets consider an example
Based on the experimental results on weighing 1 atom of Aluminium
Weight of 1 atom of Aluminimum = 27 amu
Weight of 1 atom of Aluminium in grams = 27 X 1.66 X 10-24 grams
( 1 amu = 1.66 X 10-24 grams )
Now we shall calculate the weight of 1 mole (6.023 X 1023) of Aluminium atoms
= 6.023 X 1023 X 27 X 1.66 X 10-24 grams
= 27 grams
From the above example we can conclude that
Weight of 1 Aluminium atom = 27 amu
Weight of 1 mole of Aluminium atom = 27 grams
Therefore we can state that, when an atom weight is expressed in “amu” we are taking the weight of 1 atom and when an atom weight is expressed in “grams” we are taking the weight of 1 mole of atoms
Therefore when 1mole of atoms are weight we get atomic weight in grams which is knows as “Gram Atomic Weight”
Defnition: Atomic Weight expressed in grams is known as “Gram Atomic Weight”
Gram atomic weight contains one mole of atoms.
Gram atom : One gram atomic weight of a substance is called gram atom. It is equal to the weight of Avogadro no. of atoms.Formula to calculate Number of Gram atoms
No.of gram atoms or number of moles of atoms
=\( \frac{{weight of the substance in grams}} {{gram atomic weight of the substance}} \)
Ex :- No. of gram atoms in 1 kg of Helium =\( \frac{{1000}} {4} = 250 \)