-
DISPALCEMENT AND DOUBLE DECOMPOSITION REACTIONS
DISPALCEMENT AND DOUBLE DECOMPOSITION REACTIONS
DOUBLE DISPLACEMENT REACTIONS
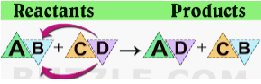
A double displacement reaction is a type of reaction in which two reactants exchange ions to form two new compounds. Double displacement reactions typically result in the formation of a product that is a precipitate Double displacement reactions take the form
AB(aq) + CD(aq) \(\to\) AD (aq)+ CB(aq).
EXAMPLES
The reaction between silver nitrate and sodium chloride is a double displacement reaction. The silver trades its nitrite ion for the sodium's chloride ion, causing the sodium to pick up the nitrate anion.This is precipitation reaction where an insoluble solid AgCl is formed.
AgNO3 + NaCl \(\to\) AgCl \(\downarrow \) + NaNO3
BaCl2(aq) + Na2SO4(aq) \(\to\) BaSO4(s)\(\downarrow \) + 2 NaCl(aq)
EXPERIMENT
Let us perform an activity using Silver nitrate, Sodium chloride to understand the chemical double displacement reactions as follows-
Activity : Experiment:
Apparatus: Two test tubes.
Chemicals: Silver nitrate, Sodium chloride, Water.
Procedure: By taking a small qunatity of silver nitrate in a test - tube and adding sufficient water, prepare silver nitrate solution. Likewise prepare sodium chlo
ride solution by taking a small quantity of sodium chloride in another test -tube and adding sufficient amount of water.Now add silver nitrate solution gradually to the sodium chloride solution.
Observation: Formation of curd - like precipitate in the test - tube containing silver chloride is observed
Conclusion: Silver nitrate and Sodium chloride are two different compounds. During chemical reaction, the constituents(ions) of the compounds get mutually exchanged. As a result,silverchloride is formed as a precipitate and sodium nitrate as a solution.
Chemical Equation:
NaCl + AgNO3 AgCl + NaNO3
Sodium chloride + Silver nitrate silverchloride + sodium nitrate(aq)
After the completion of a double displacement reaction,one of the products is usually
1) a solid precipitate,
2) a gas, or
3) a molecular compound such as water.