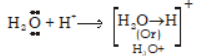-
Acids, Bases And Salts - Theories
Lewis or Electronic theory of acids and bases
According to Lewis theory, an acid is any molecule, ion or atom which can accept a pair of electron to form a co-ordinate covalent bond. Example: H+, BF3.
Base is a substance which can donate a pair of electrons to form a co-ordinate covalent bond. Example: NH3, H2O, Cl- etc.
An acid is a electron-pair acceptor and a base is electron-pair donor. Any substance which has a lone pair of electron can act as a base and acid is a substance with empty orbital.
A reaction between an acid and a base which result in the formation of co-ordinate covalent bond is called neutralisation reaction.
Types of Lewis acids :
A)Cation of d-block elements.
Example: Cu+2, Fe+2, Ag+ etc.
B)Compounds whose central atom has incomplete octet.
Example: BF3, BCl3, AlCl3 etc.
C)Compounds in which central atom has available d-orbitals and may expand its octet.
Example: SiF4, SnCl4, SF4.
D)Molecules having multiple bonds between atoms of different electro negativities.
Example: SO2, CO2, NO2 etc.
Types of Lewis bases:
A)All anions
Example: Cl-, OH-, CN-, NH2- etc.
B)Molecules with one or more lone pair of electrons.
Example: H2O, NH3, R-OH, R-NH2
C)Molecules with multiple bonds
Example:HC \( \equiv \) CH; H2C \( \equiv \) CH2Examples of neutralisation reaction:
A)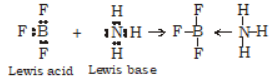
B)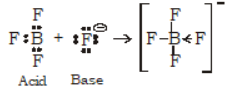
C)