Class 7 / IIT Foundation Plus Chemistry / Electronegative Ions / Monovalent And Bivalent Electronegative Ions
-
ELECTRONEGATIVE IONS
ELECTRONEGATIVE IONS
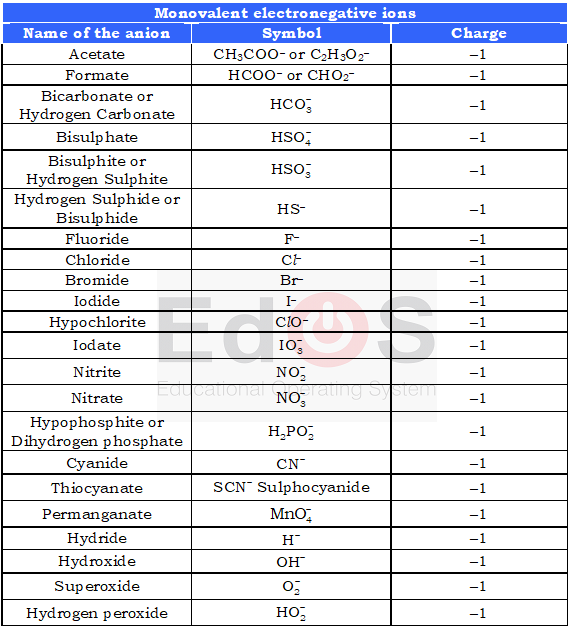
Monovalent negative ions:A radical formed when an atom gains one electron is called monovalent negative ions.
Illustration 2: If in an element X there are seven electrons present in its outer most orbit then it is a ......................
Solution : it is an example of halogens , X=9
electronic configuration = 1s22s22p5
It is a flourine and non metal
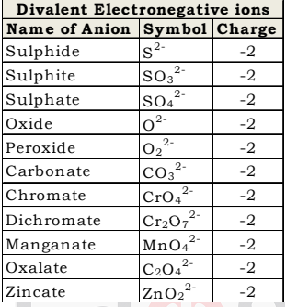
Bivalent Electronegaive ions
A Radical or Ion which is formed by gaining of two electrons is called Bivalent Electronegaive ions