-
Matter and its States
Introduction To Matter
We see different things around us. Trees, flowers and animals are found everywhere in our surrounding. We also see different objects in our homes and schools. There are mountains, rivers, stars and the Moon. We feel air around us. All these things are made up of matter. We can define matter as: Anything which has mass and occupies space is called matter.
Shape and Volume
Some things are big and some are small. A watermelon is bigger than a lemon. Bigger things occupy more space than smaller things. The space occupied by an object is called its volume. Thus, a watermelon has more volume than a lemon (Figure 5.2).
Mass
The amount of matter in an object is called its mass
-
Matter and its States
Three States of Matter
Matter exists in three physical states that are solids, liquids and gases.
Solids
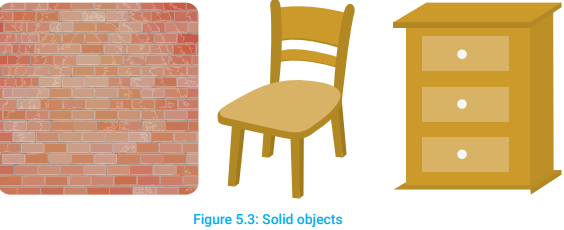
A lot of things have fixed volume and fixed shape. Such things are called solids. Table, chair, book, brick wall, pencil, cup, car, wheat grains, sugar crystals, etc., are the examples of solids (Figure 5.3).
Liquids
Many things have fixed volume but no fixed shape. They take the shape of the container in which they are put. Such things are called liquids. Water, oil, petrol, milk, fruit juices, honey, Figure 5.4: Liquid objects etc. are the examples of liquids (Figure 5.4)
Gases
There are things which have neither fixed shape nor fixed volume. They spread out to fill the container (Figure 5.5). Such things are called gases. Air, smoke, oxygen, carbon dioxide, hydrogen, steam or vapours are the examples of gases.
The properties of solids, liquids and gases regarding their volumes and shapes can be observed through the activities given below.
-
Matter and its States
Effect of Heat on Matter ( Solids, Liquids and Gases )
Things change their state on heating or cooling. Solids melt on heating and change into their liquid states. Liquids boil on heating and change into their gaseous states. Gases expand on heating and increase their pressure in the container. Liquids freeze on cooling and change into their solid states. Gases condense on cooling and change into their liquid states.
Higher Order Thinking
Matter consists of very small particles called atoms and molecules, etc. Molecules can exist independently. These particles of matter are always moving The particles of solids do not change their position during their movement. That is why solids cannot flow or spread and have fixed shape and volume. The particles of liquids have rapid movements as compared to solids. They can change their position during the motion but remain in the liquid. That is why liquids can flow and change their shape but their volume remains fixed. The particles of gases are freely moving fast and spread out to occupy the whole available space. That is why gases have neither fixed shape nor fixed volume.
-
Matter and its States
Mixing of Materials
Most of the things we find in our surroundings are not in their pure form. They are the mixtures of different substances. For example, soil is the mixture of sand, rocks, moisture and air, etc. Milk is a mixture of different food groups, i.e., carbohydrates, proteins, vitamins, fats, salts and water, etc. Similarly, air is a mixture of different gases and water vapours.
A mixture is a sample of matter containing two or more things mixed with each other.
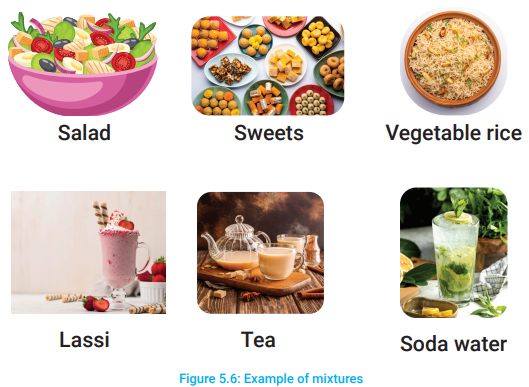
We mix many things to prepare salad. Can you name the things used in salad? We mix many things to make tea. Can you name the things used to make tea? Different things are used to prepare soda water. Can you tell the names of the things used in the preparation of soda water? Sweets, vegetable rice, lassi, yogurt, fruit juices etc., are the mixtures consisting of different substances (Figure 5.6).
-
Matter and its States
Filtration
Filtration is another method of separating the insoluble and suspended components of a mixture. In this method, the mixture is passed through a porous sheet like a filter paper, a cotton cloth or a strainer. Separating tea leaves from tea using a strainer is a well-known example of filtration
-
Matter and its States
Filtration
Filtration is another method of separating the insoluble and suspended components of a mixture. In this method, the mixture is passed through a porous sheet like a filter paper, a cotton cloth or a strainer. Separating tea leaves from tea using a strainer is a well-known example of filtration