-
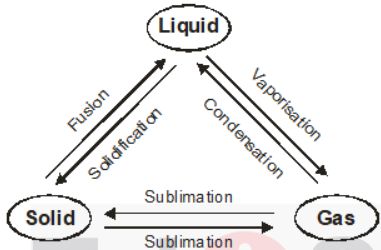
Interconversion Of Matter
EFFECT OF CHANGE OF PRESSURE
The effect of pressure on the states of matter can be discussed by the following experiment.
Experiment : Take a gas in a cylinder and apply pressure by pushing the piston down as shown in the following figure
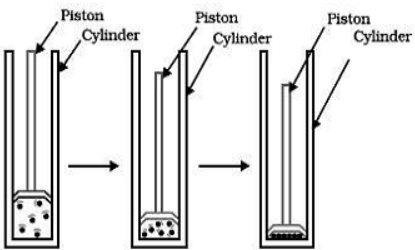
(a) (b) (c)
By applying pressure, particles of a gas come close together
Observation: A gas can be first liquefied and then converted into solid.
Liquification of gas : A gas can be liquefied by applying pressure or by lowering the temperature. For every gas, there is a minimum temperature above which gas cannot be liquefied by applying pressure. This temperature is called “critical temperature”. The minimum pressure which is required to liquefy a gas at critical temperature is called “critical pressure”.
Discussion:When the particles of fluid are present under low pressure, they are in the gaseous state as shown in the figure (a). When some high pressure is applied on the gas, the forces of attraction between gas particles become so high that they bind the gas particles together to form the liquid state [figure (b)]. Ultimately under very high pressure, the forces of attraction become so strong that the liquid may change into the solid state [figure (c)].For example, CO2 gas can be liquefied easily either by applying pressure or by reducing the temperature. However, CO2 is cooled (by reducing temperature) under high pressure, it can be directly converted into solid CO2 called ‘dry ice’.
Solid CO2 is called dry ice, because unlike ordinary ice, dry ice does not wet the surface on which it melts. It is used as a refrigerant. This is because, if pressure on dry ice is reduced to one atmosphere, it directly gets converted into gaseous state without passing through the liquid state. It is because of the reason, that dry ice is stored under high pressure.Conclusion: From above discussion, It is clear that a gas can be liquefied by increasing pressure and decreasing temperature and vice, versa hence, it follows that both pressure and temperature determine the state of a substance, whether, it will be a solid, liquid or gas. The entire change has been represented as below: